Abstract
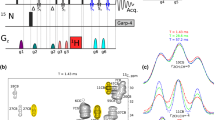
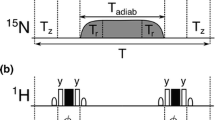
The transverse relaxation rates R 2 = 1/T 2 of protons can be determined by spin-echo sequences with multiple refocusing pulses using moderate radio-frequency field strengths and properly chosen inter-pulse delays so as to suppress echo modulations due to homonuclear scalar couplings. Combination with 2D heteronuclear correlation spectroscopy (HSQC) allows one to measure R 2 of arbitrary protons attached to nitrogen-15 or carbon-13 nuclei. Decays of six amide protons in the protein Ubiquitin that is nitrogen-15 enriched (but not deuterated) were measured at different temperatures.



Similar content being viewed by others
References
H.Y. Carr, E.M. Purcell, Phys. Rev. 94(3), 630–638 (1954)
S. Meiboom, D. Gill, Rev. Sci. Instrum. 29(8), 688–691 (1958)
J. Dittmer, G. Bodenhausen, Chem. Phys. Chem. 7(4), 831–836 (2006)
J. Dittmer, G. Bodenhausen, Chem. Phys. Chem. 5(11), 1750–1754 (2004)
G. Bodenhausen, R. Freeman, G.A. Morris, J. Magn. Reson. 23(1), 171–175 (1976)
G.A. Morris, R. Freeman, J. Magn. Reson. 29(3), 433–462 (1978)
R. Freeman, Chem. Rev. (Washington, DC, US) 91(7), 1397–1412 (1991)
R. Freeman, Prog. Nucl. Magn. Reson. Spectrosc. 32, 59–106 (1998)
K. Gopalakrishnan, N. Aeby, G. Bodenhausen, Chem. Phys. Chem. 8(12), 1791–1802 (2007)
N. Aeby, G. Bodenhausen, Chem. Phys. Lett. 463(4–6), 418–421 (2008)
T.F. Segawa, N. Aeby, G. Bodenhausen, Phys. Chem. Chem. Phys. 12(33), 9772–9776 (2010)
B. Baishya, T.F. Segawa, G. Bodenhausen, J. Am. Chem. Soc. 131(48), 17538–17539 (2009)
G.A. Morris, R. Freeman, J. Am. Chem. Soc. 101(3), 760–762 (1979)
T.F. Segawa, B. Baishya, G. Bodenhausen, Chem. Phys. Chem 11(15), 3343–3354 (2010)
B. Baishya, T.F. Segawa, G. Bodenhausen, J. Magn. Reson. 211(2), 240–242 (2011)
C. Barrère, P. Thureau, A. Thévand, S. Viel, Chem. Commun. (Cambridge, UK) 47(32), 9209–9211 (2011)
K. Takegoshi, K. Ogura, K. Hikichi, J. Magn. Reson. 84(3), 611–615 (1989)
T. Schulte-Herbruggen, Z.L. Madi, O.W. Sorensen, R.R. Ernst, Mol. Phys. 72(4), 847–871 (1991)
Z.L. Madi, B. Brutscher, T. Schulte-Herbruggen, R. Bruschweiler, R.R. Ernst, Chem. Phys. Lett. 268(3–4), 300–305 (1997)
C. Wang, A.G. Palmer, Magn. Reson. Chem. 41(10), 866–876 (2003)
A. Mittermaier, L.E. Kay, Science 312(5771), 224–228 (2006)
F. Ferrage, D. Cowburn, R. Ghose, J. Am. Chem. Soc. 131(17), 6048–6049 (2009)
G. Lipari, A. Szabo, J. Am. Chem. Soc. 104(17), 4546–4559 (1982)
G. Lipari, A. Szabo, J. Am. Chem. Soc. 104(17), 4559–4570 (1982)
P. Pelupessy, F. Ferrage, G. Bodenhausen, J. Chem. Phys. 126(13), 134508 (2007)
