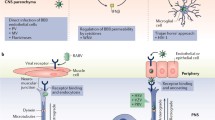
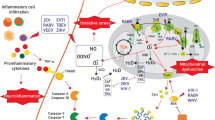
Abstract
During any viral infection of the central nervous system (CNS), the extent and nature of neural cell alterations are dictated by the localization of virus replication and, possibly, persistence. However, one additional source of CNS damage comes from the immune response that develops following CNS viral infection. Indeed, despite of its major role in controlling virus spread in the infected CNS, the immune system is equipped with numerous molecular effectors shared with the nervous system that may greatly alter the homeostasis and function of neural cells. Proinflammatory cytokines and metalloproteases belong to this inflammatory cascade. Besides neurovirulence, the crosstalk engaged between neural and immune cells is a major factor determining the outcome of neuroviral infections.

Similar content being viewed by others
References
Anderson CM, Swanson RA (2000) Astrocyte glutamate transport: review of properties, regulation, physiological functions. Glia 32(1):1–14
Appel SH (2009) CD4+ T cells mediate cytotoxicity in neurodegenerative diseases. J Clin Invest 119(1):13–15
Appel SH, Beers DR, Henkel JS (2010) T cell-microglial dialogue in Parkinson’s disease, amyotrophic lateral sclerosis: are we listening? Trends Immunol 31(1):7–17 (Epub 2009 Oct 31)
Aye MM, Matsuoka E, Moritoyo T, Umehara F, Suehara M, Hokezu Y et al (2000) Histopathological analysis of four autopsy cases of HTLV-I-associated myelopathy/tropical spastic paraparesis: inflammatory changes occur simultaneously in the entire central nervous system. Acta Neuropathol 100:245–252
Batra A, Latour LL, Ruetzler CA, Hallenbeck JM, Spatz M, Warach S, Henning EC (2010) Increased plasma and tissue MMP levels are associated with BCSFB and BBB disruption evident on post-contrast FLAIR after experimental stroke. J Cereb Blood Flow Metab (in press)
Bencsik A, Malcus C, Akaoka H, Giraudon P, Belin MF, Bernard A (1996) Selective induction of cytokines in mouse brain infected with canine distemper virus: structural, cellular and temporal expression. J Neuroimmunol 65:1–9
Bernard A, Fevre-Montange M, Bencsik A, Giraudon P, Wild TF, Confavreux C et al (1993) Brain structures selectively targeted by canine distemper virus in a mouse model infection. J Neuropathol Exp Neurol 52:471–480
Bernard A, Cohen R, Khuth ST, Vedrine B, Verlaeten O, Akaoka H et al (1999) Alteration of the leptin network in late morbid obesity induced in mice by brain infection with canine distemper virus. J Virol 73:7317–7327
Bornstein P, Sage EH (2002) Matricellular proteins: extracellular modulators of cell function. Curr Opin Cell Biol 14:608–616
Bowen JL, Olson JK (2009) Innate immune CD11b+ Gr-1+ cells, suppressor cells, affect the immune response during Theiler’s virus-induced demyelinating disease. J Immunol 183:6971–6980
Carmen J, Rothstein JD, Kerr DA (2009) Tumor necrosis factor-alpha modulates glutamate transport in the CNS and is a critical determinant of outcome from viral encephalomyelitis. Brain Res 1263:143–154
Cartier LM, Cea JG, Vergara C, Araya F, Born P (1997) Clinical and neuropathological study of six patients with spastic paraparesis associated with HTLV-I: an axomyelinic degeneration of the central nervous system. J Neuropathol Exp Neurol 56:403–413
Cauwe B, Martens E, Proost P, Opdenakker G (2009) Multidimensional degradomics identifies systemic autoantigens and intracellular matrix proteins as novel gelatinase B/MMP-9 substrates. Integr Biol (Camb) 1:404–426
Chong YH, Seoh JY, Park HK (1998) Increased activity of matrix metalloproteinase-2 in human glial and neuronal cell lines treated with HIV-1 gp41 peptides. J Mol Neurosci 10:129–141
