Abstract
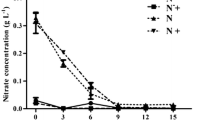
Neochloris oleoabundans is considered one of the most promising oil-rich microalgae because of its ability to store lipids under nitrogen starvation. However, high biomass densities, required for applications on medium to large scale, are not reached in this condition of growth. As previous studies on other microalgae have shown that mixotrophy allows to obtain higher biomass in comparison to autotrophic cultures, we performed morphophysiological analyses in order to test the mixotrophic growth capability of N. oleoabundans. A carbon-rich manure derived from the apple vinegar production (AWP) was added to the medium. Cells were also cultivated under nutrient starvation (tap water), to observe the expected lipids accumulation, and combining AWP to water, to test the potential of this waste in a low-cost culture system. The results highlighted that AWP in the medium allowed to obtain the highest final cell density. Moreover, starch granules were stored inside chloroplast at the beginning of the experiment. The presence of AWP did not induce variations on light harvesting complex II (LHCII)–photosystem II (PSII) assembly, even if an interesting promotion of pigment synthesis in cells was observed. On the other hand, in starved cells, chloroplast degeneration, pigment content decrease, altered LHCII–PSII assembly and accumulation of high amount of lipid globules were observed, irrespective of the presence of AWP. The results suggest that mixotrophy promotes growth in N. oleoabundans and open up the possibility of using waste products from agri-food industries for this purpose. After growth, cells could be transferred under nutrient starvation to induce lipid accumulation.








Similar content being viewed by others
References
Akazawa K, Okamoto K (1980) Biosynthesis of sucrose. In: Preiss J (ed) The biochemistry of plants, vol 3. Academic, New York, pp 199–218
Andrade MR, Costa JAV (2007) Mixotrophic cultivation of microalga Spirulina platensis using molasses as organic substrate. Aquaculture 264:130–134
Baldisserotto C, Ferroni L, Andreoli C, Fasulo MP, Bonora A, Pancaldi S (2005) Dark-acclimation of the chloroplast in Koliella antarctica exposed to a simulated austral night condition. Arctic Antarct Alpine Res 37:146–156
Borowitzka MA (1998) Vitamins and fine chemicals from microalgae. In: Borowitzka MA, Borowitzka LJ (ed) Micro-algal biotechnology. Oxford University Press, Oxford, pp 153–196
Brennan L, Owende P (2010) Biofuels from microalgae—a review of technologies for production, processing, and extractions of biofuels and co-products. Renew Sustain Energ Rev 14:557–577
Chisti Y (2007) Biodiesel from microalgae. Biotech Adv 25:294–306
de la Jara A, Mendoza H, Martel A, Molina C, Nordströn L, de la Rosa V, Diaz R (2003) Flow cytometric determination of lipid content in a marine dinoflagellate, Crypthecodinium cohnii. J App Phycol 15:433–438
Ferroni L, Baldisserotto C, Pagnoni A, Fasulo MP, Pancaldi S (2004) Adaptive modifications of the photosynthetic apparatus in Euglena gracilis Klebs exposed to manganese excess. Protoplasma 224:167–177
Ferroni L, Baldisserotto C, Pantaleoni L, Billi P, Fasulo MP, Pancaldi S (2007a) High salinity alters chloroplast morpho-physiology in a freshwater Kichneriella species (Selenastraceae) from Ethipian Lake Awasa. Am J Bot 94:1973–1983
Ferroni L, Baldisserotto C, Zennaro V, Soldani C, Fasulo MP, Pancaldi S (2007b) Acclimation to darkness in the marine chlorophyte Koliella antarctica culture under low salinity: hypotheses on its origin in the polar environment. Eur J Phycol 42:91–104
Ferroni L, Baldisserotto C, Pantaleoni L, Fasulo MP, Fagioli P, Pancaldi S (2009) Degreening of the unicellular alga Euglena gracilis: thylakoid composition, room temperature fluorescence spectra and chloroplast morphology. Plant Biology 11:631–641
Ferroni L, Baldisserotto C, Giovanardi M, Pantaleoni L, Morosinotto T, Pancaldi S (2011) Revised assignment of room-temperature chlorophyll fluorescence emission bands in single living cells of Chlamydomonas reinhardtii. J Bioenerg Biomembr 43:163–173
