Abstract
Potassium pyroantimonate precipitation, transmission electron microscopy, and X-ray microanalysis were used to investigate the subcellular localization of loosely bound calcium in Robinia pseudoacacia pulvinar motor cells during phytochrome-mediated nyctinastic closure. Calcium localization was carried out in pulvini collected in white light 2 h after the beginning of the photoperiod, immediately after a red light or a far-red light pulse applied 2 h after the beginning of the photoperiod and after 15 or 25 min of darkness respectively. Calcium antimonate precipitates were found in all the pulvinar tissues from the epidermis to the vascular bundle, independent of the light treatment. At subcellular level, precipitates were found mainly in the intercellular spaces, the inner surface of the plasma membrane, cytoplasm, colloidal vacuoles, and nuclei. Red light enhanced the nyctinastic closure of leaflets and caused an asymmetric distribution of cytosolic calcium precipitates between the extensor and flexor motor cells. Both the number and area of the cytosolic calcium precipitates drastically increased in the extensor cells compared to the flexor motor cells. Red light had a rapid and transient effect on the distribution of cytosolic calcium precipitates, which occurred during or at the end of the irradiation, before leaflet closure. By contrast, the distribution of cytosolic loosely bound calcium was similar between the extensor and flexor motor cells after irradiation with far-red light. Our results demonstrate that red light causes specific calcium mobilization in pulvinar motor cells and suggest the involvement of cytoplasmic Ca2+ as a second messenger for phytochrome during nyctinastic closure.






Similar content being viewed by others
References
Bizet F, Pereda-Loth V, Chauvet H, Gérard J, Eche B, Girousse C, Courtade M, Perbal G, Legué V (2018) Both gravistimulation onset and removal trigger an increase of cytoplasmatic free calcium in statocytes of roots grown in microgravity. Sci Rep 8:11442
Campbell NA, Thomson WW (1977) Effects of lanthanum and ethylenediaminetetraacetate on leaf movements of Mimosa. Plant Physiol 60:635–639
Charpentier M (2018) Calcium signals in the plant nucleus: origin and function. J Exp Bot 69(17):4165–4173
Costa A, Navazio L, Szabo I (2018) The contribution of organelles to plant intracellular calcium signalling. J Exp Bot 69(17):4175–4193
Edel KH, Marchadier E, Brownlee C, Kudla J, Hetherington AM (2017) The evolution of calcium-based signalling in plants. Curr Biol 27:R667–R679
Eprintsev AT, Fedorin DN, Igamberdiev AU (2013) Ca2+ is involved in phytochrome A-dependent regulation of the succinate dehydrogenase gene sdh1-2 in Arabidopsis. J Plant Physiol 170:1349–1352
Eprintsev AT, Fedorin DN, Sazonova V, Igamberdiev AU (2016) Light inhibition of fumarase in Arabidopsis leaves is phytochrome A-dependent and mediated by calcium. Plant Physiol Biochem 102:161–166
Falhof J, Pederson BP, Fuglsang AT, Palmgren M (2016) Plasma membrane H+-ATPase regulation in the center of plant physiology. Mol Plant 9:323–337
Fleurat-Lessard P, Frange N, Maeshima M, Ratajczak R, Bonnemain J-L, Martinoia E (1997) Increased expression of vacuolar aquaporin and H+-ATPase related to motor cell function in Mimosa pudica L. Plant Physiol 114:827–834
Fleurat-Lessard P, Bére E, Lallemand M, Dédaldéchamp F, Roblin G (2016) Co-occurrence of tannin and tannin-less vacuoles in sensitive plants. Protoplasma 253(3):821–834
Galon Y, Finkler A, Fromm H (2010) Calcium-regulated transcription in plants. Mol Plant 3(4):653–669
