Abstract
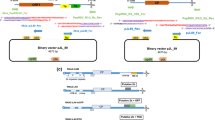
Oat blue dwarf virus (OBDV) is a small, phloem-limited marafivirus that replicates in its leafhopper vector. We have developed complete cDNA clones of OBDV from which infectious transcripts may be derived––the first such clones for any propagatively transmitted plant virus. Prior to clone construction, the reported sequences of the 5′ and 3′ ends were confirmed using 5′ RACE, primer extension, and ligation-anchored PCR. Using vascular puncture of maize seeds with capped transcripts, multiple clones were shown to be infectious at an average rate of 24.3% (range 14–36%). Aster leafhoppers successfully transmitted OBDV to oats and barley after feeding on detached, infected maize leaves. Proteins and RNAs consistent in size with those expected in OBDV infection were detected in young leaves via western and northern blotting, respectively. One construct, pOBDV-2r, was designated as the reference clone. An infectious clone of OBDV will be valuable in examining the interaction of this virus with both its insect and plant hosts.



Similar content being viewed by others
References
Agindotan BO, Ahonsi MO, Domier LL, Gray ME, Bradley CA (2009) A method for the identification of RNA viruses of miscanthus and switchgrass. Phytopathology 99:S2
Al Rwahnih M, Daubert S, Golino D, Rowhani A (2009) Deep sequencing analysis of RNAs from a grapevine showing Syrah decline symptoms reveals a multiple virus infection that includes a novel virus. Virology 387:395–401
Banttari EE, Moore MB (1962) Virus cause of blue dwarf of oats and its transmission to barley and flax. Phytopathology 52:897–902
Carstens EB, Ball LA (2009) Ratification vote on taxonomic proposals to the International Committee on Taxonomy of Viruses (2008). Arch Virol 154:1181–1188
Clark MF, Adams AN (1977) Characteristics of the microplate method of enzyme-linked immunosorbent assay for the detection of plant viruses. J Gen Virol 34:475–483
Ding S-W, Howe J, Keese P, Mackenzie A, Meek D, Osorio-Keese M, Skotnicki M, Srifah P, Torronen M, Gibbs A (1990) The tymobox, a sequence shared by most tymoviruses: its use in molecular studies of tymoviruses. Nucleic Acids Res 18:1181–1187
Dreher TW, Edwards MC, Gibbs AJ, Haenni A-L, Hammond RW, Jupin I, Koenig R, Sabanadzovic S, Abou Ghanem-Sabanadzovic N, Martelli GP (2005) Family Tymoviridae. In: Fauquet CM, Mayo MA, Maniloff J, Desselberger U, Ball LA (eds) Virus Taxonomy, Classification and Nomenclature of Viruses, 8th Report of the ICTV. Elsevier, San Diego, pp 1067–1076
Edwards MC, Weiland JJ (2009) Maize as a new host for Oat blue dwarf virus. Plant Dis 93:972
Edwards MC, Zhang Z, Weiland JJ (1997) Oat blue dwarf marafivirus resembles the tymoviruses in sequence, genome organization, and expression strategy. Virology 232:217–229
Goldbach R, Le Gall O, Wellink J (1991) Alpha-like viruses in plants. Seminars in Virol 2:19–25
Hogenhout SA, Ammar E-D, Whitfield AE, Redinbaugh MG (2008) Insect vector interactions with persistently transmitted viruses. Annu Rev Phytopathol 46:327–359
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lane LC (1986) Propagation and Purification of RNA Plant Viruses. In: Weissbach A, Weissbach H (eds) Methods in Enzymology, vol 118. Academic Press, Orlando, pp 687–696
Louie R (1995) Vascular puncture of maize kernels for the mechanical transmission of maize white line mosaic virus and other viruses of maize. Phytopathology 85:139–143
Maccheroni W, Alegria MC, Greggio CC, Piazza JP, Kamla RF, Zacharias PRA, Bar-Joseph M, Kitajima EW, Assumpção LC, Camarotte G, Cardozo J, Casagrande EC, Ferrari F, Franco SF, Giachetto PF, Girasol A, Jordão H, Silva VHA, Souza LCA, Aguilar-Vildoso CI, Zanca AS, Arruda P, Kitajima JP, Reinach FC, Ferro JA, da Silva ACR (2005) Identification and genomic organization of a new virus (Tymoviridae family) associated with citrus sudden death disease. J Virol 79:3028–3037
Madriz-Ordeñana K, Rojas-Montero R, Lundsgaard T, Ramírez P, Thordal-Christensen H, Collinge DB (2000) Mechanical transmission of maize rayado fino marafivirus (MRFV) to maize and barley by means of the vascular puncture technique. Plant Pathol 49:302–307
Redinbaugh MG, Louie R, Ngwira P, Edema R, Gordon DT, Bisaro DM (2001) Transmission of viral RNA and DNA to maize kernels by vascular puncture inoculation. J Virol Meth 98:135–143
Rozanov MN, Koonin EV, Gorbalenya AE (1992) Conservation of the putative methyltransferase domain: a hallmark of the ‘Sindbis-like’ supergroup of positive-strand RNA viruses. J Gen Virol 73:2129–2134
Sabanadzovic S, Abou Ghanem-Sabanadzovic N (2009) Identification and molecular characterization of a marafivirus in Rubus spp. Arch Virol 154:1729–1735
Sabanadzovic S, Abou Ghanem-Sabanadzovic N, Gorbalenya A (2009) Permutation of the active site of putative RNA-dependent RNA polymerase in a newly identified species of plant alpha-like virus. Virology 394:1–7
Sabanadzovic S, Abou Ghanem-Sabanadzovic N (2009) Association of a new marafivirus with ring spot symptoms in giant ragweed (Ambrosia trifida L) in Mississippi. Phytopathology 99:S112
Sambrook J, Russell DW (2001) Molecular Cloning: a laboratory manual, 3rd edn. Cold Spring Harbor Laboratory Press, Plainview
Skotnicki ML, Ding S-W, Mackenzie AM, Gibbs AJ (1993) Infectious eggplant mosaic tymovirus and ononis yellow mosaic tymovirus from cloned cDNA. Arch Virol 131:47–60
Spetz C, Moe R, Blystad D-R (2008) Symptomless infectious cDNA clone of a Norwegian isolate of Poinsettia mosaic virus. Arch Virol 153:1347–1351
Troutt AB, McHayzer-Williams MG, Pulendran B, Nossal GJV (1992) Ligation-anchored PCR: A simple amplification technique with single-sided specificity. Proc Natl Acad Sci USA 89:9823–9825
Weiland JJ, Dreher TW (1989) Infectious TYMV RNA from cloned cDNA: effects in vitro and in vivo of point substitutions in the initiation codons of two extensively overlapping ORFs. Nucleic Acids Res 17:4675–4687
Yamada M, Izu H, Nitta T, Kurihara K, Sakurai T (1998) High-temperature, nonradioactive primer extension assay for determination of a transcription-initiation site. Biotechniques 25:72–74, 76, 78
Acknowledgments
The authors wish to thank Ryne Hendrickson and Renee McClean for technical assistance. We also thank Dr. Shiaoman Chao for assistance with primer extension analysis and Dr. Margaret Redinbaugh for helpful discussion regarding the vascular puncture technique.
Author information
Authors and Affiliations
Corresponding author
Additional information
Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture.
Rights and permissions
About this article
Cite this article
Edwards, M.C., Weiland, J.J. First infectious clone of the propagatively transmitted Oat blue dwarf virus . Arch Virol 155, 463–470 (2010). https://doi.org/10.1007/s00705-010-0603-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-010-0603-6