Geider RJ, MacIntyre HI, Graziano LM, McKay RML (1998) Responses of the photosynthetic apparatus of Dunaliella tertiolecta (Chlorophyceae) to nitrogen and phosphorus limitation. Eur J Phycol 15:1–11
Gouveia L, Oliveira AC (2008) Microalgae as a raw material for biofuels production. J Ind Microbiol Biotechnol 36:269–274
Gouveia L, Marques AE, Lopes da Silva T, Reis A (2009) Neochloris oleoabundans UTEX #1185: a suitable renewable lipid source for biofuel production. J Ind Microbiol Biotechnol 36:821–826
Gushina IA, Harwood JL (2006) Lipids and lipid metabolism in eukaryotic algae. Prog Lipid Res 45:160–186
Heredia-Arroyo T, Wei W, Hu B (2010) Oil accumulation via heterotrophic/mixotrophic Chlorella protothecoides. Appl Biochem Biotech 162:1978–1995
Hu H, Gao K (2003) Optimization of growth and fatty acid composition of a unicellular marine picoplankton, Nannochloropsis sp., with enriched carbon sources. Biotechnol Lett 25:421–425
Hu Q, Sommerfeld M, Jarvis E, Ghirardi M, Posewitz M, Seibert M, Darzins A (2008) Microalgal triacylglycerols as a feedstocks for biofuel production: perspective and advances. Plant J 54:621–639
Ip P, Wong K, Chen F (2004) Enhanced production of astaxanthin by the green microalga Chlorella zofingiensis in mixotrophic culture. Process Biochem 39:1761–1776
Issa AA, Abdel-Basset RM, Adam MS (1995) Abolition of heavy metal toxicity on Kirchneriella lunaris (Chlorophyta) by calcium. Ann Bot 75:189–192
Kang R, Wang J, Shi D, Cong W, Cai Z, Ouyang F (2004) Interactions between organic and inorganic carbon sources during mixotrophic cultivation of Synechococcus sp. Biotechnol Lett 26:1429–1432
Kobayashi M, Kakizono T, Yamaguchi K, Nishio N, Nagai S (1992) Growth and astaxanthin formation of Haematococcus pluvialis in heterotrophic and mixotrophic conditions. J Ferment Bioeng 74:17–20
Latasa M, Berdalet E (1994) Effect of nitrogen and phosphorous starvation on pigment composition of cultured Heterocapsa sp. J Plankton Res 16:83–94
Lee Y (2001) Microalgal mass culture systems and methods: their limitation and potential. J Appl Phycol 13:307–315
Leonardi PI, Popovich CA, Damiani MC (2011) Feedstocks for second-generation biodiesel: microalgae's biology and oil composition. In: dos Santos Bernardes MA (ed) Economic effects of biofuel production. Tech, Croatia, pp 318–346
Levine RB, Costanza-Robinson MS, Spatafora GA (2011) Neochloris oleoabundans grown on anaerobically digested dairy manure for concomitant nutrient removal and biodiesel feedstock production. Biomass Bioenerg 35:40–49
Li Y, Horsman M, Wang B, Wu N, Lan CQ (2008a) Biofuels from microalgae. Biotechnol Prog 24:815–820
Li Y, Horsman M, Wang B, Wu N, Lan CQ (2008b) Effect on nitrogen sources on cell growth and lipid accumulation of green alga Neochloris oleoabundans. Appl Microbiol Biotechnol 81:629–636
Liang Y, Sarkany N, Cui Y (2009) Biomass and lipid productivities of Chlorella vulgaris under autotrophic, heterotrophic and mixotrophic growth conditions. Biotechnol Lett 31:1043–1049
Marquez FJ, Nishio N, Nagai S, Sasaki K (1995) Enhancement of biomass and pigment production during growth of Spirulina platensis mixotrophic culture. J Chem Tech Biotechnol 62:159–164
Martinez F, Orus MI (1991) Interactions between glucose and inorganic carbon metabolism in Chlorella vulgaris strain UAM101. Plant Physiol 95:1150–1155
Mata TM, Martins AA, Caetano NS (2010) Microalgae for biodiesel production and other applications: a review. Renew Sustain Energ Rev 14:217–232
Nagle N, Lemke P (1990) Production of methyl ester fuel from microalgae. Appl Biochem Biotech 24(25):355–361
Ogawa T, Aiba S (1981) Bioenergetic analysis of mixotrophic growth in Chlorella vulgaris and Scenedesmus acutus. Biotechnol Bioeng 23:1121–1132
Osterlind S (1950) Inorganic carbon sources of green algae: II. Carbonic anhydrase in Scenedesmus quadricauda and Chlorella pyrenoidosa. Physiol Plantarum 3:430–434
Osterlind S (1951) Inorganic carbon sources of green algae. IV. Photoactivation of some factor necessary for bicarbonate assimilation. Physiol Plantarum 4:514–527
Pancaldi S, Bonora A, Baldisserotto C, Gualandri R, Neri LM, Fasulo MP (2001) De-regulation of light-induced plastidogenesis in etiolated Euglena gracilis Klebs treated with DNA hypermethylating 3’-azido-3’-deoxythymidine. Plant Biology 3:524–535
