Abstract
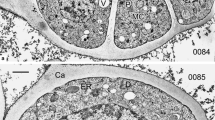
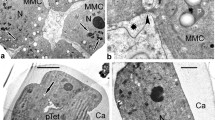
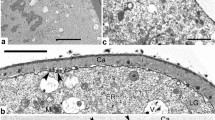
By a detailed ontogenetic study of Ambrosia trifida pollen, tracing each stage of development with TEM, we aim to understand the establishment of the pollen wall and to unravel the mechanisms underlying sporoderm development. The main steps of exine ontogeny in Ambrosia trifida, observed in the microspore periplasmic space, are as follows: spherical units, gradually transforming into columns, then to rod-like units; the appearance of the initial reticulate tectum; growth of columellae under the tectum and initial sporopollenin accumulation on them; the appearance of the endexine lamellae, first in fragments, then in long laminae; the cessation of the glycocalyx growth and its detachment from the plasma membrane, resulting in the appearance of gaps; massive accumulation of sporopollenin on the tectum, columellae, and endexine, and the appearance of the foot layer at the young post-tetrad stage, accompanied by establishment of caveae in sites of the former gaps; and final massive sporopollenin accumulation. This sequence of developmental events in all probability corresponds to the sequence of self-assembling micellar mesophases. This gives (together with earlier findings and experimental modeling of exine) strong evidence that the genome and self-assembly share control of exine formation. In this sense, self-assembly itself can be seen as an inherent mechanism of evolution.












Similar content being viewed by others
References
Atkin S, Barrier S, Cui Z, Fletcher PDI, Mackenzie G, Panel V, Sol V, Zhang X (2011) UV and visible light screening by individual sporopollenin exines derived from Lycopodium clavatum (club moss) and Ambrosia trifida (giant ragweed). J Photochem Photobiol B Biol 102:209–217. https://doi.org/10.1016/j.jphotobiol.2010.12.005
Barnes SH, Blackmore S (1986) Some functional features in pollen development. In: Blackmore S, Ferguson IK (eds) Pollen and spores: form and function. Academic, London, pp 71–80
Barnes SH, Blackmore S (1992) Ultrastructural organization of two tapetal types in angiosperms. Arch Histol Cytol 55(suppl):217–224
Blackmore S (1990) Sporoderm homologies and morphogenesis in land plants, with a discussion on Echinops sphaerocephala (Compositae). Plant Syst Evol 5:1–12
Blackmore S, Barnes SH (1985) Cosmos pollen ontogeny: a scanning electron microscope study. Protoplasma 126:91–99
Blackmore S, Barnes SH (1987) Pollen wall morphogenesis in Tragopogon porrifolius L. (Compositae: Lactuceae) and its taxonomic significance. Rev Palaeobot Palynol 52:233–246. https://doi.org/10.1016/0034-6667(87)90056-X
Blackmore S, Barnes SH (1988) Pollen ontogeny in Catananche caerulea L. (Compositae: Lactuceae). I. Premeiotic phase to establishment of tetrads. Ann Bot (Oxford) 62:605–614
Blackmore S, Claugher D (1987) Observations on the substructural organization of the exine in Fagus sylvatica L. (Fagaceae) and Scorzonera hispanica L. (Compositae: Lactuceae). Rev Palaeobot Palynol 5:175–184
Blackmore S, van Helvoort HAM, Punt W (1984) On the terminology, origins and fuctions of caveate pollen in compositae. Rev Palaeobot Palynol 43:293–301
Blackmore S, Wortley AH, Skvarla JJ, Rowley JR (2007) Pollen wall development in flowering plants. New Phytol 174:483–498
Blackmore S, Wortley AH, Skvarla JJ, Robinson H (2009) Evolution of pollen in the compositae. In: Funk VA, Susanna A, Stuessy T, Bayer R (eds) Systematics, evolution and biogeography of the Compositae. IAPT, Vienna, pp 101–130
Blackmore S, Wortley AH, Skvarla JJ, Gabarayeva NI (2010) Developmental origins of structural diversity in pollen walls of Compositae. Plant Syst Evol 284:17–32. https://doi.org/10.1007/s00606-009-0232-2