Gómez LA, Simón E (1995) Circadian rhythm of Robinia pseudoacacia leaflet movements: role of calcium and phytochromes. Photochem Photobiol 61:210–215
Hayat M (1993) Stains and cytochemical methods. Plenum Press, New York
Hayatsu M, Ono M, Hamamoto C, Suzuki S (2012) Cytochemical and electron probe X-ray microanalysis studies on the distribution change of intracellular calcium in columella cells of soybean roots under stimulated microgravity. J Electron Microsc 61(1):57–69
Hollins DL, Jaffe MJ (1997) On the role of tannin vacuoles in several nastic responses. Protoplasma 199:215–222
Hopwood D, Slidders W, Yeaman GR (1989) Tissue fixation with phenol-formaldehyde for routine histopathology. Histochem J 21(4):228–234
Hubbard KE, Siegelm RS, Valerio G, Brandt B, Schroeder JI (2012) Abscisic acid and CO2 signaling via calcium sensitivity priming in guard cells, new CDPK mutant phenotypes and a method for improved resolution of stomatal stimulus-response analysis. Ann Bot 109:5–17
Hughes J (2013) Phytochrome cytoplasmic signalling. Ann Rev Plant Biol 64:377–402
Jauneau A, Quentin M, Driouich A (1997) Micro-heterogeneity of pectins and calcium distribution in the epidermal and cortical parenchyma cell walls of flax hypocotyl. Protoplasma 198:9–19
Kashiwa HK, Tiersch NJ (1984) Evaluation of potassium pyroantimonate/sucrose /glutaraldehyde concentration and incubation time as essential variables for localizing calcium bound to organic compounds in epiphyseal chondrocytes. J Histochem Cytochem 32(10):1055–1065
Kim HY, Coté GG, Crain RC (1993) Potassium channels in Samanea saman protoplast controlled by phytochrome and the biological clock. Science 260:960–962
Kinoshita T, Nishimura M, Shimazaki K (1995) Cytosolic concentration of Ca2+ regulates the plasma membrane H+-ATPase in guard cells of fava bean. Plant Cell 7(8):1333–1342
Klein RL, Yen S-S, Thureson-Klein A (1972) Critique on the K-pyroantimonate method for semiquantitative estimation of cations in conjunction with electron microscopy. J Histochem Cytochem 20(1):65–78
Klymchuck DO, Brown CS, Chapman DK, Vorobyova TV, Martyn GM (2001) Cytochemical localization of calcium in soybean root cap cells in microgravity. Adv Space Res 27(5):967–972
Kreslavski VD, Carpentier R, Klimov VV, Allakhverdiev SI (2009) Transduction mechanism of photoreceptor signals in plant cells. J Photochem Photobiol 10:63–80
Labuz J, Samardakiewicz S, Hermanowicz P, Wyroba E, Pilarska M, Gabrys H (2016) Blue light-dependent changes in loosely bound calcium in Arabidopsis mesophyll cells: an X-ray microanalysis study. J Exp Bot 67(13):3953–3964
Lee Y, Satter RL (1989) Effects of white, blue, red light and darkness on pH of the apoplast in the Samanea pulvinus. Planta 178:31–40
Lherminier J, Benhamou J, Larrue ML, Milat E, Boudon-Padieu E, Nicole M, Blein JP (2003) Cytochemical characterization of elicitin induced protection in tobacco plants infected by Phytophthora parasitica or phytoplasm. Phytopathology 93:1308–1319
Liberman M, Mutaftschiev S, Jauneau A, Vian B, Catesson AM, Goldberg R (1999) Mung bean hypocotyl homogalacturonan: localization, organization and origin. Ann Bot 84(2):225–233
Luo YL, Luo YJ, Li QJ (2011) Subcellular localization of calcium during Alpinia mutica Roxb. (Zingiberaceae) style movement. Protoplasma 248:251–256
McEvoy RC, Koukkari WL (1972) Effects of ethylenediaminetetraacetic acid, auxin and gibberellic acid on phytochrome controlled nyctinasty in Albizzia julibrissin. Physiol Plant 26:143–147