Conrady CD, Drevets DA, Carr DJ (2009) Herpes simplex type I (HSV-1) infection of the nervous system: is an immune response a good thing? J Neuroimmunol
Danbolt NC (2001) Glutamate uptake. Prog Neurobiol 65:1–105
Edwards DR, Handsley MM, Pennington CJ (2008) The ADAM metalloproteinases. Mol Aspects Med 29:258–289
Elkington PT, O’Kane CM, Friedland JS (2005) The paradox of matrix metalloproteinases in infectious disease. Clin Exp Immunol 142(1):12–20
Fox HS (2008) Virus-host interaction in the simian immunodeficiency virus-infected brain. J Neurovirol 14(4):286–291
Garg RK (2008) Subacute sclerosing panencephalitis. J Neurol 255:1861–1871
Genis P, Jett M, Bernton EW, Boyle T, Gelbard HA, Dzenko K et al (1992) Cytokines and arachidonic metabolites produced during human immunodeficiency virus (HIV)-infected macrophage-astroglia interactions: implications for the neuropathogenesis of HIV disease. J Exp Med 176:1703–1718
Gessain A, Barin F, Vernant JC, Gout O, Maurs L, Calender A et al (1985) Antibodies to human T-lymphotropic virus type-I in patients with tropical spastic paraparesis. Lancet 2:407–410
Giraudon P, Buart S, Bernard A, Thomasset N, Belin MF (1996) Extracellular matrix-remodeling metalloproteinases and infection of the central nervous system with retrovirus human T-lymphotropic virus type I (HTLV-I). Prog Neurobiol 49:169–184
Giraudon P, Szymocha R, Buart S, Bernard A, Cartier L, Belin MF et al (2000) T lymphocytes activated by persistent viral infection differentially modify the expression of metalloproteinases and their endogenous inhibitors, TIMPs, in human astrocytes: relevance to HTLV-I-induced neurological disease. J Immunol 164:2718–2727
Giulian D, Li J, Leara B, Keenen C (1994) Phagocytic microglia release cytokines and cytotoxins that regulate the survival of astrocytes and neurons in culture. Neurochem Int 25:227–233
Gongvatana A, Schweinsburg BC, Taylor MJ, Theilmann RJ, Letendre SL, Alhassoon OM et al (2009) White matter tract injury and cognitive impairment in human immunodeficiency virus-infected individuals. J Neurovirol 15:187–195
Gonzalez-Dunia D, Watanabe M, Syan S, Mallory M, Masliah E, De La Torre JC (2000) Synaptic pathology in Borna disease virus persistent infection. J Virol 74(8):3441–3448
Gottschall PE (1996) Beta-amyloid induction of gelatinase B secretion in cultured microglia: inhibition by dexamethasone and indomethacin. Neuroreport 7:3077–3080
Griffond B, Verlaeten O, Belin MF, Risold PY, Bernard A (2004) Specific alteration of the expression of selected hypothalamic neuropeptides during acute and late mouse brain infection using a morbillivirus: relevance to the late-onset obesity? Brain Res 1022:173–181
Hamzaoui K, Maitre B, Hamzaoui A (2009) Elevated levels of MMP-9 and TIMP-1 in the cerebrospinal fluid of neuro-Behcet’s disease. Clin Exp Rheumatol 27:S52–S57
Harris JE, Nuttall RK, Elkington PT, Green JA, Horncastle DE, Graeber MB et al (2007) Monocyte-astrocyte networks regulate matrix metalloproteinase gene expression and secretion in central nervous system tuberculosis in vitro and in vivo. J Immunol 178:1199–1207
Johnson AJ, Chu CF, Milligan GN (2008) Effector CD4+ T-cell involvement in clearance of infectious herpes simplex virus type 1 from sensory ganglia and spinal cords. J Virol 82:9678–9688
Kanno T, Nagata T, Yamamoto S, Okamura H, Nishizaki T (2004) Interleukin-18 stimulates synaptically released glutamate and enhances postsynaptic AMPA receptor responses in the CA1 region of mouse hippocampal slices. Brain Res 1012:190–193
Kerschensteiner M, Meinl E, Hohlfeld R (2009) Neuro-Immune Crosstalk in CNS Diseases. Results Probl Cell Differ
