Abstract
Introduction
A genome-wide association analysis revealed a rheumatoid arthritis (RA)-risk-associated genetic locus on chromosome 9, which contained the tumor necrosis factor receptor-associated factor 1 (TRAF1). However, the detail mechanism by TRAF1 signaled to fibroblast-like synoviocytes (FLSs) apoptosis remains to be fully understood.
Materials and methods
Synovial tissue of 10 RA patients and osteoarthritis patients were obtained during joint replacement surgery. We investigated TRAF1 level and FLSs apoptosis percentage in vivo and elucidated the mechanism involved in the regulation of apoptotic process in vitro.
Results
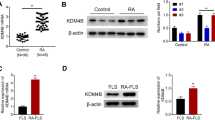
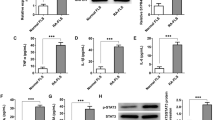
We proved the significant increase of TRAF1 level in FLSs of RA patients and demonstrated that TRAF1 level correlated positively with DAS28 score and negatively with FLSs apoptosis. Treatment with siTRAF1 was able to decrease MMPs levels and the phosphorylated forms of JNK/NF-κB in vitro. Moreover, JNK inhibitor could attenuate expression of MMPs and increase percentage of apoptosis in RA-FLSs, while siTRAF1 could not promote apoptosis when RA-FLSs were pretreated with JNK activator.
Conclusions
High levels of TRAF1 in RA synovium play an important role in the synovial hyperplasia of RA by suppressing apoptosis through activating JNK/NF-kB-dependent signaling pathways in response to the engagement of CD40.




Similar content being viewed by others
References
Nygaard G, Firestein GS (2020) Restoring synovial homeostasis in rheumatoid arthritis by targeting fibroblast-like synoviocytes. Nat Rev Rheumatol 16:316–333. https://doi.org/10.1038/s41584-020-0413-5
Korb-Pap A, Bertrand J, Sherwood J, Pap T (2016) Stable activation of fibroblasts in rheumatic arthritis-causes and consequences. Rheumatology (Oxford) 55:64–67. https://doi.org/10.1093/rheumatology/kew347
Yang Y, Ye Y, Qiu Q, Xiao Y, Huang M, Shi M, Liang L, Yang X, Xu H (2016) Triptolide inhibits the migration and invasion of rheumatoid fibroblast-like synoviocytes by blocking the activation of the JNK MAPK pathway. Int Immuno pharmacol 41:16. https://doi.org/10.1016/j.intimp
Galliqan CL, Siminovitch KA, Keystone EC, Bykerk V, Perez OD, Fish EN (2010) Fibrocyte activation in rheumatoid arthritis. Rheumatology 49:640–651. https://doi.org/10.1093/rheumatology/kep265
Xia ZB, Meng FR, Fang YX, Wu X, Zhang CW, Liu Y, Liu D, Li GQ, Feng FB, Qiu HY (2018) Inhibition of NF-κB signaling pathway induces apoptosis and suppresses proliferation and angiogenesis of human fibroblast-like synovial cells in rheumatoid arthritis. Medicine 97:e10920. https://doi.org/10.1097/MD.0000000000010920
Plenge RM, Seielstad M, Padyukov L, Lee AT, Remmers EF et al (2007) TRAF1-C5 as a risk locus for rheumatoid arthritis-a genome wide study. N Engl J Med 357:1199. https://doi.org/10.1056/NEJMoa073491
Xie P, Hostager BS, Munroe ME, Moore CR, Bishop GA (2006) Cooperation between TNF receptor-associated factors 1 and 2 in CD40 Signaling. Bishop J Immunol 176:5388–5400. https://doi.org/10.4049/jimmunol.176.9.5388
Dunn IF, Sannikova TY, Geha RS, Tsitsikov EN (2000) Identification and characterization of two CD40-inducible enhancers in the mouse TRAF1 gene locus. Mol Immunol 37:961–973. https://doi.org/10.1016/s0161-5890(01)00015-3
Cabal-Hierro L, Rodríguez M, Artime N, Lglesias J, Ugarte L, Prado MA, Lazo PS (2014) TRAF-mediated modulation of NF-kB AND JNK activation by TNFR2. Cell Signal 26:2658–2666. https://doi.org/10.1016/j.cellsig.2014.08.011
McPherson AJ, Snell LM, Mak TW, Watts TH (2012) Opposing roles for TRAF1 in the alternative versus classical NF-κB pathway in T cells. J Biol Chem 29:23010–23019. https://doi.org/10.1074/jbc.M112.350538
Choudhary S, Kalita M, Fang L, Patel KV, Tian B, Zhao Y, Edeh CB, Brasier AR (2013) Inducible TNF receptor associated factor-1 expression couples the canonical to the non-canonical NF-κB Pathway in TNF stimulation. J Biol Chem 288:14612–14623. https://doi.org/10.1074/jbc.M113.464081
