Abstract
Introduction
This study was to investigate the correlations between pyrethroid exposure and bone mineral density (BMD) and osteopenia.
Materials and methods
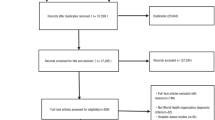
This cross-sectional study included 1389 participants over 50 years of age drawn from the 2007–2010 and 2013–2014 National Health and Nutrition Examination Survey (NHANES). Three pyrethroid metabolites, 3-phenoxybenzoic acid (3-PBA), trans-3-(2,2-dichlorovinyl)-2,2-dimethyl-cyclopropane-1-carboxylic acid (trans-DCCA), and 4-fluoro-3-phenoxybenzoic acid (4-F-3PBA) were used as indicators of pyrethroid exposure. Low BMD was defined as T-score < − 1.0, including osteopenia. Weighted multivariable linear regression analysis or logistic regression analysis was utilized to evaluate the correlation between pyrethroid exposure and BMD and low BMD. Bayesian kernel machine regression (BKMR) model was utilized to analyze the correlation between pyrethroids mixed exposure and low BMD.
Results
There were 648 (48.41%) patients with low BMD. In individual pyrethroid metabolite analysis, both tertile 2 and tertile 3 of trans-DCCA were negatively related to total femur, femur neck, and total spine BMD [coefficient (β) = − 0.041 to − 0.028; all P < 0.05]. Both tertile 2 and tertile 3 of 4-F-3PBA were negatively related to total femur BMD (P < 0.05). Only tertile 2 [odds ratio (OR) = 1.63; 95% CI = 1.07, 2.48] and tertile 3 (OR = 1.65; 95% CI = 1.10, 2.50) of trans-DCCA was correlated with an increased risk of low BMD. The BKMR analysis indicated that there was a positive tendency between mixed pyrethroids exposure and low BMD.
Conclusion
In conclusion, pyrethroids exposure was negatively correlated with BMD levels, and the associations of pyrethroids with BMD and low BMD varied by specific pyrethroids, pyrethroid concentrations, and bone sites.



Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from NHANES database, https://www.cdc.gov/nchs/nhanes/index.htm.
References
Compston JE, McClung MR, Leslie WD (2019) Osteoporosis. Lancet (London, England) 393:364–376. https://doi.org/10.1016/s0140-6736(18)32112-3
Xiao PL, Cui AY, Hsu CJ, Peng R, Jiang N, Xu XH, Ma YG, Liu D, Lu HD (2022) Global, regional prevalence, and risk factors of osteoporosis according to the world health organization diagnostic criteria: a systematic review and meta-analysis. Osteoporos Int: Established as Result of Cooperation Between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA 33:2137–2153. https://doi.org/10.1007/s00198-022-06454-3
Hendrickx G, Boudin E, Van Hul W (2015) A look behind the scenes: the risk and pathogenesis of primary osteoporosis. Nat Rev Rheumatol 11:462–474. https://doi.org/10.1038/nrrheum.2015.48
Khalil N, Chen A, Lee M, Czerwinski SA, Ebert JR, DeWitt JC, Kannan K (2016) Association of perfluoroalkyl substances, bone mineral density, and osteoporosis in the US population in NHANES 2009–2010. Environ Health Perspect 124:81–87. https://doi.org/10.1289/ehp.1307909
Guo J, Huang Y, Bian S, Zhao C, Jin Y, Yu D, Wu X, Zhang D, Cao W, Jing F, Chen G (2018) Associations of urinary polycyclic aromatic hydrocarbons with bone mass density and osteoporosis in US adults, NHANES 2005–2010. Environ Pollut (Barking, Essex : 1987). https://doi.org/10.1016/j.envpol.2018.04.108
Turan S (2021) Endocrine disrupting chemicals and bone. Best Pract Res Clin Endocrinol Metab 35:101495. https://doi.org/10.1016/j.beem.2021.101495
Zhang Y, Dong S, Wang H, Tao S, Kiyama R (2016) Biological impact of environmental polycyclic aromatic hydrocarbons (ePAHs) as endocrine disruptors. Environ Pollut (Barking, Essex : 1987). https://doi.org/10.1016/j.envpol.2016.03.050
Manolagas SC (2013) Steroids and osteoporosis: the quest for mechanisms. J Clin Investig 123:1919–1921. https://doi.org/10.1172/jci68062
Karsenty G (2012) The mutual dependence between bone and gonads. J Endocrinol 213:107–114. https://doi.org/10.1530/joe-11-0452
Xu H, Bo Y (2022) Associations between pyrethroid exposure and serum sex steroid hormones in adults: findings from a nationally representative sample. Chemosphere 300:134591. https://doi.org/10.1016/j.chemosphere.2022.134591
Green MP, Harvey AJ, Finger BJ, Tarulli GA (2021) Endocrine disrupting chemicals: impacts on human fertility and fecundity during the peri-conception period. Environ Res 194:110694. https://doi.org/10.1016/j.envres.2020.110694
