Abstract
Objective
Rathke cleft cysts (RCC) are benign lesions of the sellar region that require surgical treatment in case of visual deterioration or progression of the cyst. However, the natural course is often stable and asymptomatic.
We aimed to investigate the characteristics of patients with cyst progression during follow-up (FU) and to compare the natural history of patients with RCC with patients who underwent surgery.
Methods
Patients with an MR morphologic cystic sellar lesion classified as RCC between 04/2001 and 11/2020 were included. Functional outcomes, including ophthalmologic, endocrinologic, and MRI data, were retrospectively analyzed and compared between surgically treated patients, patients on a “watch and wait” strategy (WWS), and patients on a WWS who underwent secondary surgery due to cyst progression.
Results
One hundred forty patients (median age 42.8 years) with RCC on MRI were identified. 52/140 (37.1%) underwent primary surgery. Of 88 patients (62.9%) with initial WWS, 21 (23.9%) underwent surgery for secondary cyst progression. Patients on the WWS had significantly smaller cyst volumes (p = 0.0001) and fewer visual disturbances (p = 0.0004), but a similar rate of hormone deficiencies (p = 0.99) compared with surgically treated patients preoperatively. Postoperatively patients suffered significantly more often from hormone deficiencies than WWS patients (p = 0.001).
Patients who switched to the surgical group were significantly more likely to have preoperative T1 hyperintense signals on MRI (p = 0.0001) and visual disturbances (p = 0.001) than patients with continuous WWS. Postoperatively, these patients suffered more frequently from new hormonal deficiencies (p = 0.001). Endocrine and ophthalmologic outcomes in patients with primary and secondary surgery were comparable.
Multivariate analysis showed that WWS patients were at a higher risk of requiring surgery for cyst progression when perimetric deficits (p = 0.006), hyperprolactinemia (p = 0.003), and corticotropic deficits (p = 0.005) were present.
Conclusion
Surgical treatment of RCC may cause new hormonal deficiencies, which are rare in the natural course. Therefore, the indication for surgery should be carefully evaluated. Hyperprolactinemia and corticotropic deficits were significant indicators for a secondary cyst progression in patients with RCC. However, a significant amount of almost 25% of initially conservatively managed cysts showed deterioration, necessary for surgical intervention.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rathke cleft cysts (RCC) are benign cystic lesions of the sellar region originating from remnants of the craniopharyngeal duct. The incidence is quite high at 22% according to cadaveric studies [23]. However, the rate of symptomatic cysts requiring surgery is relatively low. Only 2–9% of all surgically treated sellar lesions are RCC [19, 28].
Surgery is indicated in space-occupying cysts that lead to worsening visual acuity and visual field loss due to compression of the optic apparatus and may lead to a significant improvement of these symptoms in up to 80% [3, 11, 21]. Surgical complications such as a new hormone deficiency of the anterior or posterior pituitary or leakage of cerebrospinal fluid may occur in about 5–43% of cases [1, 5, 17].
Therefore, incidental asymptomatic findings are often followed by regular MRI controls.
The natural course has previously been described as benign. Nevertheless, cyst progression is reported in about 10%, and/or new hormone deficits can occur over the course [12].
While risk factors for recurrence/progression in surgically treated patients such as preoperative cyst size or squamous metaplasia in the cyst wall and new-onset diabetes insipidus postoperatively have been identified [13, 21, 24], the literature on risk factors for progression in nonsurgical RCC is sparse. Only Kinoshita et al. could show that patients older than 57 years are at an increased risk for cyst progression [12].
Identification of these patients with incidental RCC and increased risk of progression is of paramount importance in determining an optimal follow-up strategy.
In addition, the natural history of patients with RCC undergoing a “watch and wait” (WWS) strategy and the outcomes of surgically treated patients need to be understood in order to weigh the risks and benefits of surgery against the natural clinical course.
Therefore, we aimed to retrospectively analyze and compare the clinical and imaging characteristics of three groups of patients with RCC. Firstly, patients with RCC undergoing WWS. Secondly, patients with progressive RCC on a WWS who underwent surgery for this reason. Thirdly, patients with RCC who underwent primary surgery.