Khuth ST, Akaoka H, Pagenstecher A, Verlaeten O, Belin MF, Giraudon P et al (2001) Morbillivirus infection of the mouse central nervous system induces region-specific upregulation of MMPs and TIMPs correlated to inflammatory cytokine expression. J Virol 75:8268–8282
Khuth ST, Strazielle N, Giraudon P, Belin MF, Ghersi-Egea JF (2005) Impairment of blood-cerebrospinal fluid barrier properties by retrovirus-activated T lymphocytes: reduction in cerebrospinal fluid-to-blood efflux of prostaglandin E2. J Neurochem 94:1580–1593
Laurenzi MA, Siden A, Persson MA, Norkrans G, Hagberg L, Chiodi F (1990) Cerebrospinal fluid interleukin-6 activity in HIV infection and inflammatory and noninflammatory diseases of the nervous system. Clin Immunol Immunopathol 57:233–241
Lees JR, Golumbek PT, Sim J, Dorsey D, Russell JH (2008) Regional CNS responses to IFN-gamma determine lesion localization patterns during EAE pathogenesis. J Exp Med 205:2633–2642
Leppert D, Ford J, Stabler G, Grygar C, Lienert C, Huber S et al (1998) Matrix metalloproteinase-9 (gelatinase B) is selectively elevated in CSF during relapses and stable phases of multiple sclerosis. Brain 121(Pt 12):2327–2334
Leriche A, Archimbaud JP, Minaire P, Berard E, Bouret J (1976) Pathology of the striated urethral sphincter in spinal cord injury patients: indications and results of external sphincterotomy (100 cases). Paraplegia 13:275–279
Levin MC, Lehky TJ, Flerlage AN, Katz D, Kingma DW, Jaffe ES et al (1997) Immunologic analysis of a spinal cord-biopsy specimen from a patient with human T-cell lymphotropic virus type I-associated neurologic disease. N Engl J Med 336:839–845
Lezin A, Buart S, Smadja D, Akaoka H, Bourdonne O, Perret-Liaudet A et al (2000) Tissue inhibitor of metalloproteinase 3, matrix metalloproteinase 9, and neopterin in the cerebrospinal fluid: preferential presence in HTLV type I-infected neurologic patients versus healthy virus carriers. AIDS Res Hum Retroviruses 16:965–972
Lipton JD, Schafermeyer RW (1995) Central nervous system infections: the usual and the unusual. Emerg Med Clin North Am 13:417–443
Liuzzi GM, Mastroianni CM, Santacroce MP, Fanelli M, D’Agostino C, Vullo V et al (2000) Increased activity of matrix metalloproteinases in the cerebrospinal fluid of patients with HIV-associated neurological diseases. J Neurovirol 6:156–163
Maccioni RB, Rojo LE, Fernández JA, Kuljis RO (2009) The role of neuroimmunomodulation in Alzheimer’s disease. Ann N Y Acad Sci 1153:240–246
Maimone D, Gregory S, Arnason BG, Reder AT (1991) Cytokine levels in the cerebrospinal fluid and serum of patients with multiple sclerosis. J Neuroimmunol 32:67–74
Maragakis NJ, Rothstein JD (2006) Mechanisms of disease: astrocytes in neurodegenerative disease. Nat Clin Pract Neurol 2(12):679–689
Matute C (2006) Oligodendrocyte NMDA receptors: a novel therapeutic target. Trends Mol Med 12:289–292
McCluskey LP, Lampson LA (2000) Local neurochemicals and site-specific immune regulation in the CNS. J Neuropathol Exp Neurol 59:177–187
Merrill JE (1992) Proinflammatory and antiinflammatory cytokines in multiple sclerosis and central nervous system acquired immunodeficiency syndrome. J Immunother 12(3):167–170
Moritoyo T, Reinhart TA, Moritoyo H, Sato E, Izumo S, Osame M et al (1996) Human T-lymphotropic virus type I-associated myelopathy and tax gene expression in CD4+ T lymphocytes. Ann Neurol 40:84–90
Morrison CJ, Butler GS, Rodriguez D, Overall CM (2009) Matrix metalloproteinase proteomics: substrates, targets, and therapy. Curr Opin Cell Biol 21:645–653
Mott JD, Werb Z (2004) Regulation of matrix biology by matrix metalloproteinases. Curr Opin Cell Biol 16:558–564
Nakagawa H, Kitagawa H, Aikawa Y (1987) Tumor necrosis factor stimulates gelatinase and collagenase production by granulation tissue in culture. Biochem Biophys Res Commun 142:791–797