Mentré P, Escaig F (1988) Localization of cations by pyroantimonate. I. Influence of fixation on distribution of calcium and sodium. An approach by analytical ion microscopy. J Histochem Cytochem 36(1):49–54
Mentré P, Halpern S (1988) Localization of cations by pyroantimonate. II Electron probe microanalysis of calcium and sodium in skeletal muscle of mouse. J Histochem Cytochem 36(1):55–64
Moran N (1990) The role of ion channels in osmotic volume changes in Samanea motor cells by patch-clamp methods. In: Satter RL, Gorton HL, Vogelmann TC (eds) The pulvinus: motor organ for leaf movement. Am Soc Plant Physiol, Rockville, pp 142–159
Moran N (2007) Osmoregulation of leaf motor cells. FEBS Lett 581:2337–2347
Moran N (2015) Rhythmic leaf movements: physiological and molecular aspects. In: Mancuso S, Shabala S (eds) Rhythms in plants. Springer, Switzerland, pp 57–95
Morse MJ, Satter RL (1979) Relationships between motor cell ultrastructure and leaf movements in Samanea saman. Plant Physiol 46:338–346
Moysset L, Simón E (1989) Role of calcium in phytochrome-controlled nyctinastic movements of Albizzia lophantha leaflets. Plant Physiol 90:1108–1114
Moysset L, Simón E (1990) Effect of EGTA on nyctinastic closure mediated by phytochrome in Albizzia lophantha. Plant Cell Physiol 31(2):187–193
Moysset L, Simón E (1991) Secondary pulvinus of Robinia pseudoacacia (Leguminosae) structural and ultrastructural features. Am J Bot 78(11):1467–1148
Moysset L, Solé-Sugrañes L, Simón E (1991) Changes in morphometry and elemental composition of Robinia pseudoacacia pulvinar motor cells during leaflet movements. J Exp Bot 42(243):1315–1323
Moysset L, Gómez LA, Simón E (1994) Effects of lanthanum on rhythmic and nyctinastic leaflet movements in Albizzia lophantha. J Exp Bot 45(270):85–93
Moysset L, Fernandez E, Cortadellas N, Simón E (2001) Intracellular localization of phytochrome in Robinia pseudoacacia pulvini. Planta 213:565–574
Plavcová L, Hacke UG (2011) Heterogeneous distribution of pectin epitopes and calcium in different pit types of four angiosperm species. New Phytol 192(4):885–897
Racusen R, Satter RL (1975) Rhythmic and phytochrome-regulated changes in transmembrane potential in Samanea pulvini. Nature 255:408–410
Raeini-Sarjaz M (2011) Circadian rhythm leaf movement of Phaseolus vulgaris and the role of calcium ions. Plant Signal Behav 6(6):962–967
Reynolds ES (1963) The use of lead citrate at high pH as an electron opaque stain in electron microscopy. J Cell Biol 17:208–212
Roblin G, Fleurat-Lessard P (1984) A possible mode of calcium involvement in dark- and light-induced movements in Cassia fasciculata Michx. Plant Cell Physiol 25:1495–1499
Roblin G, Fleurat-Lessard P, Bonmort J (1989) Effect of compounds affecting calcium channels on phytochrome- and blue pigment-mediated pulvinar movements of Cassia fasciculata. Plant Physiol 90:697–701
Roblin G, Fleurat-Lessard P, Everat-Bourbouloux A, Bonmort J (1990) Phytochrome- and blue pigment-mediated leaf movements: experimental approach in the signal transduction. Photochem Photobiol 52(1):197–202
Sakai-Wada A, Yagi S (1993) Ultrastructural studies on the Ca2+ localization in the dividing cells of the maize root tip. Cell Struct Funct 18:389–397
Satter RL, Galston AW (1981) Mechanisms of control of leaf movement. Annu Rev Plant Physiol 32:107–112
Satter RL, Marinoff P, Galston AW (1970) Phytochrome controlled nyctinastic in Albizzia julibrissin. II. Potassium flux as a basis for leaflet movement. Amer J Bot 57(8):916–926
Satter RL, Garber RC, Khairallah L, Cheng Y-S (1982) Elemental analysis of freeze-dried thin sections of Samanea motor organs: barriers to ion diffusion through the apoplast. J Cell Biol 95:893–902