Methods
Patient population
The tumor registry of the Department of Neurosurgery of the Ludwig-Maximilians-University in Munich was searched for all patients with MR morphologic diagnosis of RCC who presented between 04/2001 and 11/2020. Initially, two groups were identified, the first group consisting of patients who underwent surgery and the second group consisting of patients who underwent WWS. Then, a third group was established, consisting of patients who underwent surgery after initial WWS (Fig. 1). For surgically treated patients, MR-imaging and clinical data, including laboratory results of pituitary hormone function and ophthalmologic examination results, were assessed preoperatively within 7 days, 6 weeks, 12 months, 24 months, and later after surgery. For functional outcome analysis, outcome parameters were compared with preoperative findings.
For patients undergoing WWS, the same data were collected at the initial admission and at the last FU visit. Data from patients in the WWS group who underwent surgery for cyst progression during the FU period were analyzed in the same manner as data from patients in the surgical group. These patients are referred to as “secondary surgery” patients in the manuscript.
The 73 patients treated surgically for RCC in our study had the diagnosis of RCC confirmed histopathologically. CK7 and 20 were stained in 100% of cases positively.
In the 63 patients in our study who were managed conservatively, diagnosis of RCC was based on cMRI findings.
The study was approved by the local ethics committee of the Ludwig-Maximilians-University in Munich (reference number 21–0271).
Magnetic resonance imaging (MRI) protocol
Standard MRI (axial T2-weighted sequence with slice thickness of 2 mm, 3-dimensional T1-weighted and constructive interference in steady-state (CISS) sequences with slice thickness of 1 mm) was performed before and after intravenous administration of gadopentetate dimeglumine (Magnevist; Schering Corporation, Kenilworth, NJ) (0.1 mmol/kg body weight) on 1.5- or 3.0-T scanners (Magnetom Symphony, Siemens, Erlangen; Signa HDxt; GE Healthcare, Little Chalfont, UK) with axial, sagittal, and coronal reconstructions. Imaging findings were interpreted as RCC according to the radiological report and the interpretation of the first author, who is a board-certified neurosurgeon. Only patients that were concordantly interpreted as RCC were included. According to previous publications, mainly cystic intra- or suprasellar lesions with or without contrast-enhancing cyst wall with or without the presence of intracystic nodules and/or hemorrhage were considered RCC [4, 26].
Cyst size was calculated by volumetry. Semi-manual segmentation of pre- and postoperative T2/CISS images was performed using the SmartBrush tool of the Elements Brainlab software (Brainlab, Munich, Germany).
An increase in postoperative cyst volume or the initial volume in the conservative group of more than 25% or symptomatic progression of the residual cyst was defined as progression. If the radiologic and neurosurgical evaluation of the postoperative or follow-up MRI images showed a contradictory statement regarding a possible RCC progression or recurrence, a new follow-up MRI image was obtained after 3 months in asymptomatic patients. If postoperative or follow-up imaging did not indicate a residual cyst, any new sellar cystic pathology during FU was considered a cyst recurrence.
Ophthalmological data
Ophthalmic examinations and tests included visual acuity and visual field perimetry measurements preoperatively/at initial diagnosis (ID) and at specific time points (6 weeks, 12 months, 24 months, and later/last FU) after treatment or during the watch and wait period, as assessed by an ophthalmologist. Deficits in visual acuity were classified as severe deficit (visual acuity 0–0.4), mild deficit (visual acuity 0.5–0.9), and no deficit (visual acuity 1.0), visual field deficits as complete hemianopsia, partial anopsia, or no deficit. A postoperative increase in visual acuity of > 0.2 and/or a decreased visual field defect was defined as an improvement, whereas a postoperative decrease in visual acuity of > 0.2 and/or an increased visual field defect was assumed as deterioration. Otherwise, visual performance was classified as unchanged.
Endocrinological data and metabolic outcome
In all patients, basal serum levels of growth hormone, insulin-like growth factor 1, adrenocorticotropic hormone, baseline cortisol, prolactin, luteinizing hormone, follicle-stimulating hormone, testosterone and estradiol, thyrotropin, and thyroid hormone (fT3 and fT4) were measured.