Dickinson HG, Potter U (1976) The development of patterning in the alveolar sexine of Cosmos bipinnatus. New Phytol 76:543–550
Dobritsa AA, Coerper D (2012) The novel plant protein INAPERTURATE POLLEN1 marks distinct cellular domains and controls formation of apertures in the Arabidopsis pollen exine. Plant Cell 24:4452–4464. https://doi.org/10.1105/tpc.112.101220
Dobritsa AA, Kirkpatrick AB, Reeder SH, Li P, Owen HA (2018) Pollen aperture factor INP1 acts late in aperture formation by excluding specific membrane domains from exine deposition. Plant Physiol 176:326–339. https://doi.org/10.1104/pp.17.007201
El-Ghazaly G (1982) Ontogeny of pollen wall of Leontodon autumnalis (Hypochoeridinae, Compositae). Grana 21:103–113
El-Ghazaly G, Nilsson S (1991) Development of tapetum and orbicules of Catharanthus roseus (Apocynaceae). In: Blackmore S, Barnes SH (eds) Pollen and spores. Syst ass special vol 41. Clarendon, Oxford, pp 317–329
Fridrichsberg DA (1995) Colloidal chemistry. Chemistry, St. Petersburg
Gabarayeva NI (1993) Hypothetical ways of exine pattern determination. Grana 33(suppl 2):54–59. https://doi.org/10.1080/00173139309428980
Gabarayeva NI (1997) Ultrastructural foundations of sporoderm development in Magnoliidae. Diss. Dsc., Komarov Bot. Inst. (In Russian)
Gabarayeva NI (2000) Principles and recurrent themes in sporoderm development. In: Harley MM, Morton CM, Blackmore S (eds) Pollen and spores: morphology and biology. Royal Botanic Gardens, Kew, pp 1–17
Gabarayeva NI, Grigorjeva VV (2003) Comparative study of the pollen wall evelopment in Illicium floridanum (Illiciaceae) and Schisandra chinensis (Schisandraceae). Taiwania 48:147–167
Gabarayeva NI, Grigorjeva VV (2010) Sporoderm ontogeny in Chamaedorea microspadix (Arecaceae). Self-assembly as the underlying cause of development. Grana 49:91–114. https://doi.org/10.1080/00173131003650920
Gabarayeva NI, Grigorjeva VV (2011) Sporoderm development in Swida alba (Cornaceae), interpreted as a self-assembling colloidal system. Grana 50:81–101. https://doi.org/10.1080/00173134.2011.580448
Gabarayeva N, Grigorjeva V (2012) Sporoderm development and substructure in Magnolia sieboldii and other Magnoliaceae: an interpretation. Grana 51:119–147. https://doi.org/10.1080/00173134.2012.688863
Gabarayeva NI, Grigorjeva VV (2013) Experimental modelling of exine-like structures. Grana 52:241–257. https://doi.org/10.1080/00173134.2013.818165
Gabarayeva NI, Grigorjeva VV (2014) Sporoderm and tapetum development in Eupomatia laurina (Eupomatiaceae). An interpretation. Protoplasma 251:1321–1345. https://doi.org/10.1007/s00709-014-0631-2
Gabarayeva NI, Grigorjeva VV (2016) Simulation of exine patterns by self-assembly. Plant Syst Evol 302:1135–1156. https://doi.org/10.1007/s00606-016-1322-6
Gabarayeva NI, Grigorjeva VV (2017) Self-assembly as the underlying mechanism for exine development in Larix decidua D. C. Planta 246:471–493. https://doi.org/10.1007/s00425-017-2702-z
Gabarayeva NI, Hemsley AR (2006) Merging concepts: the role of self-assembly in the development of pollen wall structure. Rev Palaeobot Palynol 138:121–139. https://doi.org/10.1016/j.revpalbo.2005.12.001
Gabarayeva NI, Grigorjeva VV, Rowley JR, Hemsley AR (2009a) Sporoderm development in Trevesia burckii (Araliaceae). I. Tetrad period: further evidence for the participation of self-assembly processes. Rev Palaeobot Palynol 156:211–232. https://doi.org/10.1016/j.revpalbo.2008.12.001
Gabarayeva NI, Grigorjeva VV, Rowley JR, Hemsley AR (2009b) Sporoderm development in Trevesia burckii (Araliaceae). II. Post-tetrad period: further evidence for the participation of self-assembly processes. Rev Palaeobot Palynol 156:233–247. https://doi.org/10.1016/j.revpalbo.2009.01.004
