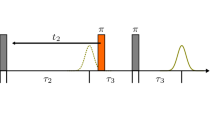
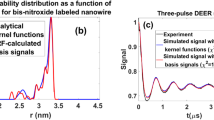
Abstract
DEER (Double Electron–Electron Resonance) kernel signals are calculated, using the double rotating-frames (DRF) technique, for the various distances, r, between two coupled Gd3+ ions, distributed randomly in a biological system. This is done numerically, using full diagonalization of the spin-Hamiltonian matrix, taking into account the zero-field splitting of the Gd3+ion with spin \(S=7/2\). These kernel signals are then used to estimate the probabilities of the distance distribution, P(r), between the pairs of various Gd3+ ions. This is accomplished using Tikhonov regularization, as implemented in the software DeerAnalysis [Jeschke et al. Appl Magn Reson 30(3): 473–498, (2006)], with the kernel signals calculated here by the DRF technique for different r values. This procedure is successfully illustrated by applying it to calculate the probabilities, P(r) from the reported experimental four-pulse DEER data for a sample of Gd ruler 15 in D2O/glycerol-d8 (i) at Q-band [Doll et al. J Magn Reson 259: 153–162, 2015] and (ii) at W-band [Dalaloyan et al. Phys Chem Chem Phys 17(28): 18464–18476, (2015)]. These results are found to be about the same, within the RMSD maximum and minimum bounds, from those obtained using analytical kernel signals, valid for hard (infinite) pulses for spin ½, so that the ZFS is not included, at the lower frequency (~ 35 GHz) of Q-band. However, at the higher frequency of W-band (~ 95 GHz), there are found slight, but distinct, differences beyond the RMSD maximum/minimum bounds, in the P(r) values calculated by the two approaches. In application scenarios to the commonly investigated proteins, where the errors are large, the results obtained by the two approaches would be about the same, at least for the data obtained at Q- and W-bands, if not at higher frequencies.





Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon a reasonable request.
Notes
To this end, the cubic spline software in Matlab was used with the command s =spline(x,y,xq), which returns a vector of interpolated values s corresponding to the query points in xq, where s is the interpolated signal at each point in time-domain range and xq are the r values corresponding to the range of non-zero P(r) in the distance distribution curve. The values of s are determined by cubic-spline interpolation of x and y, which are vectors with the values of 19 r-values for which the time-domain signals were calculated using the DRF technique and the signal for those r values at each point in the time-domain range, respectively. Use of cubic spline saves enormous computational time.
References
S.K. Misra, Chap. 7. Spin-Hamiltonians and site symmetries for transition ions, pp. 327–384; S. K. Misra and J. H. Freed, Chapter 12, Distance measurements: continuous wave (CW) and pulsed dipolar EPR, pp. 588, in Multifrequency Electron Paramagnetic Resonance: Theory and Applications. ed. by S.K. Misra (Wiley-VCH, Weinheim, 2011), pp. 545–597
S.K. Misra, P.P. Borbat, J.H. Freed, Calculation of double-quantum-coherence two-dimensional spectra: distance measurements and orientational correlations. Appl. Magn. Reson. 36, 237 (2009)
A. Dalaloyan, M. Qi, S. Ruthstein, S. Vega, A. Godt, A. Feintuch, D. Goldfarb, Gd (III)–Gd (III) EPR distance measurements–the range of accessible distances and the impact of zero field splitting. Phys. Chem. Chem. Phys. 17(28), 18464–18476 (2015)
A.M. Raitsimring, C. Gunanathan, A. Potapov, I. Efremenko, J.M. Martin, D. Milstein, D. Goldfarb, Gd3+ complexes as potential spin labels for high field pulsed EPR distance measurements. J. Am. Chem. Soc. 129(46), 14138–14139 (2007)