Oussa NA, Soumounou Y, Sabbagh L (2013) TRAF1 phosphorylation on Serine 139 modulates NF-κB activity downstream of 4-1BB in T cells. Biochem Biophys Res Commun 432:129–134. https://doi.org/10.1016/j.bbrc.2013.01.073
Bin W, Ming X, Wen-Xia C (2019) TRAF1 meditates lipopolysaccharide-induced acute lung injury by up regulating JNK activation. Biochem Biophys Res Commun 511:49–56. https://doi.org/10.1016/j.bbrc.2019.01.041
Greenfeld H, Takasaki K, Walsh MJ, Ersing I, Bernhardt K, Ma Y, Fu B, Ashbaugh CW, Cabo J, Mollo SB, Zhou H, Li S, Gewurz BE (2015) TRAF1 coordinates polyubiquitin signaling to enhance Epstein-Barr Virus LMP1-mediated growth and survival pathway activation. PLos Pathog 11:e1004890. https://doi.org/10.1371/journal.ppat.1004890
Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS, Healey LA, Kaplan SR, Liang MH, Luthra HS (1988) The American rheumatism association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 31:315–324. https://doi.org/10.1002/art.1780310302
Zhang X, Li W, Zhang X, Zhang X, Jiang L, Guo Y, Wang X (2014) Association between polymorphism in TRAF1/C5 gene and risk of rheumatoid arthritis: a meta-analysis. Mol Biol Rep 41:317–324. https://doi.org/10.1007/s11033-013-2864-0
Van Steenbergen HW, Rodríguez-Rodríguez L, Berglin E, Zhernakova A, Knevel R, Ivorra-Cortés J, Huizinga TW, Fernández-Gutiérrez B, Gregersen PK, Rantapää-Dahlqvist S, van der Helm-van Mil AHM (2015) A genetic study on C5-TRAF1 and progression of joint damage in rheumatoid arthritis. Arthritis Res Ther 17:1. https://doi.org/10.1186/s13075-014-0514-0
Edilova MI, Abdul-Sater AA, Watts TH (2018) TRAF1 signaling in human health and disease. Front Immunol 9:2969. https://doi.org/10.3389/fimmu.2018.02969
Youn J, Kim HY, Park JH, Hwang SH, Lee SY, Cho CS, Lee SK (2002) Regulation of TNF-a-mediated hyperplasia through TNF receptors, TRAFs, and NF-κB in synoviocytes obtained from patients with rheumatoid arthritis. Immunol Lett 83:85–93. https://doi.org/10.1016/s0165-2478(02)00079-2
Nishimoto T, Seta N, Anan R, Yamamoto T, Kaneko Y, Takeuchi T, Kuwana M (2014) A single nucleotide polymorphism of TRAF1 predicts the clinical response to anti-TNF treatment in Japanese patients with rheumatoid arthritis. Clin Exp Rheumatol 32:211–217
Okuda J, Arikawa Y, Takeuchi Y, Mahmoud MM, Suzaki E, Kataoka K, Suzuki T, Okinaka Y, Nakai T (2006) Intracellular replication of Edwardsiella tarda in murine macrophage is dependent on the type III secretion system and induces an up-regulation of anti-apoptotic NF-kappaB target genes protecting the macrophage from staurosporine-induced apoptosis. Microb Pathog 41:226–240. https://doi.org/10.1016/j.micpath.2006.08.002
Dürkop H, Hirsch B, Hahn C, Foss HD, Stein H (2005) Differential expression and function of A20 and TRAF1 in Hodgkin lymphoma and anaplastic large cell lymphoma and their induction by CD30 stimulation. Anticancer Res 25:2367–2379. https://doi.org/10.1002/path.1351
Lu Y-Y, Li Z-Z, Jiang D-S, Wang L, Zhang Y, Chen K, Zhang X-F, Liu Y, Fan G-C, Chen Y, Yang Q, Zhou Y, Zhang X-D, Liu D-P, Li H (2013) TRAF1 is a critical regulator of cerebral ischaemia-reperfusion injury and neuronal death. Nat Commun 4:2852. https://doi.org/10.1038/ncomms3852
Zhang XF, Zhang R, Huang L, Wang PX, Zhang Y, Jiang DS, Zhu LH, Tian S, Zhang XD, Li H (2014) TRAF1 is a key mediator for hepatic ischemia/reperfusion injury. Cell Death Dis 5:e1467. https://doi.org/10.1038/cddis.2014.411
Dunn IF, Geha RS, Tsitsikov EN (1999) Structure of the murine TRAF1 gene. Mol Immunol 36:611–617
Wang CY, Mayo MW, Korneluk RG, Goeddel DV, Baldwin AS Jr (1988) NF-κB antiapoptosis: Induction of TRAF-1 and TRAF-2 and cIAP2 to suppress caspase-8 activation. Science 281:1680–1683
Duckett CS, Gedrich RW, Gilfillan MC, Thompson CB (1997) Induction of NF-kB by the CD30 receptor is mediated by TRAF1 and TRAF2. Mol Cell Biol 17:1535–1542. https://doi.org/10.1128/MCB.17.3.1535
Schwenzer R, Siemienski K, Liptay S, Schuber G, Peters N, Scheurich P, Schmid RM, Wajant H (1999) The human tumor necrosis factor (TNF) receptor-associated factor 1 gene (TRAF1) is up-regulated by cytokines of the TNF ligand family and modulates TNF-induced activation of NF-kB and c-Jun N-terminal kinase. J Biol Chem Actions Search 274:19368–19374. https://doi.org/10.1074/jbc.274.27.19368
