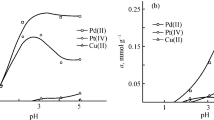
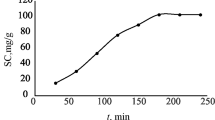
Abstract
Natural sources of noble metals (Ru, Rh, Pd, Os, Ir, Pt, Au) are exhaustible, thus the extraction of metal from anthropogenic sources seems very promising. This work presents an investigation of new N-heterocyclic sorbents based on polyvinylpyridine for reversible extraction of noble metals from complex matrix solutions. This type of sorbents shows good resistance to aggressive acidic media and sufficient sorption capacity in both dynamic and static modes. It was found, to achieve quantitative sorption of Rh and Ir it is necessary to convert these analytes completely into their chloride form at the stage of sample preparation. Based on a series of experimental data, possible mechanisms of retention of noble metals’ chlorocomplexes on this type of sorbents are proposed. The UV–Vis spectra of various noble metals chlorocomplexes were studied in the presence of model compounds that simulate the repeating units of the sorbents. We assume that the chlorocomplexes of Rh, Ir, and possibly Ru form simple ion pairs with positively charged sorption sites. While Pd, Pt, and Au form more stable complexes (possibly intra-sphere) with sorbent units. The mechanism of Os sorption is unclear and requires a more detailed investigation. This information helps to select suitable desorption solutions for the extraction of analytes from the sorbent. The desorption solution should contain electrolytes, acid, and complexing reagents necessary for the quantitative recovery of noble metals due to the different sorption mechanisms and the various contribution of electrostatic forces to the efficiency of chlorocomplexes retention.
Graphic abstract








Similar content being viewed by others
References
Fontàs C, Hidalgo M, Salvadó V (2009) Solvent Extr Ion Exch 27:83
Kononova ON, Goncharova EL, Melnikov AM, Kashirin DM, Kholmogorov AG, Konontsev SG (2010) Solvent Extr Ion Exch 28:388
Kramer J, Driessen WL, Koch KR, Reedijk J (2005) Sep Sci Technol 39:63
Harland CE (1994) Ion exchange theory and practice. The Royal Society of Chemistry, Cambridge, p 285
Xue D, Wang H, Liu Y, Shen P (2015) Miner Eng 81:149
Kramer J, Driessen WL, Koch KR, Reedijk J (2002) Hydrometallurgy 64:59
Hidalgo M, Uheida A, Salvadó V, Fontàs C (2006) Solvent Extr Ion Exch 24:931
Pavlova LA, Davankov VA, Lependina OL (2014) Sorbtsionnyye i khromatograficheskiye protsessy 14:75
Karimi M, Feiz M, Aboufazeli F, Hamid R, Sadeghi O (2012) Curr World Environ 7:227
Lotfi Zadeh Zhad HR, Sadeghi O, Amini MM, Tavassoli N, Banitaba MH, Davarani SSH (2011) Sep Sci Technol 46:648
Garcia L, Torrent A, Anticó E, Fontàs C, Roglans A (2008) React Funct Polym 68:1088
Usher A, McPhail DC, Brugger J (2009) Geochim Cosmochim Acta 73:3359
Zolotov YA, Varshal GM, Ivanov VM (2003) Analytical chemistry of platinum group metals. Editorial URSS, Moscow, p 592
Dubenskiy AS, Yakurnova ED, Krasilnikova YA, Seregina IF, Pavlova LA, Davankov VA, Bolshov MA (2020) Anal Lett 53:1266
Dubenskiy AS, Yakurnova ED, Seregina IF, Pavlova LA, Tsyurupa MP, Davankov VA, Bol’shov MA (2018) Inorg Mater 54:17
Hughes EB, Jellinek HHG, Ambrose BA (1949) J Phys Chem 53:410
Acknowledgements
The financial support from the Russian Foundation for Basic Research (RFBR), Grant 20-03-00354A is acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Maksimova, Y.A., Dubenskiy, A.S., Davankov, V.A. et al. Conditions and mechanisms of noble metals ions sorption in the process of their preconcentration on the new polyvinylpyridine sorbents. Monatsh Chem 151, 1291–1303 (2020). https://doi.org/10.1007/s00706-020-02662-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-020-02662-x