Abstract
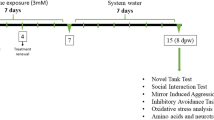
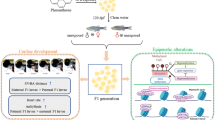
An increase in plasma l-methionine (Met) levels, even if transitory, can cause important toxicological alterations in the affected individuals. Met is essential in the regulation of epigenetic mechanisms and its influence on the subsequent generation has been investigated. However, few studies have explored the influence of a temporary increase in Met levels in parents on their offspring. This study evaluated the behavioral and neurochemical effects of parental exposure to high Met concentration (3 mM) in zebrafish offspring. Adult zebrafish were exposed to Met for 7 days, maintained for additional 7 days in tanks that contained only water, and then used for breeding. The offspring obtained from these fish (F1) were tested in this study. During the early stages of offspring development, morphology, heart rate, survival, locomotion, and anxiety-like behavior were assessed. When these animals reached the adult stage, locomotion, anxiety, aggression, social interaction, memory, oxidative stress, and levels of amino acids and neurotransmitters were analyzed. F1 larvae Met group presented an increase in the distance and mean speed when compared to the control group. F1 adult Met group showed decreased anxiety-like behavior and locomotion. An increase in reactive oxygen species was also observed in the F1 adult Met group whereas lipid peroxidation and antioxidant enzymes did not change when compared to the control group. Dopamine, serotonin, glutamate, and glutathione levels were increased in the F1 adult Met group. Taken together, our data show that even a transient increase in Met in parents can cause behavioral and neurochemical changes in the offspring, promoting transgenerational effects.









Similar content being viewed by others
Availability of data and material
Data will be made available on reasonable request.
Code availability
Not applicable.
References
Aebi H (1984) Oxygen radicals in biological systems. Methods Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Altenhofen S, Nabinger DD, Wiprich MT et al (2017a) Tebuconazole alters morphological, behavioral and neurochemical parameters in larvae and adult zebrafish (Danio rerio). Chemosphere 180:483–490. https://doi.org/10.1016/j.chemosphere.2017.04.029
Altenhofen S, Wiprich MT, Nery LR et al (2017b) Manganese(II) chloride alters behavioral and neurochemical parameters in larvae and adult zebrafish. Aquat Toxicol 182:172–183. https://doi.org/10.1016/j.aquatox.2016.11.013
Barbazuk WB, Korf I, Kadavi C et al (2000) The syntenic relationship of the zebrafish and human genomes. Genome Res 10:1351–1358. https://doi.org/10.1101/gr.144700
Bevilaqua F, Sachett A, Chitolina R et al (2020) A mixture of fipronil and fungicides induces alterations on behavioral and oxidative stress parameters in zebrafish. Ecotoxicology 29:140–147. https://doi.org/10.1007/s10646-019-02146-7
Blank M, Guerim LD, Cordeiro RF, Vianna MRM (2009) A one-trial inhibitory avoidance task to zebrafish: rapid acquisition of an NMDA-dependent long-term memory. Neurobiol Learn Mem 92:529–534. https://doi.org/10.1016/j.nlm.2009.07.001
Blumfield ML, Collins CE (2014) High-protein diets during pregnancy: healthful or harmful for offspring? Am J Clin Nutr 100:993–995. https://doi.org/10.3945/ajcn.114.096511
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Brisch R, Saniotis A, Wolf R et al (2014) The role of dopamine in schizophrenia from a neurobiological and evolutionary perspective: old fashioned, but still in vogue. Front Psychiatry 5:47. https://doi.org/10.3389/fpsyt.2014.00047