In patients undergoing WWS hormone, levels were assessed at first admission and at last FU, in surgically treated patients preoperatively and at 6 weeks, 12 months, 24 months, and later after surgery. In patients who underwent WWS and underwent surgery due to progression of cysts during the course, hormonal status was assessed at initial admission and thereafter in the same manner as in surgically treated patients. Neurohypophyseal function was measured by serum sodium and osmolality levels, fluid intake and output as well as urine specific gravity level. Endocrinological deficits were classified as anterior or posterior hypopituitarism, which includes complete and incomplete insufficiencies, accordingly to the impacted lobe. In the case of partial or complete involvement of both lobes, insufficiency was defined as panhypopituitarism. If hormone replacement was necessary due to endogenously reduced hormone production, the corresponding hormone axis was considered insufficient. Postoperative improvement was assumed in case of complete recovery of at least one affected hormonal axis and at least unchanged status of the remaining axes. A new complete/partial insufficiency of each hormonal axis was classified as deterioration. Otherwise, the endocrinological function was classified as unchanged.
Treatment protocol
Treatment decisions were invariably made by the interdisciplinary tumor board of the Ludwig-Maximilians-University in Munich. Surgery was usually indicated in patients with visual impairment and a corresponding cyst compressing the optic apparatus. In asymptomatic patients, surgery was offered if the cyst compressed the optic apparatus or if the cyst had increased in size by at least 25%. In these patients, the decision was made on a shared basis according to patient preference and comorbidities. This approach for indicating surgery was used in both the primary and secondary surgery groups. Symptomatic cysts were principally approached via a transnasal, transsphenoidal route. In individual cases, a transcranial approach was chosen based on the patient’s anatomical conditions, especially the cyst configuration and the proximity to the carotid arteries and optic nerves. Conservative treatment of RCC was considered, if patients were asymptomatic and/or the cyst showed no compression of the optic chiasm.
Risk assessment
Perioperative morbidity rates were derived from medical records, and all medical, neurologic, and approach-related adverse events were assessed and classified as either transient or permanent. Functional morbidity was analyzed separately. Depending on the occurrence of morbidity, the disease courses were classified as complicated or unremarkable.
Statistical methods
The reference point of this study was the date of first admission to the hospital. The last FU date was November 2020. Overall and recurrence respectively progression rates were specified by number of patients/total case number. Results were tested by using a two-way analysis of variance (ANOVA), Student’s t-test, and Fisher’s exact test. For risk factor analyses uni- and multivariate tests were conducted. GraphPad PRISM8.0 software was used for statistical analysis (GraphPad, San Diego, CA, USA). Statistical significance was set at p < 0.05.
Results
Patient characteristics and study population
One hundred forty patients with RCC were identified with 52 patients (37.1%) undergoing primary surgery. Of 88 patients (62.9%) with WWS, 21 patients (23.9%) underwent surgery for cyst progression during FU (Fig. 1). Mean age of patients with primary surgery was 43.9 ± 19.1 years, with secondary surgery 46.6 ± 21.2 years, and with WWS 40.8 ± 17.1 years (p = 0.4). Sex was equally distributed in the three groups, with slightly more male patients undergoing surgery (36.5% vs. 28.6% vs. 25.4%; p = 0.4). Mean FU for all surgically treated patients was 55.7 ± 50.0 months. Mean FU for conservatively treated patients was 52.2 ± 38.8 months (p = 0.53). Surgery was performed via transnasal transsphenoidal approach in 70 cases (95.9%) and via transcranial approach in 3 patients (4.1%). These 3 patients had RCC which, in addition to an intrasellar portion, also had a correspondingly large supra- and parasellar cystic portion, so that a transcranial surgical approach was chosen to achieve better access to the carotid arteries and optic nerves. The other cysts with suprasellar and intrasellar parts were operated via using a transsphenoidal approach. In 65 patients (89.0%), fenestrations with an augmented cyst wall resection were conducted. In the remaining patients, only cyst fenestration was performed as previously described by our group [21]. Demographic data are shown in Table 1.
Comparison of imaging, symptoms, and functional outcome
Surgery vs. WWS
Of 73 patients who underwent surgery, 52 patients were surgically treated after the initial diagnosis and 21 patients after the initial WWS. Mean time to surgery was 19.9 ± 11.1 months in those patients undergoing secondary surgery.
Compared to patients with a WWS, surgically treated cysts were significantly larger (p < 0.0001), were more likely to show signs of hemorrhage on MRI (p = 0.038) as well as suprasellar growth (p < 0.0001), and patients were significantly more likely to suffer from visual deterioration (p = 0.0004). In both the surgical group and the WWS group, around 27% of patients reported headaches, which improved significantly over time regardless of the group.