A. Potapov, H. Yagi, T. Huber, S. Jergic, N.E. Dixon, G. Otting, D. Goldfarb, Nanometer-scale distance measurements in proteins using Gd3+ spin labeling. J. Am. Chem. Soc. 132, 9040 (2010)
H. Yagi, D. Banerjee, B. Graham, T. Huber, D. Goldfarb, G. Otting, Gadolinium tagging for high-precision measurements of 6 nm distances in protein assemblies by EPR. J. Am. Chem. Soc. 133, 10418 (2011)
D.T. Edwards, T. Huber, S. Hussain, K.M. Stone, M. Kinnebrew, I. Kaminker, E. Matalon, M.S. Sherwin, D. Goldfarb, S. Han, Determining the oligomeric structure of proteorhodopsin by Gd3+-based pulsed dipolar spectroscopy of multiple distances. Structure 22, 1677 (2014)
A. Doll, M. Qi, N. Wili, S. Pribitzer, A. Godt, G. Jeschke, Gd (III)–Gd (III) distance measurements with chirp pump pulses. J. Magn. Reson. 259, 153–162 (2015)
Y. Song, T. Meade, A. Astashkin, E. Klein, J. Enemark, A. Raitsimring, Pulsed dipolar spectroscopy distance measurements in biomacromolecules labeled with Gd (III) markers. J. Magn. Reson. 210, 59 (2011)
F. Wojciechowski, A. Groß, I.T. Holder, L. Knorr, M. Drescher, J.S. Hartig, Pulsed EPR spectroscopy distance measurements of DNA internally labelled with Gd3+-DOTA. Chem. Commun. 51, 13850 (2015)
M. Gordon-Grossman, I. Kaminker, Y. Gofman, Y. Shai, D. Goldfarb, W-Band pulse EPR distance measurements in peptides using Gd3+–dipicolinic acid derivatives as spin labels. Phys. Chem. Chem. Phys. 13, 10771 (2011)
E. Matalon, T. Huber, G. Hagelueken, B. Graham, V. Frydman, A. Feintuch, G. Otting, D. Goldfarb, Gadolinium (III) spin labels for high-sensitivity distance measurements in transmembrane helices. Angew. Chem. 125, 12047 (2013)
N. Manukovsky, V. Frydman, D. Goldfarb, spin labels report the conformation and solvent accessibility of solution and vesicle bound Melittin. J. Phys. Chem. 119, 13732 (2015)
M. Yulikov, P. Lueders, M.F. Warsi, V. Chechik, G. Jeschke, Distance measurements in Au nanoparticles functionalized with nitroxide radicals and Gd3+-DTPA chelate complexes. Phys. Chem. Chem. Phys. 14, 10732 (2012)
A. Martorana, G. Bellapadrona, A. Feintuch, E. Di Gregorio, S. Aime, D. Goldfarb, Probing protein conformation in cells by EPR distance measurements using Gd3+ spin labeling. J. Am. Chem. Soc. 136, 13458 (2014)
M. Qi, A. Groß, G. Jeschke, A. Godt, M. Drescher, Gd (III)-PyMTA label is suitable for in-cell EPR. J. Am. Chem. Soc. 136, 15366 (2014)
F.C. Mascali, H. Ching, R.M. Rasia, S. Un, L.C. Tabares, Using genetically encodable self-assembling gdiii spin labels to make in-cell nanometric distance measurements. Angew. Chem. 128, 11207 (2016)
F.-X. Theillet, A. Binolfi, B. Bekei, A. Martorana, H.M. Rose, M. Stuiver, S. Verzini, D. Lorenz, M. van Rossum, D. Goldfarb, Structural disorder of monomeric α-synuclein persists in mammalian cells. Nature 530, 45 (2016)
Y. Yang, F. Yang, Y. Gong, J. Chen, D. Goldfarb, X. Su, A reactive, rigid GdIII Labeling tag for In-Cell EPR distance measurements in proteins. Angew. Chem. 129, 2960 (2017)
S. Razzaghi, M. Qi, A.I. Nalepa, A. Godt, G. Jeschke, A. Savitsky, M. Yulikov, RIDME spectroscopy with Gd (III) centers. J. Phys. Chem. Lett. 5, 3970 (2014)
A. Collauto, V. Frydman, M.D. Lee, E.H. Abdelkader, A. Feintuch, J.D. Swarbrick, B. Graham, G. Otting, D. Goldfarb, RIDME distance measurements using Gd (III) tags with a narrow central transition. Phys. Chem. Chem. Phys. 18, 19037 (2016)
D.T. Edwards, Z. Ma, T.J. Meade, D. Goldfarb, S. Han, M.S. Sherwin, Extending the distance range accessed with continuous wave EPR with Gd3+ spin probes at high magnetic fields. Phys. Chem. Chem. Phys. 15, 11313 (2013)