Namba Y, Oka S, Shimada K, Nakano I, Murayama S, Kimura S et al (1991) Post-mortem diagnosis of human T lymphotrophic virus type-1 (HTLV-1) associated myelopathy by detection of HTLV-1 DNA in the spinal cord of a patient with post-transfusional myelopathy. Mol Cell Probes 5:381–384
Nathanson N (2008) The pathogenesis of poliomyelitis: what we don’t know. Adv Virus Res 71:1–50
Neumann H, Schweigreiter R, Yamashita T, Rosenkranz K, Wekerle H, Barde YA (2002) Tumor necrosis factor inhibits neurite outgrowth and branching of hippocampal neurons by a rho-dependent mechanism. J Neurosci 22:854–862
Oldstone MB, Sinha YN, Blount P, Tishon A, Rodriguez M, von Wedel R et al (1982) Virus-induced alterations in homeostasis: alteration in differentiated functions of infected cells in vivo. Science 218:1125–1127
Opdenakker G, Dillen C, Fiten P, Martens E, Van Aelst I, Van den Steen PE et al (2006) Remnant epitopes, autoimmunity and glycosylation. Biochim Biophys Acta 1760:610–615
Park KP, Rosell A, Foerch C, Xing C, Kim WJ, Lee S, Opdenakker G, Furie KL, Lo EH (2009) Plasma and brain matrix metalloproteinase-9 after acute focal cerebral ischemia in rats. Stroke 40(8):2836–2842
Peterson KE, Chesebro B (2006) Influence of proinflammatory cytokines and chemokines on the neuropathogenesis of oncornavirus and immunosuppressive lentivirus infections. Curr Top Microbiol Immunol 303:67–95
Peterson KE, Du M (2009) Innate immunity in the pathogenesis of polytropic retrovirus infection in the central nervous system. Immunol Res 43(1–3):149–159
Prat CM, Schmid S, Farrugia F, Cenac N, Le Masson G, Schwemmle M, Gonzalez-Dunia D (2009) Mutation of the protein kinase C site in borna disease virus phosphoprotein abrogates viral interference with neuronal signaling and restores normal synaptic activity. PLoS Pathog 5(5):e1000425
Ransohoff RM, Perry VH (2009) Microglial physiology: unique stimuli, specialized responses. Annu Rev Immunol 27:119–145
Roberts ES, Huitron-Resendiz S, Taffe MA, Marcondes MC, Flynn CT, Lanigan CM, Hammond JA, Head SR, Henriksen SJ, Fox HS (2006) Host response and dysfunction in the CNS during chronic simian immunodeficiency virus infection. J Neurosci 26(17):4577–4585
Romanic AM, White RF, Arleth AJ, Ohlstein EH, Barone FC (1998) Matrix metalloproteinase expression increases after cerebral focal ischemia in rats: inhibition of matrix metalloproteinase-9 reduces infarct size. Stroke 29(5):1020–1030
Rossi D, Volterra A (2009) Astrocytic dysfunction: insights on the role in neurodegeneration. Brain Res Bull 80(4–5):224–232 (Epub 2009 Jul 22)
Saas P, Boucraut J, Walker PR, Quiquerez AL, Billot M, Desplat-Jego S et al (2000) TWEAK stimulation of astrocytes and the proinflammatory consequences. Glia 32:102–107
Salazar-Mather TP, Hokeness KL (2006) Cytokine and chemokine networks: pathways to antiviral defense. Curr Top Microbiol Immunol 303:29–46
Sato S, Reiner SL, Jensen MA, Roos RP (1997) Central nervous system cytokine mRNA expression following Theiler’s murine encephalomyelitis virus infection. J Neuroimmunol 76:213–223
Scarisbrick IA (2008) The multiple sclerosis degradome: enzymatic cascades in development and progression of central nervous system inflammatory disease. Curr Top Microbiol Immunol 318:133–175
Schwab C, McGeer PL (2008) Inflammatory aspects of Alzheimer disease and other neurodegenerative disorders. J Alzheimers Dis 13(4):359–369
Sellner J, Simon F, Meyding-Lamade U, Leib SL (2006) Herpes-simplex virus encephalitis is characterized by an early MMP-9 increase and collagen type IV degradation. Brain Res 1125(1):155–162
