Abstract
Introduction
Hypophosphatasia (HPP) is a rare inherited disorder, caused by mutations in the alkaline phosphatase (ALPL) gene, which encodes for the tissue non-specific alkaline phosphatase (TNSALP) isoform of alkaline phosphatase (ALP). Adult HPP is one of the mild forms that presents with unspecific signs such as osteopenia, osteomalacia and muscle involvement. Our purpose was to identify and characterize possibly misdiagnosed adult HPP patients at a clinical and biochemical level.
Material and Methods
At the laboratory of Miguel Servet University Hospital we retrospectively reviewed serum ALP levels in adults over a 48-month period. The clinical records of individuals with consistently low ALP levels were reviewed to exclude secondary causes. Those with persistent hypophosphatasemia were screened for symptoms of HPP. The study participants were evaluated at biochemical and genetic levels.
Results
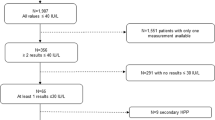
We identified 705 ALP determinations (out of 384,000 processed) in 589 patients below the reference range (30 U/l). Only 21 patients with clinical signs and symptoms of HPP were selected for genetic testing. Finally, only 12 patients participated in the study, 83.3% of whom (10/12) harbored a pathogenic or likely pathogenic variant in a heterozygous state. The major symptoms of our cohort were the presence of musculoskeletal pain (100% of patients) and muscular weakness (83.3% patients).
Conclusion
Mild HPP patients presenting with diffuse symptoms such as musculoskeletal pain may be undiagnosed or misdiagnosed as osteoporosis patients by routine diagnosis. It is important to identify these individuals, to avoid inappropriate treatment with antiresorptive drugs.


Similar content being viewed by others
Availability of data and materials
The study provides full data transparency and availability of data and material. Web resources: ALPL gene variant database: https://alplmutationdatabase.jku.at/. Last accessed April 7th gnomAD variant database: https://gnomad.broadinstitute.org/. Last accessed April 7th EVS database: https://evs.gs.washington.edu /EVS/. Last accessed April 7th.
References
Millán JL, Whyte MP (2016) Alkaline phosphatase and hypophosphatasia. Calcif Tissue Int 98:398. https://doi.org/10.1007/S00223-015-0079-1
Whyte MP (2016) Hypophosphatasia - aetiology, nosology, pathogenesis, diagnosis and treatment. Nat Rev Endocrinol 12:233–246. https://doi.org/10.1038/nrendo.2016.14
Mornet E (2013) Genetics of hypophosphatasia. Clin Rev Bone Miner Metab 11:71–77. https://doi.org/10.1007/s12018-013-9140-7
Mornet E, Taillandier A, Peyramaure S et al (1998) Identification of fifteen novel mutations in the tissue-nonspecific alkaline phosphatase (TNSALP) gene in European patients with severe hypophosphatasia. Eur J Hum Genet 6:308–314. https://doi.org/10.1038/sj.ejhg.5200190
Mornet E (2015) Molecular genetics of hypophosphatasia and phenotype-genotype correlations. Subcell Biochem 76:25–43. https://doi.org/10.1007/978-94-017-7197-9_2
Mornet E, Yvard A, Taillandier A et al (2011) A molecular-based estimation of the prevalence of hypophosphatasia in the european population. Ann Hum Genet 75:439–445. https://doi.org/10.1111/j.1469-1809.2011.00642.x
Mornet E, Taillandier A, Domingues C et al (2020) Hypophosphatasia: a genetic-based nosology and new insights in genotype-phenotype correlation. Eur J Hum Genet. https://doi.org/10.1038/s41431-020-00732-6
Bianchi ML (2015) Hypophosphatasia: an overview of the disease and its treatment. Osteoporos Int 26:2743–2757. https://doi.org/10.1007/s00198-015-3272-1
McKiernan FE, Berg RL, Fuehrer J (2014) Clinical and radiographic findings in adults with persistent hypophosphatasemia. J Bone Miner Res 29:1651–1660. https://doi.org/10.1002/jbmr.2178
Tenorio J, Álvarez I, Riancho-Zarrabeitia L et al (2017) Molecular and clinical analysis of ALPL in a cohort of patients with suspicion of hypophosphatasia. Am J Med Genet Part A 173:601–610. https://doi.org/10.1002/ajmg.a.37991