Barr DB, Olsson AO, Wong LY, Udunka S, Baker SE, Whitehead RD, Magsumbol MS, Williams BL, Needham LL (2010) Urinary concentrations of metabolites of pyrethroid insecticides in the general U.S. population: national health and nutrition examination survey 1999–2002. Environ Health Perspect 118:742–748. https://doi.org/10.1289/ehp.0901275
Wang J, Lin K, Taylor A, Gan J (2018) In vitro assessment of pyrethroid bioaccessibility via particle ingestion. Environ Int 119:125–132. https://doi.org/10.1016/j.envint.2018.05.043
Cunha SC, Menezes-Sousa D, Mello FV, Miranda JAT, Fogaca FHS, Alonso MB, Torres JPM, Fernandes JO (2022) Survey on endocrine-disrupting chemicals in seafood: occurrence and distribution. Environ Res 210:112886. https://doi.org/10.1016/j.envres.2022.112886
Lehmler HJ, Simonsen D, Liu B, Bao W (2020) Environmental exposure to pyrethroid pesticides in a nationally representative sample of U.S. adults and children: the national health and nutrition examination survey 2007–2012. Environ Pollut (Barking, Essex : 1987) 267:115489. https://doi.org/10.1016/j.envpol.2020.115489
Saillenfait AM, Ndiaye D, Sabaté JP (2015) Pyrethroids: exposure and health effects–an update. Int J Hyg Environ Health 218:281–292. https://doi.org/10.1016/j.ijheh.2015.01.002
Ye X, Liu J (2019) Effects of pyrethroid insecticides on hypothalamic-pituitary-gonadal axis: a reproductive health perspective. Environ Pollut (Barking, Essex : 1987) 245:590–599. https://doi.org/10.1016/j.envpol.2018.11.031
Wang Q, Shen JY, Zhang R, Hong JW, Li Z, Ding Z, Wang HX, Zhang JP, Zhang MR, Xu LC (2020) Effects and mechanisms of pyrethroids on male reproductive system. Toxicology 438:152460. https://doi.org/10.1016/j.tox.2020.152460
CDC, 2020. Laboratory procedure manual-specific organophosphorus pesticides, synthetic pyrethroids, and select herbicides (Universal Pesticides). Available: https://wwwn.cdc.gov/nchs/data/nhanes/2013-2014/labmethods/UPHOPM-H-MET-508.pdf. Accessed 20 Aug 2022
CDC, 2007. National health and nutrition examination survey, dual energy x-ray absorptiometry (DXA) procedures manua. Available. https://wwwn.cdc.gov/nchs/data/nhanes/2007-2008/manuals/manual_dexa.pdf. Accessed 14 Aug 2022
Looker AC, Orwoll ES, Johnston CC Jr, Lindsay RL, Wahner HW, Dunn WL, Calvo MS, Harris TB, Heyse SP (1997) Prevalence of low femoral bone density in older U.S. adults from NHANES III. J Bone Mineral Res: Off J Am Soc Bone Mineral Res 12:1761–1768. https://doi.org/10.1359/jbmr.1997.12.11.1761
Classification and Diagnosis of Diabetes (2020) Standards of medical care in diabetes-2020. Diabetes Care 43:S14-s31. https://doi.org/10.2337/dc20-S002
Whelton PK, Carey RM (2017) The 2017 clinical practice guideline for high blood pressure. JAMA 318:2073–2074. https://doi.org/10.1001/jama.2017.18209
Grundy SM, Stone NJ, Bailey AL, Beam C, Birtcher KK et al (2019) 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the american college of cardiology/american heart association task force on clinical practice guidelines. Circulation 139:e1082–e1143. https://doi.org/10.1161/cir.0000000000000625
Zhang Y, Lu Y, Ma H, Xu Q, Wu X (2021) Combined exposure to multiple endocrine disruptors and uterine leiomyomata and endometriosis in US women. Front Endocrinol 12:726876. https://doi.org/10.3389/fendo.2021.726876
Bopp SK, Barouki R, Brack W, Dalla Costa S, Dorne JCM et al (2018) Current EU research activities on combined exposure to multiple chemicals. Environ Int 120:544–562. https://doi.org/10.1016/j.envint.2018.07.037
Drakvik E, Altenburger R, Aoki Y, Backhaus T, Bahadori T et al (2020) Statement on advancing the assessment of chemical mixtures and their risks for human health and the environment. Environ Int 134:105267. https://doi.org/10.1016/j.envint.2019.105267
Almeida M, Laurent MR, Dubois V, Claessens F, O’Brien CA, Bouillon R, Vanderschueren D, Manolagas SC (2017) Estrogens and androgens in skeletal physiology and pathophysiology. Physiol Rev 97:135–187. https://doi.org/10.1152/physrev.00033.2015
Patel S, Homaei A, Raju AB, Meher BR (2018) Estrogen: The necessary evil for human health, and ways to tame it. Biomed Pharmacother = Biomedecine Pharmacotherapie 102:403–411. https://doi.org/10.1016/j.biopha.2018.03.078
Chakraborty D, Sarkar A, Mann S, Agnihotri P, Saquib M, Malik S, Kumavat R, Mathur A, Biswas S (2022) Estrogen-mediated differential protein regulation and signal transduction in rheumatoid arthritis. J Mol Endocrinol 69:R25-r43. https://doi.org/10.1530/jme-22-0010
Agas D, Lacava G, Sabbieti MG (2018) Bone and bone marrow disruption by endocrine-active substances. J Cell Physiol 234:192–213. https://doi.org/10.1002/jcp.26837