Bröer S, Bröer A (2017) Amino acid homeostasis and signalling in mammalian cells and organisms. Biochem J 474:1935–1963. https://doi.org/10.1042/BCJ20160822
Burdge GC, Lillycrop KA (2010) Nutrition, epigenetics, and developmental plasticity: implications for understanding human disease. Annu Rev Nutr 30:315–339. https://doi.org/10.1146/annurev.nutr.012809.104751
Capiotti KM, Menezes FP, Nazario LR et al (2011) Early exposure to caffeine affects gene expression of adenosine receptors, DARPP-32 and BDNF without affecting sensibility and morphology of developing zebrafish (Danio rerio). Neurotoxicol Teratol 33:680–685. https://doi.org/10.1016/j.ntt.2011.08.010
Capiotti KM, Fazenda L, Nazario LR et al (2013) Arginine exposure alters ectonucleotidase activities and morphology of zebrafish larvae (Danio rerio). Int J Dev Neurosci 31:75–81. https://doi.org/10.1016/j.ijdevneu.2012.09.002
Chandrashekar V, Leathem JH (1977) Effect of excess dietary methionine on rat pregnancy: influence on ovarian Δ5 3β hydroxysteroid dehydrogenase activity. Fertil Steril 28:590–593. https://doi.org/10.1016/S0015-0282(16)42563-X
Chang CA, Wei X-C, Martin SR et al (2019) Transiently elevated plasma methionine, S-adenosylmethionine and S-adenosylhomocysteine: Unreported laboratory findings in a patient with NGLY1 deficiency, a congenital disorder of deglycosylation. JIMD Rep 49:21–29. https://doi.org/10.1002/jmd2.12064
Colwill RM, Creton R (2011) Imaging escape and avoidance behavior in zebrafish larvae. Rev Neurosci 22:63–73. https://doi.org/10.1515/RNS.2011.008
Cools R, D’Esposito M (2011) Inverted-U–shaped dopamine actions on human working memory and cognitive control. Biol Psychiatry 69:e113–e125. https://doi.org/10.1016/j.biopsych.2011.03.028
de Lima DSC, Francisco ES, Lima CB, Guedes RCA (2017) Neonatal l-glutamine modulates anxiety-like behavior, cortical spreading depression, and microglial immunoreactivity: analysis in developing rats suckled on normal size- and large size litters. Amino Acids 49:337–346. https://doi.org/10.1007/s00726-016-2365-2
Elango R, Ball RO (2016) Protein and amino acid requirements during pregnancy. Adv Nutr 7:839S-844S. https://doi.org/10.3945/an.115.011817
Gaiday AN, Tussupkaliyev AB, Bermagambetova SK et al (2018) Effect of homocysteine on pregnancy: a systematic review. Chem Biol Interact 293:70–76. https://doi.org/10.1016/j.cbi.2018.07.021
Gerlai R, Lahav M, Guo S, Rosenthal A (2000) Drinks like a fish: Zebra fish (Danio rerio) as a behavior genetic model to study alcohol effects. Pharmacol Biochem Behav 67:773–782. https://doi.org/10.1016/S0091-3057(00)00422-6
Green LC, Wagner DA, Glogowski J et al (1982) Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal Biochem 126:131–138. https://doi.org/10.1016/0003-2697(82)90118-x
Gulsun M, Oznur T, Aydemir E et al (2016) Possible relationship between amino acids, aggression and psychopathy. Int J Psychiatry Clin Pract 20:91–100. https://doi.org/10.3109/13651501.2016.1144771
Hassan TH, Abdelrahman HM, Abdel Fattah NR et al (2013) Blood and brain glutamate levels in children with autistic disorder. Res Autism Spectr Disord 7:541–548. https://doi.org/10.1016/j.rasd.2012.12.005
Horzmann KA, Freeman JL (2018) Making waves: new developments in toxicology with the zebrafish. Toxicol Sci 163:5–12. https://doi.org/10.1093/toxsci/kfy044
Hrncic D, Mikić J, Rasic-Markovic A et al (2016) Anxiety-related behavior in hyperhomocysteinemia induced by methionine nutritional overload in rats: Role of the brain oxidative stress. Can J Physiol Pharmacol 94:1074–1082. https://doi.org/10.1139/cjpp-2015-0581