Gabarayeva NI, Grigorjeva VV, Rowley JR (2010a) A new look at sporoderm ontogeny in Persea americana. Micelles and the hidden side of development. Ann Bot (Oxford) 105:939–955. https://doi.org/10.1093/aob/mcq075
Gabarayeva NI, Grigorjeva VV, Rowley JR (2010b) Sporoderm development in Acer tataricum (Aceraceae). An interpretation. Protoplasma 247:65–81. https://doi.org/10.1007/s00709-010-0141-9
Gabarayeva N, Grigorjeva V, Polevova S (2011a) Exine and tapetum development in Symphytum officinale (Boraginaceae). Exine substructure and its interpretation. Plant Syst Evol 296:101–120. https://doi.org/10.1007/s00606-011-0479-2
Gabarayeva NI, Grigorjeva VV, Marquez G (2011b) Ultrastructure and development during meiosis and the tetrad period of sporogenesis in the leptosporangiate fern Alsophila setosa (Cyatheaceae) compared with corresponding stages in Psilotum nudum (Psilotaceae). Grana 50:235–262. https://doi.org/10.1080/00173134.2011.631759
Gabarayeva N, Grigorjeva V, Kosenko Y (2013a) I Primexine development in Passiflora racemosa Brot. Overlooked aspects of development. Plant Syst Evol 299:1013–1035. https://doi.org/10.1007/s00606-013-0757-2
Gabarayeva N, Grigorjeva V, Kosenko Y (2013b) II. Exine development in Passiflora racemosa Brot.: post-tetrad period. Overlooked aspects of development. Plant Syst Evol 299:1037–1055. https://doi.org/10.1007/s00606-013-0756-3
Gabarayeva N, Grigorjeva V, Polevova S (2014) Sporoderm and tapetum ontogeny in Juniperus communis (Cupressaceae). Connective structures between tapetum and microspores. Rev Palaeobot Palynol 206:23–44
Gabarayeva NI, Grigorjeva VV, Blackmore S (2016) Pollen wall substructure and development in Tanacetum vulgare (Compositae: Anthemideae): revisiting hypotheses on pattern formation in complex cell walls. Int J Plant Sci 177:347–370. https://doi.org/10.1086/684946
Gabarayeva N, Grigorjeva V, Polevova S, Hemsley AR (2017) Pollen wall and tapetum development in Plantago major L. (Plantaginaceae): assisting self-assembly. Grana 56:81–111. https://doi.org/10.1080/00173134.2016.1159729
Gabarayeva NI, Polevova SV, Grigorjeva VV, Blackmore S (2018) Assembling the thickest plant cell wall: exine development in Echinops (Asteraceae, Cynareae). Planta 248(2):323–346
Griffiths PC, Hemsley AR (2001) Rasberries and muffins—mimicking biological pattern formation. Colloids Surf B: Biointerfaces 25:163–170. https://doi.org/10.1016/S0927-7765(01)00316-2
Grigorjeva V, Gabarayeva N (2015) The development of sporoderm, tapetum and Ubisch bodies in Dianthus deltoides (Caryophyllaceae): self-assembly in action. Rev Palaeobot Palynol 219:1–27. https://doi.org/10.1016/j.revpalbo.2015.03.005
Grigorjeva VV, Gabarayeva N (2018) Pollen wall ontogeny in Polemonium caeruleum (Polemoniaceae) and suggested underlying mechanisms of development. Protoplasma 255:109–128. https://doi.org/10.1007/s00709-017-1121-0
Hemsley AR (1998) Non-linear variation in simulated complex pattern development. J Theor Biol 192:73–79
Hemsley AR, Gabarayeva NI (2007) Exine development: the importance of looking through colloidal chemistry “window”. Plant Syst Evol 263:25–49. https://doi.org/10.1007/s00606-006-0465-2
Hemsley AR, Griffiths PC (2000) Architecture in the microcosm: biocolloids, self-assembly and pattern formation. Philos Trans R Soc Lond A 358:547–564
Hemsley AR, Jenkins PD, Collinson ME, Vincent B (1996) Experimental modelling of exine self-assembly. Bot J Linn Soc 121:177–187. https://doi.org/10.1111/j.1095-8339.1996.tb00752.x
Hemsley AR, Vincent B, Collinson ME, Griffiths PC (1998) Simulated self-assembly of spore exines. Ann Bot (Oxford) 82:105–109
Heslop-Harrison J (1972) Pattern in plant cell wall morphogenesis in miniature. Proc R Inst G B 45:335–351