Preoperatively, 33/73 (45.2%) patients suffered from visual field deficits, which improved in 22/33 (66.7%) patients after surgery. Accordingly, surgery led to a significant decrease of cyst size (8.2 cm3 ± 6.5 vs. 1.1 cm3 ± 0.2; p < 0.0001). In the WWS group, only 3/67 (4.5%) patients showed visual field deficits that remained stable in two patients and improved in one patient. The size of the cysts on MRI remained relatively unchanged in the WWS group (3.4 cm3 ± 3.0 at first diagnosis vs. 3.2 cm3 ± 3.0 at last follow-up, p = 0.6).
Preoperatively, there was no difference in hormone deficiencies in both groups. Postoperatively, 14/73 (19.4%) patients developed new hypopituitarism, with 9 patients having partial anterior hypopituitarism predominantly of the corticotropic axis, 1 patient having new-onset diabetes insipidus, and 4 patients having new panhypopituitarism. In contrast, only one patient (1.5%) under WWS developed new partial anterior hypopituitarism (p = 0.02), while in another patient under WWS, the initial diabetes insipidus resolved. A detailed presentation of the clinical symptoms of the surgery and WWS group as well as in the further subdivision as primary and secondary surgery groups are listed in Tables 2 and 3. A further presentation of the clinical data can be found in the supplementary tables: patient characteristics of primary and secondary surgery groups (supplementary Table 1), clinical symptoms of secondary surgery group and WWS (supplementary Table 2), and functional outcome data (supplementary Table 3 and 4).
Comparison of imaging, symptoms, and functional outcome
Secondary surgery vs. WWS
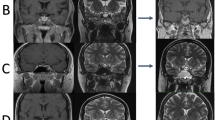
The demographic data of 21 patients who underwent a secondary operation and 67 patients who continued to undergo WWS were comparable. An important marker to determine between the secondary surgery group and WWS was the cyst size, which appeared clearly progressive in the course and extended to the chiasm: cyst size was significantly larger in the secondary surgery group (p < 0.0001), and significantly more patients had T1 hyperintense cysts on MRI (p = 0.0001). After surgery, the cyst volume decreased to a significantly smaller volume than in patients who continued to undergo WWS (0.8 cm3 ± 0.3 vs. 3.2 cm3 ± 3.0, p = 0.0006). An exemplary case is shown in Fig. 2.
Exemplary case of a patient with cyst progression after initial WWS. a, d RCC without optic nerve compression at initial diagnosis on MRI CISS sequence. b, e Contrast-enhanced T1 sequences with evidence of cyst progression 144 months after initial diagnosis. c, f Postoperative contrast-enhanced T1 sequences without signs of residual cyst
Hormonal status and typical symptoms such as headache and diplopia occurred equally frequently in both groups.
On the other hand, visual field defects were more frequent in patients who underwent secondary surgery than in those who did not (p = 0.01). After surgery, visual field deficits and visual impairment improved significantly and were equally common in both groups. However, visual disturbances and partial anopia were still more common even after surgery than in patients who received WWS. While endocrine function was comparable preoperatively, 19.0% suffered from a new partial pituitary insufficiency postoperatively (p = 0.0001).
Comparison of imaging, symptoms, and functional outcome
Primary surgery vs. secondary surgery
Demographic data, imaging findings, and preoperative symptoms did not diverge in 51 patients who underwent primary surgery and 21 patients who underwent secondary surgery. The surgical outcome in terms of improvement in vision and hormonal function was also comparable in both groups with 19% of new anterior or posterior hypopituitarism. There was a trend towards better postoperative improvement in pituitary function in patients who underwent primary surgery, but this was not statistically significant (0/21 vs. 8/52 patients; p = 0.09).
In a total of 99 surgeries, including 15 surgeries for cyst recurrence and 11 revision surgeries for complications, 8/99 (8.1%) CSF leaks occurred after transsphenoidal microsurgical cyst wall fenestration after a mean period of 53.6 ± 30.7 days. Six out of eight patients underwent in total 9 transsphenoidal microscopical revision surgeries, and 2/8 patients were treated with lumbar CSF drainage. Further, two postoperative intrasellar abscesses (2.0%) after transsphenoidal microsurgery occurred after 205 and 324 days, both of which were surgically revised and subsequently treated with intravenous antibiotics. All these complications occurred in the primary surgery group (transsphenoidal approach) while no perioperative complications were found in the secondary surgery group (p = 0.05). All complications are shown in Fig. 1 and listed in Table 4. In 12 (12.1%) cases, transient postoperative electrolyte disorders were diagnosed in the primary surgery group, while only 4 (4.0%) electrolyte disorders were found in the secondary surgery group (p = 0.99).