Le Floc’h N, Otten W, Merlot E (2011) Tryptophan metabolism, from nutrition to potential therapeutic applications. Amino Acids 41:1195–1205. https://doi.org/10.1007/s00726-010-0752-7
LeBel CP, Ali SF, McKee M, Bondy SC (1990) Organometal-induced increases in oxygen reactive species: The potential of 2′,7′-dichlorofluorescin diacetate as an index of neurotoxic damage. Toxicol Appl Pharmacol 104:17–24. https://doi.org/10.1016/0041-008X(90)90278-3
Lee US, Cui J (2010) BK channel activation: structural and functional insights. Trends Neurosci 33:415–423. https://doi.org/10.1016/j.tins.2010.06.004
Legind CS, Broberg BV, Mandl RCW et al (2019) Heritability of cerebral glutamate levels and their association with schizophrenia spectrum disorders: a 1[H]-spectroscopy twin study. Neuropsychopharmacology 44:581–589. https://doi.org/10.1038/s41386-018-0236-0
Lowry OH, Rosebrough A, Farr L, Randall RJ (1994) Protein measurement with the folin phenol reagent. Anal Biochem 217:220–230. https://doi.org/10.1016/0304-3894(92)87011-4
Lutte AH, Capiotti KM, da Silva NLG et al (2015) Contributions from extracellular sources of adenosine to the ethanol toxicity in zebrafish larvae. Reprod Toxicol 53:82–91. https://doi.org/10.1016/j.reprotox.2015.04.001
Macchi FS, Pissinate K, Villela AD et al (2018) 1H-Benzo[d]imidazoles and 3,4-dihydroquinazolin-4-ones: design, synthesis and antitubercular activity. Eur J Med Chem 155:153–164. https://doi.org/10.1016/j.ejmech.2018.06.005
Manta-Vogli PD, Schulpis KH, Dotsikas Y, Loukas YL (2020) The significant role of amino acids during pregnancy: nutritional support. J Matern Neonatal Med 33:334–340. https://doi.org/10.1080/14767058.2018.1489795
Marklund S, Marklund G (1974) Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem 47:469–474. https://doi.org/10.1111/j.1432-1033.1974.tb03714.x
Martínez Y, Li X, Liu G et al (2017) The role of methionine on metabolism, oxidative stress, and diseases. Amino Acids 49:2091–2098. https://doi.org/10.1007/s00726-017-2494-2
Miao L, St Clair DK (2009) Regulation of superoxide dismutase genes: implications in disease. Free Radic Biol Med 47:344–356. https://doi.org/10.1016/j.freeradbiomed.2009.05.018
Mocelin R, Marcon M, da Rosa Araujo AS et al (2019) Withdrawal effects following repeated ethanol exposure are prevented by N-acetylcysteine in zebrafish. Prog Neuro Psychopharmacol Biol Psychiatry 93:161–170. https://doi.org/10.1016/j.pnpbp.2019.03.014
Moriyama TS, Felicio AC, Chagas MHN et al (2011) Increased dopamine transporter density in Parkinson’s disease patients with social anxiety disorder. J Neurol Sci 310:53–57. https://doi.org/10.1016/j.jns.2011.06.056
Mudd SH (2011) Hypermethioninemias of genetic and non-genetic origin: a review. Am J Med Genet Part C Semin Med Genet 157:3–32. https://doi.org/10.1002/ajmg.c.30293
Nabinger DD, Altenhofen S, Bitencourt PER et al (2018) Nickel exposure alters behavioral parameters in larval and adult zebrafish. Sci Total Environ 624:1623–1633. https://doi.org/10.1016/j.scitotenv.2017.10.057
Ocal P, Ersoylu B, Cepni I et al (2012) The association between homocysteine in the follicular fluid with embryo quality and pregnancy rate in assisted reproductive techniques. J Assist Reprod Genet 29:299–304. https://doi.org/10.1007/s10815-012-9709-y
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358. https://doi.org/10.1016/0003-2697(79)90738-3
Penas C, Navarro X (2018) Epigenetic modifications associated to neuroinflammation and neuropathic pain after neural trauma. Front Cell Neurosci 12:158