Hesse M, Hess MW (1993) Recent trends in tapetum research. A cytological and methodological review. Plant Syst Evol 7:127–145
Horner HT, Pearson CB (1978) Pollen wall and aperture development in Helianthus annuus (Compositae: Heliantheae). Am J Bot 65:293–309
Ingber DE (2003a) Tensegrity I. Cell structure and hierarchical systems biology. J Cell Sci 116:1157–1173. https://doi.org/10.1242/jcs.00359
Ingber DE (2003b) Tensegrity II. How structural networks influence cellular information processing networks. J Cell Sci 116:1397–1408. https://doi.org/10.1242/jcs.00360
Kamelina OP (1988) Types of the tapetum in angiosperms. Ann Sci Univ Reims ARERS 23:71–73
Larson DA (1966) On the significance on the detailed structure of Passiflora caerulea exines. Bot Gaz 127:40–48
Lersten NR, Curtis JD (1990) Invasive tapetum and tricelled pollen in (Asteraceae, tribe Heliantheae). Plant Syst Evol 169:237–243
Makra L, Juhász M, Béczi R, Borsos E (2005) The history and impacts of airborne Ambrosia (Asteraceae) pollen in Hungary. Grana 44:57–64. https://doi.org/10.1080/00173130510010558
Martin MD, Chamecki M, Brush GS, Meneveau C, Parlange MB (2009) Pollen clumping and wind pollination in an invasive angiosperm. Am J Bot 96:1703–1711. https://doi.org/10.3732/ajb.0800407
McLauchlan KK, Barnes CS, Craine JS (2011) Interannual variability of pollen productivity and transport in mid-North America from 1997 to 2009. Aerobiologia 27:181–189. https://doi.org/10.1007/s10453-010-9186-7
Pacini E, Franchi GG (1991) Diversification and evolution of the tapetum. In: Blackmore S, Barnes SH (eds) Pollen and spores: patterns of diversification. Clarendon, Oxford, pp 301–316
Pacini E, Juniper BE (1983) The ultrastructure of the formation and development of the amoeboid tapetum in Arum italicum Miller. Protoplasma 117:116–129. https://doi.org/10.1007/BF01288350
Pacini E, Keijer CJ (1989) Ontogeny of intruding non-periplasmodial tapetum in the wild chicory, Cichorium intybus (Compositae). Plant Syst Evol 67:149–164
Pacini E, Franchi GG, Hesse M (1985) The tapetum: its form, function and possible phylogeny in Embryophyta. Plant Syst Evol 149:155–185
Pettitt JM (1979) Ultrastructure and cytochemistry of spore wall morphogenesis. In: Dyer AF (ed) The experimental biology of ferns. Academic, London, pp 211–252
Punt W, Hoen PP (2009) The Northwest European Pollen Flora, 70 Asteraceae—Asteroideae. Rev Palaeobot Palynol 157:22–183
Reinitzer F (1888) Zur Kenntnis des Cholesterins. Monatshefte 9:421–441
Rowley JR (1973) Formation of pollen exine bacules and microchannels on a glycocalyx. Grana 13:129–138
Rowley JR (1990) The fundamental structure of the pollen exine. Plant Syst Evol 5:13–29
Rowley JR, Dahl AO (1977) Pollen development in Artemisia vulgaris with special reference to glycocalyx material. Pollen Spores 19:169–284
Rowley JR, Walles B (1985) Cell differentiation in microsporangia of Pinus sylvestris. III. Late pachytene. Nord J Bot 5:255–271. https://doi.org/10.1111/j.1756-1051.1985.tb01655.x
Rowley JR, Dahl AO, Rowley JS (1981) Substructure in exines of Artemisia vulgaris (Asteraceae). Rev Palaeobot Palynol 35:1–38
Rowley JR, Gabarayeva NI, Walles B (1992) Cyclic invasion of tapetal cells into loculi during microspore development in Nymphaea colorata (Nymphaceaе). Am J Bot 79:801–808
Sampathkumar A, Yan A, Krupinksi P, Meyerowitz EM (2014) Physical forces regulate plant development and morphogenesis. Curr Biol 24:475–483. https://doi.org/10.1016/j.cub.2014.03.014
Sheldon JM, Dickinson HG (1983) Determination of patterning in the pollen wall of Lilium henryi. J Cell Sci 63:191–208
Sitte P (1981) Role of lipid self-assembly in subcellular morphogenesis. In: Kiermayer O (ed) Cytomorphogenesis in plants. Cell biology monographs, 8th edn. Springer, Wien, pp 401–421. https://doi.org/10.1007/978-3-7091-8602-2_15