Guma M, Firestein GS (2012) c-Jun N-terminal kinase in inflammation and rheumatic diseases. Open Rheumatol J 6:220–231. https://doi.org/10.2174/1874312901206010220
Cheng T, Wang M, Chen L, Guo Y, Chen Z, Wu J (2018) Increased expression of CD40/TRAF1 and activation of nuclear factor-κB-dependent proinflammatory gene expression in collagen-induced arthritis. Scand J Rheumatol 47:455–460. https://doi.org/10.1080/03009742.2018.1432684
Yamamoto H, Ryu J, Min E, Oi N, Bai R, Zykova TA, Yu DH, Moriyama K, Bode AM, Dong Z (2017) TRAF1 Is Critical for DMBA/Solar UVR-Induced Skin Carcinogenesis. J Invest Dermatol 137:1322–1332. https://doi.org/10.1016/j.jid.2016.12.026
Xu W, Zhang L, Zhang Y, Zhang K, Wu Y, Jin D (2019) TRAF1 Exacerbates Myocardial Ischemia Reperfusion Injury via ASK1-JNK/p38 Signaling. J Am Heart Assoc 8:e012575. https://doi.org/10.1161/JAHA.119.012575
Xiang M, Wang PX, Wang AB, Zhang XJ, Zhang Y, Zhang P, Mei FH, Chen MH, Li H (2016) Targeting hepatic TRAF1-ASK1 signaling to improve inflammation, insulin resistance, and hepatic steatosis. J Hepatol 64:1365–1377. https://doi.org/10.1016/j.jhep.2016.02.002
Eeva J, Ropponen A, Nuutinen U, Eeva ST, Mättö M, Eray M, Pelkonen J (2007) The CD40-induced protection against CD95-mediated apoptosis is associated with a rapid upregulation of anti-apoptotic c-FLIP. Mol Immunol 44:1230–1237. https://doi.org/10.1016/j.molimm.2006.06.005
Wang D, Freeman GJ, Levine H, Ritz J, Robertson MJ (1997) Role of the CD40 and CD95 (APO-1/Fas) antigens in the apoptosis of human B-cell malignancies. Br J Haematol 97:409–417. https://doi.org/10.1046/j.1365-2141.1997.422688.x
Fang W, Nath KA, Mackey MF, Noelle RJ, Mueller DL, Behrens TW (1997) CD40 inhibits B cell apoptosis by upregulating bcl-xL expression and blocking oxidant accumulation. Am J Physio 272:C950-956. https://doi.org/10.1152/ajpcell.1997.272.3.C950
Afford SC, Ahmed-Choudhury J, Randhawa S, Russell C, Youster J, Crosby HA, Eliopoulos A, Hubscher SG, Young LS, Adams DH (2001) CD40 activation-induced, Fas-dependent apoptosis and NF-kappaB/AP-1 signaling in human intrahepatic biliary epithelial cells. FASEB J 15:2345–2354. https://doi.org/10.1096/fj.01-0088com
Longo CR, Arvelo MB, Patel VI, Daniel S, Mahiou J, Grey ST, Ferran C (2003) A20 protects from CD40-CD40 ligand-mediated endothelial cell activation and apoptosis. Circulation 108:1113–1118. https://doi.org/10.1161/01.CIR.0000083718.76889.D0
Villarroel Dorrego M, Whawell SA, Speight PM, Barrett AW (2006) Transfection and ligation of CD40 in human oral keratinocytes affect proliferation, adhesion and migration but not apoptosis in vitro. Clin Exp Dermatol 31:266–271. https://doi.org/10.1111/j.1365-2230.2005.02018.x
Kim L-H, Eow G-I, Peh SC, Poppema S (2003) The role of CD30, CD40 and CD95 in the regulation of proliferation and apoptosis in classical Hodgkin’s lymphoma. Pathology 35:428–435. https://doi.org/10.1080/00313020310001602567
Funding
This work was supported by Suzhou Minsheng science and technology project (SYS2020108, SYS2019043).
Author information
Authors and Affiliations
Contributions
TC and JW participated in study design, data collection, data analysis, data interpretation, and drafting the paper. MW, YX, CL and HZ participated in patient recruitment, experiments, and data collection. TC and MW supervised the whole research, designed the study, interpreted the data, and wrote the paper. All the authors contributed to the article and approved the submitted version.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent for publication
All the authors agree to publish.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Cheng, T., Wu, J., Xu, Y. et al. CD40/TRAF1 decreases synovial cell apoptosis in patients with rheumatoid arthritis through JNK/NF-κB pathway. J Bone Miner Metab 40, 819–828 (2022). https://doi.org/10.1007/s00774-022-01350-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-022-01350-6