Prick BW, Hop WCJ, Duvekot JJ (2011) Maternal phenylketonuria and hyperphenylalaninemia in pregnancy: pregnancy complications and neonatal sequelae in untreated and treated pregnancies. Am J Clin Nutr 95:374–382. https://doi.org/10.3945/ajcn.110.009456
Quintana AM, Yu HC, Brebner A et al (2017) Mutations in THAP11 cause an inborn error of cobalamin metabolism and developmental abnormalities. Hum Mol Genet 26:2838–2849. https://doi.org/10.1093/hmg/ddx157
Raffaelli G, Saviane C, Mohajerani MH et al (2004) BK potassium channels control transmitter release at CA3–CA3 synapses in the rat hippocampus. J Physiol 557:147–157. https://doi.org/10.1113/jphysiol.2004.062661
Rao SD, Yin HZ, Weiss JH (2003) Disruption of glial glutamate transport by reactive oxygen species produced in motor neurons. J Neurosci 23:2627–2633. https://doi.org/10.1523/JNEUROSCI.23-07-02627.2003
Rees WD, Wilson FA, Maloney CA (2006) Sulfur amino acid metabolism in pregnancy: the impact of methionine in the maternal diet. J Nutr 136:1701S-1705S. https://doi.org/10.1093/jn/136.6.1701S
Ryan DP, Henzel KS, Pearson BL et al (2018) A paternal methyl donor-rich diet altered cognitive and neural functions in offspring mice. Mol Psychiatry 23:1345–1355. https://doi.org/10.1038/mp.2017.53
Sarter M, Bruno JP, Parikh V (2007) Abnormal neurotransmitter release underlying behavioral and cognitive disorders: toward concepts of dynamic and function-specific dysregulation. Neuropsychopharmacology 32:1452–1461. https://doi.org/10.1038/sj.npp.1301285
Savio LEB, Vuaden FC, Kist LW et al (2013) Proline-induced changes in acetylcholinesterase activity and gene expression in zebrafish brain: reversal by antipsychotic drugs. Neuroscience 250:121–128. https://doi.org/10.1016/j.neuroscience.2013.07.004
Schweinberger BM, Wyse ATS (2016) Mechanistic basis of hypermethioninemia. Amino Acids 48:2479–2489. https://doi.org/10.1007/s00726-016-2302-4
Schweinberger BM, Schwieder L, Scherer E et al (2014) Development of an animal model for gestational hypermethioninemia in rat and its effect on brain Na+, K+-ATPase/Mg2+-ATPase activity and oxidative status of the offspring. Metab Brain Dis 29:153–160. https://doi.org/10.1007/s11011-013-9451-x
Schweinberger BM, Rodrigues AF, Turcatel E et al (2017) Maternal hypermethioninemia affects neurons number, neurotrophins levels, energy metabolism, and Na+, K+-ATPase expression/content in brain of rat offspring. Mol Neurobiol 55:980–988. https://doi.org/10.1007/s12035-017-0383-z
Schweinberger BM, Rodrigues AF, dos Santos TM et al (2018) Methionine administration in pregnant rats causes memory deficit in the offspring and alters ultrastructure in brain tissue. Neurotox Res 33:239–246. https://doi.org/10.1007/s12640-017-9830-x
Shioda K, Nisijima K, Yoshino T, Kato S (2004) Extracellular serotonin, dopamine and glutamate levels are elevated in the hypothalamus in a serotonin syndrome animal model induced by tranylcypromine and fluoxetine. Prog Neuro Psychopharmacol Biol Psychiatry 28:633–640. https://doi.org/10.1016/j.pnpbp.2004.01.013
Soares MSP, Viau CM, Saffi J et al (2017) Acute administration of methionine and/or methionine sulfoxide impairs redox status and induces apoptosis in rat cerebral cortex. Metab Brain Dis 32:1693–1703. https://doi.org/10.1007/s11011-017-0054-9
Stefanello FM, Matté C, Scherer EB et al (2007a) Chemically induced model of hypermethioninemia in rats. J Neurosci Methods 160:1–4. https://doi.org/10.1016/j.jneumeth.2006.07.029
Stefanello FM, Scherer EB, Kurek AG et al (2007b) Effect of hypermethioninemia on some parameters of oxidative stress and on Na(+), K (+)-ATPase activity in hippocampus of rats. Metab Brain Dis 22:172–182. https://doi.org/10.1007/s11011-007-9052-7
