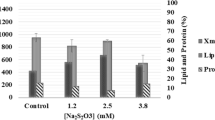
Abstract
Energy crises, global warming, and climatic changes call for technological and commercial advances in manufacturing high-quality transportation fuels from unconventional feedstocks. Microalgae is one of the most promising sources of biofuels due to the high yields attained per unit area and because it does not displace food crops. Neochloris oleabundans (Neo) microalga is an important promising microbial source of single-cell oil (SCO). Different experimental growth and lipid production conditions were evaluated and compared by using optical density (540 nm), dry-weight determination, and flow cytometry (FC). Best Neo average biomass productivity was obtained at 30°C under conditions of nitrogen-sufficiency and CO2 supplementation (N+/30°C/CO2), with an average doubling time of 1.4 days. The second and third highest productivities occurred with N-sufficient cultures without CO2 supplementation at 26°C (N+/26°C) and at 30°C (N+/30°C), with doubling times of 1.7 and 2.2 days, respectively. Microbial lipid production was monitored by flow cytometry using Nile red (NR), a lipophilic fluorochrome that possesses several advantageous characteristics for in situ screening near real time (at line). Results showed maximum lipid content (56%) after 6 days of nitrogen depletion under nitrogen starvation without CO2 supplementation (N−/30°C), followed by N−/30°C/CO2 and N−/26°C conditions with 52% lipid content, after 5 and 6 days of N starvation, respectively. The adequate fatty acid profile and iodine value of Neo lipids reinforced this microalga as a good source of SCO, in particular for use as biodiesel.





Similar content being viewed by others
References
American Public Health Association, America Water Works Association, and Water Environment Federation (1998) Standard methods for the examination of water and wastewater. NO3 − ultraviolet spectrophotometric screening method, 20th edn. APHA, AWWA, WEF, Washington DC, pp 4–115
Bligh EG, Dyer WJ (1959) A rapid method for total lipid extraction and purification. Can J Biochem Physiol 37:911–917
Bourelly P (1972) Les algues D’eu Douce: algues vertes. N. Boubée & Cie, France
Chelf P (1990) Environmental control of lipid and biomass production in two diatom species. J Appl Phycol 2:121–129. doi:10.1007/BF00023373
Chisti Y (2007) Biodiesel from microalgae. Biotechnol Adv 25:294–306. doi:10.1016/j.biotechadv.2007.02.001
Chisti Y (2008) Biodiesel from microalgae beats bioethanol. Trends Biotechnol 26:126–131. doi:10.1016/j.tibtech.2007.12.002
da Silva TL, Reis A, Medeiros R, Oliveira C, Gouveia L (2008) Oil production towards biofuel from autotrophic microalgae semi-continuous cultivations monitorized by flow cytometry. Appl Biochem Biotechnol. doi:10.1007/s12010-008-8443-5
de la Jara A, Mendoza H, Martel A, Molina C, Nordström L, de la Rosa V, Diaz R (2003) Flow cytometric determination of lipid content in a marine dinoflagellate, Crypthecodinium cohnii. J Appl Phycol 15:433–438. doi:10.1023/A:1026007902078
Elsey D, Jameson D, Raleigh B, Cooney MJ (2007) Fluorescent measurement of microalgal neutral lipids. J Microbiol Methods 68:639–642. doi:10.1016/j.mimet.2006.11.008
European Standard EN 14111 (2003) Fat and oil derivatives—fatty acid methyl esters (FAME)—determination of iodine value
European Standard EN 5509 (2000) Animal and vegetable fats and oils—preparation of methyl esters of fatty acids
European Standard EN 14103 (2003) Fat and oil derivatives—fatty acid methyl esters (FAME)—determination of ester and linolenic acid methyl ester contents
European Standard EN 14214 (2004) Automotive fuels—fatty acid methyl esters (FAME) for diesel engines—requirements and test methods. AFNOR, Saint-Denis
Firestone D (ed) (1990) Oils and fats. In: Official methods of analysis of the AOAC. AOAC, Champaign, IL, pp 951–986
Gouveia L, Oliveira C (2008) Microalgae as a raw material for biofuels production. J Ind Microbiol Biotechnol 36:269–274. doi:10.1007/s10295-008-0495-6
James DE (1978) Culturing algae. Carolina Biological Supply Company, Burlington, NC, USA
Rodolfi L, Chini Zittelli G, Bassi N, Padovani G, Biondi N, Bonini G, Tredici MR (2008) Microalgae for oil: strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol Bioeng 102(1):110–112. doi:10.1002/bit.22033
Sheehan J, Dunahay T, Benemann J, Roessler P (1998) A look back at the US Department of Energy’s Aquatic Species Program—biodiesel from algae. National Renewable Energy Laboratory, Golden, CO, USA
Suen H, Hubbard JS, Holzer G, Tornabene TG (1987) Total lipid production of the green alga Nannochloropsis sp QII under different nitrogen regimes. J Phycol 23:289–296. doi:10.1111/j.1529-8817.1987.tb04137.x
Taylor G (2008) Biofuels and the biorefinery concept. Energy Policy 36:4406–4409. doi:10.1016/j.enpol.2008.09.069
Tornabene TG, Holzer G, Lien S, Burris N (1983) Lipid composition of the nitrogen starved green alga Neochloris oleabundans. Enzyme Microb Technol 5:435–440. doi:10.1016/0141-0229(83)90026-1
Aknowledgements
The authors would like to acknowledge Doutora Narcisa Bandarra from IPIMAR for the fatty acid analysis and also Doutora Ana Cristina Oliveira for the iodine value determinations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gouveia, L., Marques, A.E., da Silva, T.L. et al. Neochloris oleabundans UTEX #1185: a suitable renewable lipid source for biofuel production. J Ind Microbiol Biotechnol 36, 821–826 (2009). https://doi.org/10.1007/s10295-009-0559-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-009-0559-2