N. Manukovsky, A. Feintuch, I. Kuprov, D. Goldfarb, Time domain simulation of Gd3+– Gd3+ distance measurements by EPR. J. Chem. Phys. 147(4), 044201 (2017)
G. Jeschke, V. Chechik, P. Ionita, A. Godt, H. Zimmermann, J. Banham, C.R. Timmel, D. Hilger, H. Jung, DeerAnalysis2006—a comprehensive software package for analyzing pulsed ELDOR data. Appl. Magn. Reson. 30(3), 473–498 (2006)
T.H. Edwards, S. Stoll, Optimal Tikhonov regularization for DEER spectroscopy. J. Magn. Reson. 288, 58–68 (2018)
Y.W. Chiang, P.P. Borbat, J.H. Freed, The determination of pair distance distributions by pulsed ESR using Tikhonov regularization. J. Magn. Reson. 172(2), 279–295 (2005)
S.K. Misra, H.R. Salahi, Calculation of pulsed EPR DEER signal for two coupled Gd3+ ions by dipolar interaction using rotating frames. Physica B 637, 413903 (2022)
L. Fabregas Ibanez, G. Jeschke, S. Stoll, DeerLab: a comprehensive software package for analyzing dipolar electron paramagnetic resonance spectroscopy data. Magn. Reson. 1, 209–224 (2020)
G. Jeschke, DeerAnalysis2013.2 User Manual 03810: DeerAnalysis2013b-manual.pdf (http://www.epr.ethz.ch/software/index), p. 31.
Acknowledgements
SKM acknowledges helpful discussions with Professor Daniella Goldfarb, Weizmann Institute of Science, Israel and the hospitality offered during his 3-month sabbatical as a visiting professor at the Weizmann Institute in 2015, when this project was initially started. Some preliminary discussions on this subject with Profs. Shimon Vega and Akiva Feintuch are gratefully acknowledged. The original experimental Q- and W-band data analyzed in this paper were kindly provided to us by Professors Gunnar Jeschke and Daniella Goldfarb, respectively. We thank Professor Jeschke for some helpful discussion on how to analyze the data.
Funding
This work was partially supported by Natural Sciences and Engineering Research Council of Canada.
Author information
Authors and Affiliations
Contributions
SKM conceived the presented idea and developed a Fortran code to do the simulations. HRS improved the code and converted it into a Matlab code. The theory was developed further jointly by SKM and HRS. HRS performed the numerical simulations under the supervision of SKM. Both authors discussed the results and contributed to the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical Approval
Not applicable.
Consent for Publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix A.1
1.1 Flowchart for the Calculation of Four-Pulse DEER Signal

Appendix A.2
2.1 Expressions Referred to in the Flowchart A.1
The evolution of the density matrix under the application of a pulse is described by
where \({H}_{p};\, \mathrm{with/} p = \mathrm{obs},\mathrm{ pump},\) denote the Hamiltonians for the observer and pump pulses, respectively [27], given as
In Eqs. (A.2a) and (A.2b) \({\omega }_{1p};\mathrm{with} p=\mathrm{obs},\mathrm{ pump}\) are the amplitudes of the observer and pump pulses, respectively, in frequency units \((={\upgamma }_{e}{B}_{1p});{B}_{1p}=\) amplitudes of the pulses in Gauss (Table 1) and \({\upgamma }_{e}\) is the gyromagnetic ratio of the electron \(;\upphi\) is the phase of the pulse and\({S}_{k\pm }\); \(k=\mathrm{1,2}\) are the raising/lowering operators of the \(k\) th electronic spin of the coupled \(G{d}^{3+}\) ions system in the \(64 \times 64\) direct product Hilbert space. The indices in (A.2a) and (A.2b) indicate that the pulse Hamiltonians are adjusted to the specific transitions, \(-3/2 \leftrightarrow -1/2\) and\(-1/2 \leftrightarrow 1/2\), which define the observer and pump pulses, respectively.