McKiernan FE, Dong J, Berg RL et al (2017) Mutational and biochemical findings in adults with persistent hypophosphatasemia. Osteoporos Int 28:2343–2348. https://doi.org/10.1007/s00198-017-4035-y
Whyte MP (2017) Hypophosphatasia: an overview for 2017. Bone 102:15–25
Meah F, Basit A, Emanuele N, Emanuele MA (2017) Hypophosphatasia: review of bone mineral metabolism, pathophysiology, clinical presentation, diagnosis, and treatment. Clin Rev Bone Miner Metab 15:24–36. https://doi.org/10.1007/s12018-016-9225-1
Riancho-Zarrabeitia L, García-Unzueta M, Tenorio JA et al (2016) Clinical, biochemical and genetic spectrum of low alkaline phosphatase levels in adults. Eur J Intern Med 29:40–45. https://doi.org/10.1016/J.EJIM.2015.12.019
Whyte MP, Coburn SP, Ryan LM et al (2018) Hypophosphatasia: Biochemical hallmarks validate the expanded pediatric clinical nosology. Bone 110:96–106. https://doi.org/10.1016/J.BONE.2018.01.022
Deeb A, Elfatih A (2018) Could alerting physicians for low alkaline phosphatase levels be helpful in early diagnosis of hypophosphatasia? JCRPE J Clin Res Pediatr Endocrinol 10:19–24. https://doi.org/10.4274/jcrpe.4426
Whyte MP, Rockman-Greenberg C, Ozono K et al (2016) Asfotase alfa treatment improves survival for perinatal and infantile hypophosphatasia. J Clin Endocrinol Metab 101:334–342. https://doi.org/10.1210/jc.2015-3462
Whyte MP, Greenberg CR, Salman NJ et al (2012) Enzyme-replacement therapy in life-threatening hypophosphatasia. N Engl J Med 366:904–913. https://doi.org/10.1056/nejmoa1106173
Cundy T, Michigami T, Tachikawa K et al (2015) Reversible deterioration in hypophosphatasia caused by renal failure with bisphosphonate treatment. J Bone Miner Res 30:1726–1737. https://doi.org/10.1002/jbmr.2495
Richards S, Aziz N, Bale S et al (2015) Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American college of medical genetics and genomics and the association for molecular pathology. Genet Med 17(5):405–424. https://doi.org/10.1038/gim.2015.30
Kircher M, Witten DM, Jain P et al (2014) A general framework for estimating the relative pathogenicity of human genetic variants. Nat Genet 46:310–315. https://doi.org/10.1038/ng.2892
Abecasis GR, Altshuler D, Auton A et al (2010) A map of human genome variation from population-scale sequencing. Nature 467:1061–1073. https://doi.org/10.1038/nature09534
Fauvert D, Brun-Heath I, Lia-Baldini AS et al (2009) Mild forms of hypophosphatasia mostly result from dominant negative effect of severe alleles or from compound heterozygosity for severe and moderate alleles. BMC Med Genet 10:51. https://doi.org/10.1186/1471-2350-10-51
Weber TJ, Sawyer EK, Moseley S et al (2016) Burden of disease in adult patients with hypophosphatasia: results from two patient-reported surveys. Metabolism 65:1522–1530. https://doi.org/10.1016/j.metabol.2016.07.006
Berkseth KE, Tebben PJ, Drake MT et al (2013) Clinical spectrum of hypophosphatasia diagnosed in adults. Bone 54:21–27. https://doi.org/10.1016/j.bone.2013.01.024
Fonta C, Salles J-P (2017) Neuromuscular features of hypophosphatasia. Arch. Pédiatrie 24:5S85-5S88. https://doi.org/10.1016/S0929-693X(18)30021-6
Otero JE, Gottesman GS, McAlister WH et al (2013) Severe skeletal toxicity from protracted etidronate therapy for generalized arterial calcification of infancy. J Bone Miner Res 28:419–430
Fedde KN, Blair L, Silverstein J et al (1999) Alkaline phosphatase knock-out mice recapitulate the metabolic and skeletal defects of infantile hypophosphatasia. J Bone Miner Res 14:2015–2026. https://doi.org/10.1359/jbmr.1999.14.12.2015
López-Delgado L, Riancho-Zarrabeitia L, García-Unzueta MT et al (2018) Abnormal bone turnover in individuals with low serum alkaline phosphatase. Osteoporos Int 29:2147–2150. https://doi.org/10.1007/s00198-018-4571-0
Schmidt T, Mussawy H, Rolvien T et al (2017) Clinical, radiographic and biochemical characteristics of adult hypophosphatasia. Osteoporos Int 28:2653–2662. https://doi.org/10.1007/s00198-017-4087-z