Pancaldi S, Baldisserotto C, Ferroni L, Bonora A, Fasulo MP (2002) Room temperature microspectrofluorimetry as a useful tool for studying the assembly of the PSII chlorophyll–protein complexes in single living cells of etiolated Euglena gracilis Klebs during the greening process. J Exp Bot 53:1753–1763
Pruvost J, Van Vooren G, Cogne G, Legrand J (2009) Investigation of biomass and lipids production with Neochloris oleoabundans in photobioreactor. Bioresource Technol 100:5988–5995
Pulz O, Gross W (2004) Valuable products from biotechnology of microalgae. Appl Microbiol Biotechnol 65:635–648
Raven JA (1968) The mechanism of photosynthetic use of HCO3 by Hydrodictyon africanum. J Exp Bot 19:193–206
Sawayama S, Inoue S, Dote Y, Yokoyama SY (1995) CO2 fixation and oil production through microalga. Energ Conver Manage 36:729–731
Scott SA, Davey MP, Dennis JS, Horst I, Howe CJ, Lea-Smith DJ, Smith AG (2011) Biodiesel from algae: challenges and prospects. Curr Opin Biotech 21:277–286
Shenk PM, Thomas-Hall SR, Stephens E, Marx UC, Mussgnug JH, Posten C, Kruse O, Hankamer B (2008) Second generation biofuels: high-efficiency microalgae for biodiesel production. Bioenerg Res 1:20–43
Shi XM, Chen F (1999) Production and rapid extraction of lutein and the other lipid-soluble pigments from Chlorella protothecoides grown under heterotrophic and mixotrophic conditions. Nahrung 43:109–113
Šiffel P, Braunová Z (1999) Release and aggregation of the light harvesting complex in intact leaves subjected to strong CO2 deficit. Photosynth Res 61:217–226
Smith FA (1968) Rates of photosynthesis in characean cells. II. Photosynthetic 14CO2 fixation and 14C-bicarbonate uptake by characean cells. J Exp Bot 19:207–217
Spolaore P, Joannis-Cassan C, Duran E, Isambert A (2006) Commercial applications of microalgae. J Biosci Bioeng 101:87–96
Stephens E, Ross IL, Mussgnug JH, Wagner LD, Borowitzka MA, Posten C, Kruse O, Hankamer B (2010) Future prospects of microalgal biofuel production systems. Trends Plant Sci 15:554–564
Tornabene TG, Holzer G, Lien S, Burris N (1983) Lipid composition of the nitrose starved green alga Neochloris oleoabundans. Enzyme Microb Tech 5:435–440
Wang B, Lan CQ (2011) Biomass production and nitrogen and phosphorus removal by the green alga Neochloris oleoabundans in simulated wastewater and secondary municipal wastewater effluent. Bioresource Technol 102:5639–5644
Welburn AR (1994) The spectral determination of Chlorophylls a and b, as well as total carotenoids, using various solvents with spectrophotometer of different resolution. Plant Physiol 144:307–313
Wijffels RH, Barbosa MJ (2011) An outlook on microalgal biofuels. Science 329:796–799
Xu H, Miao X, Wu K (2006) High quality biodiesel production from a microalga Chlorella prototecoides by heterotrophic growth in fermenters. J Biotechnol 126:499–507
Yang C, Hua Q, Shimizu K (2000) Energetics and carbon metabolism during growth of microalgal cells under photoautotrophic, mixotrophic and cyclic light-autotrophic/dark-heterotrophic conditions. Biochem Eng J 6:87–102
Yang Y, Xu J, Vail D, Weathers P (2011) Ettlia oleoabundans growth and oil production on agricultural anaerobic waste effluents. Bioresource Technol 102:5076–5082
Young EB, Beardall J (2003) Photosynthetic function in Dunaliella tertiolecta (Chlorophyta) during a nitrogen starvation and recovery cycle. J Phycol 39:897–905
Yu H, Jia S, Dai Y (2009) Growth characteristics of the cyanobacterium Nostoc flagelliforme in photoautotrophic, mixotrophic and heterotrophic cultivation. J Appl Phycol 21:127–133
Acknowledgements
This work was financially supported by grants from the Consorzio Universitario Italiano per l’Argentina (CUIA) and from the University of Ferrara.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Peter Nick
Rights and permissions
About this article
Cite this article
Giovanardi, M., Ferroni, L., Baldisserotto, C. et al. Morphophysiological analyses of Neochloris oleoabundans (Chlorophyta) grown mixotrophically in a carbon-rich waste product. Protoplasma 250, 161–174 (2013). https://doi.org/10.1007/s00709-012-0390-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-012-0390-x