Shacklock PS, Read ND, Trewavas AJ (1992) Cytosolic free calcium mediates red light-induced photomorphogenesis. Nature 358:753–755
Slocum RD, Roux SJ (1982) An improved method for the subcellular localization of calcium using a modification of the antimonate precipitation technique. J Histochem Cytochem 30(7):617–629
Spurr AR (1969) A low viscosity epoxy resin embedding medium for electron microscopy. J Ultrastruct Res 26:31–43
Suh SJ, Moran N, Lee Y (2000) Blue light activates potassium-efflux channels in flexor cells from Samanea saman motor organs via two mechanisms. Plant Physiol 123:833–843
Tonetto de Freitas S, Padda S, Wu Q, Park S, Mitcham E (2011) Dynamic alterations in cellular and molecular components during blossom-end root development in tomatoes expressing sCAX1, a constitutively active Ca2+/H+ antiporter from Arabidopsis. Plant Physiol 156(2):844–855
Toriyama H, Jaffe MJ (1972) Migration of calcium and its role in the regulation of seismonasty in the motor cell of Mimosa pudica L. Plant Physiol 49:72–81
Tretyn A, Kendrick E, Kopcewicz J (1992) Cytochemical studies on phytochrome mediated changes of Ca2+ localization in etiolated oat coleoptile cells. J Exp Bot 43(249):439–448
Wick SM, Hepler PK (1982) Selective localization of intracellular Ca2+ with potassium pyroantimonate. J Histochem Cytochem 30:1190–1204
Yao H, Xu Q, Yuan M (2008) Actin dynamics mediates the changes of calcium level during the pulvinus movement of Mimosa pudica. Plant Signal Behav 3(11):954–960
Zhang S, Chen S, Chen F, Jiang B (2011) The role of ionic calcium in the gravitropic response of a creeping chrysanthemum cultivar. Russian J Plant Physiol 58(4):696–702
Acknowledgements
We would like to thank the Experimental Fields Service and the Transmission Electron Microscopy Unit of the Scientific and Technological Centers (CCITUB) at the University of Barcelona for their valuable technical assistance. Thanks to P. Rovira and M. Pinto for their assistance in the preparation of the figures and Michael Maudsley (Language Services, University of Barcelona) for correcting the English text.
Funding
This work was supported by a research project (CGL2011-26310) from the Ministry of Science and Innovation, Spain.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Klaus Harter
Electronic supplementary material
Supplementary Figure 1
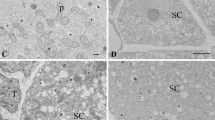
Comparison of fixative procedures in extensor cortical motor cells of R. pseudoacacia secondary pulvini fixed in: (a) 2.5% GA, 2% PFA and 2% OsO4; (b) 2.5% GA, 2% PFA, 1% PH and 2% OsO4; (c) 2.5% GA and 2% PFA; and (d) 2.5% GA, 2% PFA and 1% PH. In (a - d) note the absence of calcium antimonate precipitates. In (a) and (b) the integrity of the plasma membrane, tonoplast, chloroplast envelope and thylakoid membranes is preserved, but in (c) poor preservation of the chloroplast membranes is evident. In (d) increased electron density of cell wall and chloroplasts and preservation of the tissue were more satisfactory than in c. Legends as in Fig. 3, 4 and 5. Thin arrow and arrowhead indicate plasma membrane and tonoplast respectively. Scale bar: (a – d) 0.5 μm (JPG 1858 kb)
Rights and permissions
About this article
Cite this article
Moysset, L., Llambrich, E. & Simón, E. Calcium changes in Robinia pseudoacacia pulvinar motor cells during nyctinastic closure mediated by phytochromes. Protoplasma 256, 615–629 (2019). https://doi.org/10.1007/s00709-018-1323-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-018-1323-0