Abstract
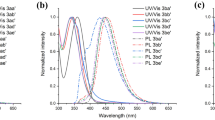
A series of 2′,7′-diarylspiro[cyclopentane-1,9′-fluorene] symmetrical derivatives containing different terminal aromatic units have been synthesized by employing palladium-catalyzed C–C cross-coupling reactions and characterized with respect to the optical and thermal properties. Their absorption and fluorescence properties were investigated. These compounds possess good fluorescence-emitting ability with φ FL values in the region of 0.67–0.92 in solution and display strong blue emission. The derivatives also showed good thermal stabilities with decomposition temperatures (T d) in the range of 265–386 °C. Structure–optical behavior characteristics and further details of the electronic properties from cyclic voltammetry measurements and theoretical calculations were discussed.
Graphical abstract






Similar content being viewed by others
References
Komino T, Nomura H, Koyanagi T, Adachi C (2013) Chem Mater 25:3038
Wang CL, Dong HL, Hu WP, Liu YQ, Zhu DB (2012) Chem Rev 112:2208
He GS, Tan LS, Zheng QD, Prasad P (2008) Chem Rev 108:1245
Wu WC, Heh HC, Chan LH, Chen CT (2002) Adv Mater 14:1072
Okumoto K, Kanno H, Hamaa Y, Takahashi H, Shibata K (2006) Appl Phys Lett 89:063504
Han J, An J, Im C, Cho NS, Shim HK, Majima T (2009) J Photochem Photobiol A 205:98
Lin BC, Qiu L, Qiu B, Peng Y, Yan FA (2011) Macromolecules 44:9642
Lakhwani G, Janssen R, Meskers S (2009) J Phys Chem B 113:14165
Meng G, Velayudham S, Smith A, Luck R, Liu HY (2009) Macromolecules 42:1995
Lee TH, Lüssem B, Kim K, Giri G, Bao ZN (2013) ACS Appl Mater Inter 5:2337
Seo HS, Kim DK, Oh JD, Shin ES, Choi JH (2013) J Phys Chem C 117:4764
Lai WY, Zhu R, Fan QL, Hou LT, Cao Y, Huang W (2006) Macromolecules 39:3707
Wang ZX, Shao HX, Ye JC, Zhang L, Lu P (2007) Adv Funct Mater 17:253
Jung SH, Suh DH, Cho HN (2003) Polym Bull 50:251
Tyler AM, Paul GW, Jacqueline M (2013) J Phys Chem C 117:8429
Tirapattur S, Belletete M, Drolet N, Bouchard J, Ranger M, Leclerc M, Durocher G (2002) J Phys Chem B 106:8959
Kotaka H, Konishi G, Mizuno K (2010) Tetrahedron Lett 51:181
Xing X, Xiao LX, Zheng LL, Hu SY, Chen ZJ, Qu B, Gong QH (2012) J Mater Chem 22:15136
Lee H, Oh J, Chu HY, Lee JI, Kim SH, Yang YS (2003) Tetrahedron 59:2773
Wu FI, Shu CF, Chien CH, Tao YT (2005) Synth Met 148:133
Wong KT, Wang ZJ, Chien YY, Wang CL (2001) Org Lett 3:2285
Li ZH, Wong MS (2006) Org Lett 8:1499
Hughes G, Wang CS, Batsanow AS, Fern M, Frank S, Bryce MR (2003) Org Biomol Chem 1:3069
Leclerc MJ (2001) Polym Chem 39:2867
Neher D (2001) Macromol Rapid Commun 22:1365
Scherf U, List EJM (2002) Adv Mater 14:477
Wong KT, Fang F, Peng YC (2006) Org Lett 8:1415
Destri S, Pasini M, Botta C, Porzio W, Marchi FBL (2002) J Mater Chem 12:924
Destri S, Porzio W, Botta C, Pasini M, Bertini F (2001) Synth Met 121:1647
Destri S, Porzio W, Botta C, Pasini M, Bertini F (2001) Synth Met 121:7
Destri S, Pasini M, Giovanella U, Porzio W (2003) Mater Sci Eng C 23:291
Li H, Yang Y, Hou Y, Tang R, Duan T, Chen J, Wang H, Han H, Peng T, Chen X, Li Q, Li Z (2014) ACS Sustain Chem Eng 2:1776
Zhu MR, Ye TL, Ma DG, Yang CL (2011) J Phys Chem C 115:17965
Li Y, Köse ME, Schanze KS (2013) J Phys Chem B 117:9025
Peng ZK, Tao SL, Zhang XH (2008) J Phys Chem C 112:2165
Adachi K, Mita T, Yamate T, Yamazaki S, Takechi H, Watarai H (2010) Langmuir 26:117
Polo F, Rizzo F, Veiga-Gutierrez M, De Cola L, Quici S (2012) 2012. J Am Chem Soc 134:15402
Hao ZS, Li MJ, Lin HX, Gu ZB, Cui YM (2014) Dyes Pigm 109:54
Xiao HB, Yin HY, Zhang XY (2012) Org Lett 14:5282
Price JDW, Tour JM (2003) Tetrahedron 59:3131
Acknowledgments
The authors are grateful for supports from the National Natural Science Foundation of China (Project No. 21272154, 81202402). The authors also thank Drs. H. Deng and M. Shao, The Instrumental Analysis and Research Center of Shanghai University for structural analysis.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gu, ZB., Lin, HX., Cui, YM. et al. Synthesis and characterization of 2′,7′-diarylspiro[cyclopentane-1,9′-fluorene] derivatives. Monatsh Chem 146, 1519–1527 (2015). https://doi.org/10.1007/s00706-015-1432-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-015-1432-9