Summary.
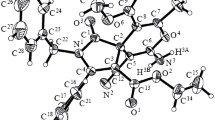
The reaction of 2-benzylidenecyclohexanone and 1-(4-methoxyphenyl)-2-methyl-1-penten-3-one with guanidine did not yield the expected 4-phenylhexahydro-2-quinazolinamine and 4-(p-methoxyphenyl)-dihydro-2-pyrimidinamine, respectively, but nitrogen-free products which turned out as [3+3]- and [4+2]-cycloadducts of two molecules of the applied vinylogous ketone each. According to elemental analyses, mass spectra, and, in particular, NMR analyses (1H and 13CNMR, HH-COSY, gs-HSQC, gs-HMBC, 1D TOCSY, NOESY, and 1D NOE difference spectra), the prepared dimers were identified as racemic (5E)-5-benzylidene-9β-phenyl-trans-4a-1,2,3,4,4a,5,6,7,8,9a-decahydroxanthen-4aα-ol and 3β,5β-bis-(4-methoxyphenyl)-2α,4β,6α-trimethyl-4α- propionylcyclohexanone, respectively. Structure and stereochemistry of the dimers are elucidated, and mechanisms for their formation are proposed.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received April 11, 2001. Accepted (revised) May 18, 2001
Rights and permissions
About this article
Cite this article
Gößnitzer, E., Wendelin, W., Schermanz, K. et al. Special Reactions of α,β-Unsaturated Ketones III [1]. Formation and Structure Elucidation of Dimers of 2-Aryl-methyleneketones: (5E)-5-Benzylidene-9β-phenyl-trans-4a-1,2,3,4,4a,5,6,7,8,9a-decahydroxanthen-4aα-ol and 3β,5β-Bis-(4-methoxyphenyl)-2α,4β,6α-trimethyl-4α-propionyl-cyclohexanone [2]. Monatshefte fuer Chemie 132, 1081–1093 (2001). https://doi.org/10.1007/s007060170049
Issue Date:
DOI: https://doi.org/10.1007/s007060170049