Agas D, Sabbieti MG, Marchetti L (2013) Endocrine disruptors and bone metabolism. Arch Toxicol 87:735–751. https://doi.org/10.1007/s00204-012-0988-y
Wang N, Wang Y, Zhang H, Guo Y, Chen C, Zhang W, Wan H, Han J, Lu Y (2020) Association of bone mineral density with nine urinary personal care and consumer product chemicals and metabolites: a national-representative, population-based study. Environ Int 142:105865. https://doi.org/10.1016/j.envint.2020.105865
Min KB, Min JY (2014) Urinary phthalate metabolites and the risk of low bone mineral density and osteoporosis in older women. J Clin Endocrinol Metab 99:E1997-2003. https://doi.org/10.1210/jc.2014-2279
Wang A, Wan Y, Mahai G, Qian X, Li Y, Xu S, Xia W (2023) Association of prenatal exposure to organophosphate, pyrethroid, and neonicotinoid insecticides with child neurodevelopment at 2 years of age: a prospective cohort study. Environ Health Perspect 131:107011. https://doi.org/10.1289/ehp12097
Bao W, Liu B, Simonsen DW, Lehmler HJ (2020) Association between exposure to pyrethroid insecticides and risk of all-cause and cause-specific mortality in the general US adult population. JAMA Intern Med 180:367–374. https://doi.org/10.1001/jamainternmed.2019.6019
Zepeda-Arce R, Rojas-García AE, Benitez-Trinidad A, Herrera-Moreno JF, Medina-Díaz IM, Barrón-Vivanco BS, Villegas GP, Hernández-Ochoa I, Sólis Heredia MJ, Bernal-Hernández YY (2017) Oxidative stress and genetic damage among workers exposed primarily to organophosphate and pyrethroid pesticides. Environ Toxicol 32:1754–1764. https://doi.org/10.1002/tox.22398
Funding
This research was supported by the Jilin Provincial Scientific and Technological Development Program (No. YDZJ202301ZYTS126).
Author information
Authors and Affiliations
Contributions
ZS and FY designed the study. ZS wrote the manuscript. FZ, XG, ZL, YZ, DZ, RW, QX, WM, RZ and LG collected, analyzed, and interpreted the data. FY critically reviewed, edited, and approved the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethics approval and consent to participate
Protocols of NHANES have been approved by the National Center for Health Statistics Research Review Board. All participants provided written informed consent. This study used publicly available de-identified data and did not require additional ethical approval.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
774_2024_1499_MOESM1_ESM.tif
Supplementary file1 Supplementary Figure 1. Correlations between different pyrethroid metabolites. The closer the absolute value of the correlation coefficient is to 1 the stronger the correlation. Positive values represent positive correlations and negative values represent negative correlations. 3-PBA, 3-phenoxybenzoic acid; trans-DCCA, trans-3-(2,2-dichlorovinyl)-2,2-dimethyl-cyclopropane-1-carboxylic acid; 4-F-3PBA, 4-fluoro-3-phenoxybenzoic acid. (TIF 1026 KB)
774_2024_1499_MOESM2_ESM.tif
Supplementary file2 Supplementary Figure 2. Association between exposure 1 with low BMD (T-score < − 1.0), while fixing exposure 2 at different percentiles (there are the 10th, 50th, and 90th percentiles) and all the others at their median levels. When the three quantile lines (10th, 50th, and 90th percentiles) are parallel, it indicates that there is no interaction between the two corresponding exposures. Model was adjusted for age, gender, race, BMI, parental fracture, and anti-osteoporosis therapy. 3-PBA, 3-phenoxybenzoic acid; trans-DCCA, trans-3-(2,2-dichlorovinyl)-2,2-dimethyl-cyclopropane-1-carboxylic acid; 4-F-3PBA, 4-fluoro-3-phenoxybenzoic acid. (TIF 2354 KB)
774_2024_1499_MOESM3_ESM.tif
Supplementary file3 Supplementary Figure 3. Weight coefficients of each component of the pyrethroid metabolites mixture for low BMD in the quantile-based g computation (qgcomp) model. The longer the column, the greater the effect of the chemical. Model adjusted for age, gender, race, BMI, parental fracture, and anti-osteoporosis therapy. 3-PBA, 3-phenoxybenzoic acid; trans-DCCA, trans-3-(2,2-dichlorovinyl)-2,2-dimethyl-cyclopropane-1-carboxylic acid; 4-F-3PBA, 4-fluoro-3-phenoxybenzoic acid. (TIF 899 KB)
About this article
Cite this article
Shen, Z., Zhang, F., Guan, X. et al. Associations of pyrethroid exposure with bone mineral density and osteopenia in adults. J Bone Miner Metab 42, 242–252 (2024). https://doi.org/10.1007/s00774-024-01499-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-024-01499-2