The density matrix for the system of coupled \(G{d}^{3+}\) ions evolves during free evolution, i.e., in the absence of any pulse, governed by the LVN (Liouville von Neumann) equation, as follows:
where \(\rho \left({t}_{0}\right)\) is the density matrix just after the application of a pulse at time \({t}_{0}\) and \(\rho \left({t}_{0}+t\right)\) is the density matrix after free evolution over the time interval \(t\), subsequent to the application of a pulse.
The complex time-domain DEER signal for this case is expressed as
for the orientation, (\(\uptheta ,\upphi\)), of the static magnetic field with respect to the dipolar axis of the two \(G{d}^{3+}\) ions, whose dipole moments are oriented with respect to the respective molecular frames, denoted by five independent Euler angles \(\{{\upbeta }_{1},{\upgamma }_{1},{\mathrm{\alpha }}_{2},{\upbeta }_{2},{\upgamma }_{2}\}\), choosing the arbitrary angle \({\mathrm{\alpha }}_{1}=0,\) as shown in Fig. 2, exhibiting the configuration of the two coupled \(G{d}^{3+}\) ions [27]. In Eq. (A.4), \({\rho }_{f}\left(t\right)\) is the final density matrix \(.\)
Appendix B
3.1 Estimation of the Probability Distribution, \({\varvec{P}}\left({\varvec{r}}\right),\) from the Experimental Signal Using DeerAnalysis, Modified for Double Rotating-Frames (DRF)-Calculated Kernel Signals
-
1.
Determine the range of the distance, \(r\), between the two \(G{d}^{3+}\) ions, over which the probability distribution is non-zero, as calculated by the application of DeerAnalysis software.
-
2.
Simulate the four-pulse DEER signals using the DRF technique for a good number of evenly distributed points, \({r}_{n}\), over this range.
-
3.
Use the Spline function in Matlab to derive 512 time-domain signals by the cubic-spline technique, each calculated as a function of time in 512 even steps over the range of time from 0 to \({{\varvec{\uptau}}}_{2}\) (listed inTable 1) from the m DRF-calculated signals in the previous step.
-
4.
Normalize the time-domain signals, as simulated by the DRF technique, for the various \({r}_{n}\) values, by dividing each by its maximum value.
-
5.
Stack these signals in a 512 \(\times\) 512 matrix, \(K(t,r)\), where \(t,r\) denote the rows and columns, respectively.
-
6.
In the DeerAnalysis Matlab file, “make_bas_Tikh.m”, make the following replacements: (i) \(rmin\) and rmax by the minimum and maximum of range of distance \(r\), respectively, determined in step 1; (ii) \(dt\) by \({\uptau }_{2}/512\); (iii) the already present analytically calculated kernel signals,\(K(t,r)\),by the DRF-calculated kernel signals, \(K(t,r)\), as described in step 5
-
7.
7. In the Matlab file “get_Tikhonov_new.m” in DeerAnalysis folder, replace the conditional statement “if length(tdip) > 2048”, which allows one to use already calculated and stored analytically calculated kernel signals,\(K(t,r)\), by the statement “if length(tdip) > \({n}_{RF}\), where the integer \({n}_{RF}\) < 512”, i.e., \({n}_{RF}=200\) in the present case, for one to use the DRF-calculated kernel signals of size (512, 512) as in step 5.
-
8.
Delete “pake_base_tikh_512.mat” file from the DeerAnalysis folder, so that the already stored analytically calculated kernel signals, \(K(t,r)\), are removed, allowing the use of the DRF-calculated kernel signals, \(K(t,r)\). Then open the “DeerAnalysis.m” file, run it, and wait until the DeerAnalysis workspace loads up (about 5 min).
-
9.
When the workspace opens, click on the button “load”, then from the “Data sets” box load the experimental data file (with the ending.DTA).
-
10.
In the “Distance Analysis” box, select “Tikhonov” and “L curve” to obtain plots of L-shaped curve and the probability distribution as a function of distance.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Misra, S.K., Salahi, H.R. Estimation of Distance Distributions Between Gd3+ Ion Pairs with a Significant Zero-Field Splitting from Pulsed EPR DEER Data. Appl Magn Reson 54, 383–400 (2023). https://doi.org/10.1007/s00723-022-01521-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00723-022-01521-8