Baron R, Ferrari S, Russell RGG (2011) Denosumab and bisphosphonates: different mechanisms of action and effects. Bone 48:677–692
Roca-Ayats N, Balcells S, Garcia-Giralt N et al (2017) GGPS1 mutation and atypical femoral fractures with bisphosphonates. N Engl J Med 376:1794–1795. https://doi.org/10.1056/NEJMc1612804
Peris P, González-Roca E, Rodríguez-García SC et al (2019) Incidence of mutations in the ALPL, GGPS1, and CYP1A1 genes in patients with atypical femoral fractures. JBMR Plus 3:29–36. https://doi.org/10.1002/jbm4.10064
Alonso N, Larraz-Prieto B, Berg K et al (2019) Loss-of-function mutations in the ALPL gene presenting with adult onset osteoporosis and low serum concentrations of total alkaline phosphatase. J Bone Miner Res. https://doi.org/10.1002/jbmr.3928
Silvent J, Gasse B, Mornet E, Sire J-Y (2014) Molecular evolution of the tissue-nonspecific alkaline phosphatase allows prediction and validation of missense mutations responsible for hypophosphatasia. J Biol Chem 289:24168–24179. https://doi.org/10.1074/jbc.M114.576843
Whyte MP, Zhang F, Wenkert D et al (2015) Hypophosphatasia: validation and expansion of the clinical nosology for children from 25years experience with 173 pediatric patients. Bone 75:229–239. https://doi.org/10.1016/j.bone.2015.02.022
Spentchian M, Brun-Heath I, Taillandier A et al (2006) Characterization of missense mutations and large deletions in the ALPL gene by sequencing and quantitative multiplex PCR of short fragments. Genet Test 10:252–257. https://doi.org/10.1089/gte.2006.10.252
Taillandier A, Domingues C, Dufour A et al (2017) Genetic analysis of adults heterozygous for ALPL mutations. J Bone Miner Metab 1:1–11
del Angel G, Reynders J, Negron C et al (2020) Large-scale in vitro functional testing and novel variant scoring via protein modeling provide insights into alkaline phosphatase activity in hypophosphatasia. Hum Mutat 41:humu.24010. https://doi.org/10.1002/humu.24010
Villa-Suárez JM, García-Fontana C, Andújar-Vera F et al (2021) Hypophosphatasia: a unique disorder of bone mineralization. Int J Mol Sci 22:4303
Funding
Not applicable for that section.
Author information
Authors and Affiliations
Contributions
PC carried out the conception and design and data interpretation, drafting and reviewing the manuscript and approved the final version of the manuscript. CL contributed to data analysis and interpretation, revising the manuscript and approved the final version of the manuscript. MMG contributed to the acquisition and interpretation of data, revising the manuscript and approved the final version of the manuscript. JO contributed to the acquisition and interpretation of data, revising the manuscript and approved the final version of the manuscript. JAP contributed to the acquisition and analysis of data, revising the manuscript and approved the final version of the manuscript. JBA contributed to the acquisition and interpretation of data, revising the manuscript and approved the final version of the manuscript. EG-R contributed to data analysis and interpretation, the drafting and reviewing of the manuscript and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
"Author 1, Author 2, Author 3, Author 4, Author 5, Author 6 and Author 7 declare that they have no conflict of interest." Eva González-Roca (Author 8): has been on the advisory board and received honoraria for lectures from Alexion.
Ethical approval
The study was approved by the Research Ethics Committee of the Autonomous Community of Aragon (CEICA) and all participants provided informed written consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Calmarza, P., Lapresta, C., Martínez García, M. et al. Musculoskeletal pain and muscular weakness as the main symptoms of adult hypophosphatasia in a Spanish cohort: clinical characterization and identification of a new ALPL gene variant. J Bone Miner Metab 41, 654–665 (2023). https://doi.org/10.1007/s00774-023-01440-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-023-01440-z