Skvarla JJ, Larsen DA (1965) An electron microscopic study of pollen morphology in the Compositae with special reference to the Ambrosiinae. Grana 6:210–269
Southworth D (1983) Exine development in Gerbera jamesonii (Asteraceae: Mutisieae). Am J Bot 70:1038–1047. https://doi.org/10.1002/j.1537-2197.1983.tb07904.x
Stix E (1960) Pollenmorphologische Untersuchungen an Compositen. Grana 2:41–104
Takahashi M (1989) Development of the echinate pollen wall in Farfugium japonicum (Compositae: Senecioneae). Bot Mag Tokyo 102:219–239
Taylor ML, Osborn JM (2006) Pollen ontogeny in Brasenia (Cabombaceae, Nymphaeales). Am J Bot 93:344–356
Taylor ML, Hudson PJ, Rigg JM, Strandquist JN, Green JS, Thiemann TC, Osborn JM (2012) Tapetum structure and ontogeny in Victoria (Nymphaeaceae). Grana 51:107–118
Taylor ML, Hudson PJ, Rigg JM, Strandquist JN, Green JS, Thiemann TC, Osborn JM (2013) Pollen ontogeny in Victoria (Nymphaeales). Int J Plant Sci 174:1259–1276. https://doi.org/10.1086/673246
Taylor ML, Cooper RL, Schneider EL, Osborn JM (2015) Pollen structure and development in Nymphaeales: insights into character evolution in an ancient angiosperm lineage. Am J Bot 102:1685–1702. https://doi.org/10.3732/ajb.1500249
Thio BJR, Lee JH, Carson Meredith J (2009) Characterization of ragweed pollen adhesion to polyamides and polystyrene using atomic force microscopy. Environ Sci Technol 43:4308–4313. https://doi.org/10.1021/es803422s
Tompson DA (1917) On growth and form. Cambridge University Press, Cambridge
Varotto S, Parrini P, Mariani P (1996) Pollen ontogeny in Cichorium intybus L. Grana 35:154–161. https://doi.org/10.1080/00173139609429076
Vinckier S, Smets E (2005) A histological study of microsporogenesis in Tarenna gracilipes (Rubiaceae). Grana 44:30–44
Walles B, Rowley JR (1982) Cell differentiation in microsporangia of Pinus sylvestris with special attention to the tapetum. I. The pre- and early-meiotic periods. Nord J Bot 2:53–70
Willemse MTM (1971) Morphological and quantitative changes in the population of cell organelles during microsporogenesis of Pinus sylvestris L. II. Morphological changes from prometaphase I until the tetrad stage. Acta Bot Neerl 20:411–427. https://doi.org/10.1111/j.1438-8677.1971.tb00727.x
Wodehouse RP (1928) Pollen grains in the identication and classification of plants. I. The Ambrosiaceae. Bull Torrey Bot Club 55:181–198
Wodehouse RP (1934) An oil drop theory of pollen-grain pattern formation. Am J Bot 21:18–22
Acknowledgements
The work was carried out in the framework of the institutional research project “Morphology, ultrastructure and development of sporoderm in higher plants” of the Komarov Botanical Institute of the Russian Academy of Sciences (no. АААА-А18-118032190134-3) on the equipment of the Centre for Collective Use (“Cellular and molecular technology study of plants and fungi”) of the Komarov Botanical Institute (St. Petersburg). We also thank our anonymous reviewers for their valuable advice and suggestions.
Author information
Authors and Affiliations
Contributions
Conceptualization: N.G. and S.B.
Methodology: S.P., E.S., O.V., and V.G.
Investigation: N.G.
Writing of the original draft, review, and editing: N.G. and S.B.
Funding acquisition: N.G.
Resources: N.G.
Supervision: N.G. and S.B.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Peter Nick
Rights and permissions
About this article
Cite this article
Gabarayeva, N., Polevova, S., Grigorjeva, V. et al. Suggested mechanisms underlying pollen wall development in Ambrosia trifida (Asteraceae: Heliantheae). Protoplasma 256, 555–574 (2019). https://doi.org/10.1007/s00709-018-1320-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-018-1320-3