Szretter KJ, Samuel MA, Gilfillan S, Fuchs A, Colonna M, Diamond MS (2009) The immune adaptor molecule SARM modulates tumor necrosis factor alpha production and microglia activation in the brainstem and restricts West Nile Virus pathogenesis. J Virol 83(18):9329–9338 (Epub 2009 Jul 8)
Szymocha R, Akaoka H, Brisson C, Beurton-Marduel P, Chalon A, Bernard A et al (2000a) Astrocytic alterations induced by HTLV type 1-infected T lymphocytes: a role for Tax-1 and tumor necrosis factor alpha. AIDS Res Hum Retroviruses 16:1723–1729
Szymocha R, Akaoka H, Dutuit M, Malcus C, Didier-Bazes M, Belin MF et al (2000b) Human T-cell lymphotropic virus type 1-infected T lymphocytes impair catabolism and uptake of glutamate by astrocytes via Tax-1 and tumor necrosis factor alpha. J Virol 74:6433–6441
Templeton SP, Perlman S (2008) Role of IFN-gamma responsiveness in CD8 T-cell-mediated viral clearance and demyelination in coronavirus-infected mice. J Neuroimmunol 194:18–26
Thomsen AR (2009) Lymphocytic choriomeningitis virus-induced central nervous system disease: a model for studying the role of chemokines in regulating the acute antiviral CD8+ T-cell response in an immune-privileged organ. J Virol 83:20–28
Tishon A, Lewicki H, Andaya A, McGavern D, Martin L, Oldstone MB (2006) CD4 T cell control primary measles virus infection of the CNS: regulation is dependent on combined activity with either CD8 T cells or with B cells: CD4, CD8 or B cells alone are ineffective. Virology 347:234–245
Ulrich R, Baumgärtner W, Gerhauser I, Seeliger F, Haist V, Deschl U, Alldinger S (2006) MMP-12, MMP-3, and TIMP-1 are markedly upregulated in chronic demyelinating theiler murine encephalomyelitis. J Neuropathol Exp Neurol 65(8):783–793
Verlaeten O, Griffond B, Khuth ST, Giraudon P, Akaoka H, Belin MF et al (2001) Down regulation of melanin concentrating hormone in virally induced obesity. Mol Cell Endocrinol 181:207–219
Verma S, Kumar M, Gurjav U, Lum S, Nerurkar VR (2010) Reversal of West Nile virus-induced blood-brain barrier disruption and tight junction proteins degradation by matrix metalloproteinases inhibitor. Virology 397(1):130–138
Vuaillat C, Varrin-Doyer M, Bernard A, Sagardoy I, Cavagna S, Chounlamountri I et al (2008) High CRMP2 expression in peripheral T lymphocytes is associated with recruitment to the brain during virus-induced neuroinflammation. J Neuroimmunol 193:38–51
Wang Z, Pekarskaya O, Bencheikh M, Chao W, Gelbard HA, Ghorpade A et al (2003) Reduced expression of glutamate transporter EAAT2 and impaired glutamate transport in human primary astrocytes exposed to HIV-1 or gp120. Virology 312:60–73
Wang B, Hsu SH, Majumder S, Kutay H, Huang W, Jacob ST et al (2009) TGFbeta-mediated upregulation of hepatic miR-181b promotes hepatocarcinogenesis by targeting TIMP3. Oncogene 21:1–11
Wang S, Quang Le T, Chida J, Cisse Y, Yano M, Kido H (2010) Mechanisms of matrix metalloproteinase-9 upregulation and tissue destruction in various organs in influenza A virus infection. J Med Invest 57(1–2):26–34
Yamanouchi K (1980) Comparative aspects of pathogenicity of measles, canine distemper, and rinderpest viruses. Jpn J Med Sci Biol 33:41–66
Yu N, Billaud JN, Phillips TR (1998) Effects of feline immunodeficiency virus on astrocyte glutamate uptake: implications for lentivirus-induced central nervous system diseases. Proc Natl Acad Sci USA 95:2624–2629
Acknowledgments
We particularly thank Dr. Serge Nataf (U842 Inserm) for help in writing manuscript and helpful discussions. The French Association of Research on multiple sclerosis ARSEP has supported the work cited in this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Giraudon, P., Bernard, A. Inflammation in neuroviral diseases. J Neural Transm 117, 899–906 (2010). https://doi.org/10.1007/s00702-010-0402-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-010-0402-y