Abstract
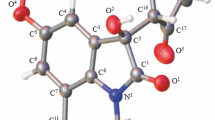
A novel and efficient annulation of pentane-2,4-dione with 2-(2-bromophenyl)quinazolin-4(3H)-one was disclosed in DMF catalyzed by CuI/l-proline in the presence of Cs2CO3. This procedure experienced the consecutive α-arylation of carbonyl, deacylation but no dehydration reaction for the synthesis of 6-hydroxy-6-methyl-5,6-dihydro-8H-isoquinolino[1,2-b]quinazolin-8-ones in good yields.
Graphical abstract



Similar content being viewed by others
References
Ayala-Ramirez M, Feng L, Habra MA, Rich T, Dickson PV, Perrier N, Phan A, Waguespack S, Patel S, Jimenez C (2012) Cancer 118:2804
Lantermann AB, Chen D, McCutcheon K, Hoffman G, Frias E, Ruddy D, Akiec RD, Korn J, McAllister G, Stegmeier F, Meyer MJ, Sharma SV (2015) Cancer Res 75:4937
Chen CH, Statt S, Chiu CL, Thai P, Arif M, Adler KB, Wu R (2014) Am J Respir Crit Care Med 190:1127
Carli P, Militello L, Miolo GM, Quitadamo D, Lombardi D, Torrisi E, Scalone S, Crivellari D, Spazzapan S (2014) Tumori 100:605
Odia Y, Shih JH, Kreisl TN, Fine HA (2014) J NeuroOncol 120:431
Kamal A, Babu KS, Chandrasekhar C, Nagaraju B, Visweswara Sastry KN, Ganesh Kumar C (2015) Tetrahedron Lett 56:6373
Beaudegnies R, Quaranta Murphy L, Kessabi F, Lamberth C, Knauf-Beiter G, Fraser T (2016) Bioorg Med Chem 24:444
Zhang H, Jin P, Wang S, Li F, Guan L, Quan Z (2015) Arch Pharm 348:564
El-Hashash MA, Elshahawi MM, Ragab EA, Nagdy S (2015) Synth Commun 45:2240
Paine HA, Nathubhai A, Woon ECY, Sunderland PT, Wood PJ, Mahon MF, Lloyd MD, Thompson AS, Haikarainen T, Narwal M, Lehtio L, Threadgill MD (2015) Bioorg Med Chem 23:5891
Park J, Lee G, Lee S, Hien TT, Kim S, Yang JW, Cho J, Ko H, Lim S, Kim Y, Kang K, Kim Y (2015) J Med Chem 58:2114
Notte GT (2013) Ann Rep Med Chem 48:451
Pyatakov DA, Sokolov AN, Astakhov AV, Chernenko AY, Fakhrutdinov AN, Rybakov VB, Chernyshev VV, Chernyshev VM (2015) J Org Chem 80:10694
Zhang Y, Xu J, Li C, Wang XS (2015) Tetrahedron 71:8732
Nowak M, Malinowski Z, Fornal E, Jozwiak A, Parfieniuk E, Gajek G, Kontek R (2015) Tetrahedron 71:9463
Laha JK, Tummalapalli KSS, Nair A, Patel N (2015) J Org Chem 80:11351
Duan Z, Gao Y, Yuan D, Ding M (2015) Synlett 26:2598
Zhang M, Zhang H, Ruan W, Wen T (2015) Eur J Org Chem 2015:5914
Fuentes N, Kong W, Fernandez-Sanchez L, Merino E, Nevado C (2015) J Am Chem Soc 137:964
Bonetti A, Beccalli E, Caselli A, Clerici F, Pellegrino S, Gelmi ML (2015) Chem Eur J 21:1692
Sole D, Mariani F, Fernandez I (2014) Adv Synth Catal 356:3237
Gawande SD, Zanwar MR, Kavala V, Kuo C-W, Rajawinslin RR, Yao C-F (2015) Adv Synth Catal 357:168
Reddy PG, Reddy ID, Manjula A (2018) Synlett 2018:1717
Xie F, Chen Q-H, Xie R, Jiang H-F, Zhang M (2018) ACS Catal 8:5869
Yamashita M, Nishizono Y, Himekawa S, Iida A (2016) Tetrahedron 72:4123
Shawakfeh KQ, Ishtaiwi ZN, Al-Said NH (2014) Tetrahedron Lett 55:1329
Hannigan K, Kulkarni SS, Bdzhola VG, Golub AG, Yarmoluk SMT, Tanaji T (2013) Bioorg Med Chem Lett 23:5790
Neel AJ, Hehn JP, Tripet PF, Toste FD (2013) J Am Chem Soc 135:14044
Adepu R, Sunke R, Meda CLT, Rambabu D, Krishna GR, Reddy CM, Deora GS, Parsa KVL, Pal M (2013) Chem Commun 49:190
Kumar KS, Kumar PM, Reddy MA, Ferozuddin M, Sreenivasulu M, Jafar AA, Krishna GR, Reddy CM, Rambabu D, Shiva Kumar K, Sarbani P, Manojit P (2011) Chem Commun 47:10263
Palucki M, Buchwald SL (1997) J Am Chem Soc 119:11108
Hamann BC, Hartwig JF (1997) J Am Chem Soc 119:12382
Fu WC, So CM, Yuen OY, Lee ITC, Kwong FY (2016) Org Lett 18:1872
Zheng B, Jia TZ, Walsh PJ (2013) Org Lett 15:4190
Xie RL, Ling Y, Fu H (2012) Chem Commun 48:12210
Shi Y, Zhu X, Mao H, Hu H, Zhu C, Cheng Y (2013) Chem Eur J 19:11553
Danoun G, Tlili A, Mounnier F, Taillefer M (2012) Angew Chem Int Ed 51:12815
Lu YJ, Fu H (2011) J Org Chem 76:4600
Lei AW, He C, Guo S, Huang L (2010) J Am Chem Soc 132:8273
Miao WQ, Liu JQ, Wang XS (2017) Org Biomol Chem 15:5325
Li C, Zhang WT, Wang XS (2014) J Org Chem 79:5847
Feng BB, Liu JQ, Wang XS (2017) J Org Chem 82:1817
Liu JQ, Ma YG, Zhang MM, Wang XS (2017) J Org Chem 82:4918
Chen DS, Zhang MM, Li YL, Liu Y, Wang XS (2014) Tetrahedron 70:2889
Li C, Zhang WT, Wang XS (2014) Tetrahedron 70:8919
Acknowledgements
This work was financially supported by NSFC of China (No. 21702078), Natural Science Foundation of Jiangsu Higher Education Institution (17KJA150003), the Priority Academic Program Development of Jiangsu Higher education Institution, and TAPP.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Miao, WQ., Liu, JQ. & Wang, XS. An efficient synthesis of 6-hydroxy-6-methyl-5,6-dihydro-8H-isoquinolino[1,2-b]quinazolin-8-ones via a CuI-catalyzed deacylation and no dehydration reaction. Monatsh Chem 150, 1305–1315 (2019). https://doi.org/10.1007/s00706-019-02430-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-019-02430-6