J. Dittmer, G. Bodenhausen, J. Am. Chem. Soc. 126(5), 1314–1315 (2004)
V.Y. Orekhov, D.M. Korzhnev, L.E. Kay, J. Am. Chem. Soc. 126(6), 1886–1891 (2004)
M. Verde, S. Ulzega, F. Ferrage, G. Bodenhausen, J. Chem. Phys. 130(7), 074506 (2009)
S. Ulzega, M. Verde, F. Ferrage, G. Bodenhausen, J. Chem. Phys. 131(22), 224503 (2009)
S. Ulzega, N. Salvi, T.F. Segawa, F. Ferrage, G. Bodenhausen, Chem. Phys. Chem. 12(2), 333–341 (2011)
B. Boulat, G. Bodenhausen, J. Biomol. NMR 3(3), 335–348 (1993)
R. Ishima, P.T. Wingfield, S.J. Stahl, J.D. Kaufman, D.A. Torchia, J. Am. Chem. Soc. 120(40), 10534–10542 (1998)
T.S. Ulmer, I.D. Campbell, J. Boyd, J. Magn. Reson. 157(2), 181–189 (2002)
P. Lundstrom, M. Akke, J. Biomol. NMR 32(2), 163–173 (2005)
R. Ishima, D.A. Torchia, J. Biomol. NMR 25(3), 243–248 (2003)
D.M. Korzhnev, K. Kloiber, L.E. Kay, J. Am. Chem. Soc. 126(23), 7320–7329 (2004)
L. Braunschweiler, R.R. Ernst, J. Magn. Reson. 53(3), 521–528 (1983)
A. Bax, D.G. Davis, J. Magn. Reson. 63(1), 207–213 (1985)
C. Griesinger, G. Otting, K. Wuthrich, R.R. Ernst, J. Am. Chem. Soc. 110(23), 7870–7872 (1988)
G. Bodenhausen, D.J. Ruben, Chem. Phys. Lett. 69(1), 185–189 (1980)
A.G. Palmer, J. Cavanagh, P.E. Wright, M. Rance, J. Magn. Reson. 93(1), 151–170 (1991)
L.E. Kay, P. Keifer, T. Saarinen, J. Am. Chem. Soc. 114(26), 10663–10665 (1992)
J. Schleucher, M. Schwendinger, M. Sattler, P. Schmidt, O. Schedletzky, S.J. Glaser, O.W. Sorensen, C. Griesinger, J. Biomol. NMR 4(2), 301–306 (1994)
F. Delaglio, S. Grzesiek, G.W. Vuister, G. Zhu, J. Pfeifer, A. Bax, J. Biomol. NMR 6(3), 277–293 (1995)
C. P. Slichter, Principles of magnetic resonance, Third enlarged and updated edn. (Springer, Berlin, 1996)
P. Schanda, Prog. Nucl. Magn. Reson. Spectrosc. 55(3), 238–265 (2009)
D. Jeannerat, J. Magn. Reson. 186(1), 112–122 (2007)
P. Schanda, B. Brutscher, J. Am. Chem. Soc. 127(22), 8014–8015 (2005)
L. Frydman, T. Scherf, A. Lupulescu, Proc. Natl. Acad. Sci. USA 99(25), 15858–15862 (2002)
P. Pelupessy, J. Am. Chem. Soc. 125(40), 12345–12350 (2003)
H. Geen, R. Freeman, J. Magn. Reson. 93(1), 93–141 (1991)
Acknowledgments
We thank Martial Rey for technical support and are indebted to the Swiss National Science Foundation, the Commission for Technology and Innovation, the Ecole Polytechnique Fédérale de Lausanne and the CNRS for funding.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Segawa, T.F., Baishya, B. & Bodenhausen, G. Transverse Relaxation of Scalar Coupled Protons in Magnetic Resonance of Non-Deuterated Proteins. Appl Magn Reson 42, 353–361 (2012). https://doi.org/10.1007/s00723-011-0298-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00723-011-0298-1