Steinberg EE, Janak PH (2013) Establishing causality for dopamine in neural function and behavior with optogenetics. Brain Res 1511:46–64. https://doi.org/10.1016/j.brainres.2012.09.036
Tang S, Fang Y, Huang G et al (2017) Methionine metabolism is essential for SIRT1-regulated mouse embryonic stem cell maintenance and embryonic development. EMBO J. https://doi.org/10.15252/embj.201796708
Tsuchiyama A, Oyanagi K, Nakata F et al (1982) A new type of hypermethioninemia in neonates. Tohoku J Exp Med 138:281–288. https://doi.org/10.1620/tjem.138.281
Vuaden FC, Savio LEB, Piato AL et al (2012) Long-term methionine exposure induces memory impairment on inhibitory avoidance and alters acetylcholinesterase activity and expression in Zebrafish (Danio rerio). Neurochem Res 37:1545–1553. https://doi.org/10.1007/s11064-012-0749-6
Vuaden FC, Savio LEB, Rico EP et al (2016) Methionine exposure alters glutamate uptake and adenine nucleotide hydrolysis in the zebrafish brain. Mol Neurobiol 53:200–209. https://doi.org/10.1007/s12035-014-8983-3
Wager K, Mahmood F, Russell C (2014) Modelling inborn errors of metabolism in zebrafish. J Inherit Metab Dis 37:483–495. https://doi.org/10.1007/s10545-014-9696-5
Westerfield M (2007) The Zebrafish Book, A guide for the laboratory use of zebrafish (Danio rerio), 5th edn. University of Oregon Press, Eugene
Yu M, Guo L, Li N et al (2018) Overexpression of Kcnmb2 in dorsal CA1 of offspring mice rescues hippocampal dysfunction caused by a methyl donor-rich paternal diet. Front Cell Neurosci 12:360
Zanandrea R, Abreu MSMS, Piato A et al (2018) Lithium prevents scopolamine-induced memory impairment in zebrafish. Neurosci Lett 664:34–37. https://doi.org/10.1016/j.neulet.2017.11.010
Zanandrea R, Wiprich MT, Altenhofen S et al (2020) Withdrawal effects following methionine exposure in adult zebrafish. Mol Neurobiol. https://doi.org/10.1007/s12035-020-01970-x
Zhang N (2018) Role of methionine on epigenetic modification of DNA methylation and gene expression in animals. Anim Nutr 4:11–16. https://doi.org/10.1016/j.aninu.2017.08.009
Funding
This study was funded in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—finance code 001, Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (Proc. 420695/2018-4), Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (Proc. 17/2551-0000977-0), and Instituto Nacional de Ciências e Tecnologia para Doenças Cerebrais, Excitotoxicidade e Neuroproteção. R.Z (133202/2018-6) and C.D.B. (Proc. 304450/2019-7) were the recipients of a fellowship from CNPq.
Author information
Authors and Affiliations
Contributions
Conceptualization: RZ, CDB; Data Curation: RZ; Methodology: RZ; Formal analysis and investigation: RZ, MTW, SA, GR, TMdS; Writing—original draft preparation: RZ; Writing—review and editing: RZ; ATSW; CDB; Funding acquisition: CDB; Resources: ATSW, CDB; Supervision: CDB.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethics approval
This study was approved by the Comissão de Ética no Uso de Animais of Pontifícia Universidade Católica do Rio Grande do Sul (3758-CEUA-PUCRS) and was registered in the Sistema Nacional de Gestão do Patrimônio Genético e Conhecimento Tradicional Associado—SISGEN (Protocol No. A3B073D).
Additional information
Handling editor: J. G. López.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zanandrea, R., Wiprich, M.T., Altenhofen, S. et al. Paternal exposure to excessive methionine altered behavior and neurochemical activities in zebrafish offspring. Amino Acids 53, 1153–1167 (2021). https://doi.org/10.1007/s00726-021-03019-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-021-03019-2