Indicators for progression during WWS
Univariate analysis revealed that the presence of visual disturbances (p = 0.009), and visual field deficits (p = 0.0009) was associated with a higher risk of cyst progression (Table 5). Suprasellar cyst localization (p = 0.04) as well as corticotropic pituitary deficiency (p = 0.02) and hyperprolactinemia (p = 0.0009) were also independent indicators for progression. Although T1 hyperintense cysts were significantly more common in patients who underwent secondary surgery, cyst hemorrhage could not be identified as a distinct indicator for progression (p = 0.9).
Visual field deficits (p = 0.006) as well as corticotropic pituitary insufficiency (p = 0.005) and hyperprolactinemia (p = 0.003) were also significant indicators for the progression of the cyst in the multivariate analysis.
Discussion
This study investigates clinical and radiological data of 140 patients with RCC that were divided into three groups: (1) patients on a WWS, (2) patients undergoing surgery after initial WWS, and (3) patients undergoing primary surgery.
In patients on a WWS, RCC were either asymptomatic or showed stable or even improved symptoms over time, such as headache, diplopia, pituitary dysfunction, and visual disturbances. While most RCC remained stable during FU, approximately 23.9% of patients with WWS experienced cyst progression and subsequent surgical intervention. These patients typically had visual field defects, corticotropic pituitary deficiency, and hyperprolactinemia.
Surgery proved to be effective in improving visual symptoms by reducing the size of the cyst, and outcomes and complications did not differ between patients who underwent primary surgery and those who underwent surgery after initial WWS. The number of new onset partial pituitary insufficiencies of 19% is remarkable, compared to the stable endocrine course of patients with WWS.
Patients’ characteristics such as age and sex distribution with a male to female ratio of 1:2.3 were characteristic of RCC [2, 15]. The mean FU time of 50 months in both the surgical and WWS groups is rather long compared to previous publications on the dynamics of mainly surgically treated RCC, which ranges from 24 to 72 months [1, 2, 6, 7, 12, 17, 20, 22, 25]. According to the standard of care, the microsurgical approach was transnasal, transsphenoidal in 95.9% of cases. As the clinical outcome and recurrence rates after surgery were comparable to previous studies using microsurgical or endoscopic techniques, we do not consider the microsurgical technique a disadvantage of the study [15].
We found a significant difference in cyst volume between the operated and the WWS group. The difference in preoperative RCC volumes reflects that the indication for surgery is usually made for larger cysts. This again confirms the natural course of RCC with the WWS strategy, so that it must be assumed that the smaller volumes generally only rarely lead to complaints. The difference between postoperative RCC volumes and RCC volumes of the WWS group at the last FU demonstrates the efficacy of surgery in reducing RCC volumes.
Headache and endocrine dysfunction were typical symptoms at initial diagnosis, and the frequency was comparable to previous publications [10, 14]. Interestingly, the number of patients with headache was equal in the surgically treated (26.4%) and WWS (27.9%) groups, and both groups experienced significant improvement in headache over time. At the last FU, only 4.3% and 1.4% respectively had persistent headaches. Previous studies have reported improvement in headache with surgery [9, 16] and suggested that inflammatory processes in the cyst wall are responsible for the headache [16]. In view of the significant improvement in headaches in the natural course, we believe that the indication for surgery in patients with headaches must be restrictive. However, targeted studies focusing in particular on the symptom of headache are warranted.
The endocrine course of patients with WWS was very stable throughout the follow-up period, with only 1/67 (1.5%) patients experiencing new anterior pituitary insufficiency. These results are consistent with the findings of Kinoshita et al. [12] describing the natural history of RCC in a large cohort of 229 patients, with 2 patients (0.9%) developing new pituitary hormone deficiency.
In the surgical group, new hormone deficiencies occurred in around 19% of cases, particularly of the corticotropic (p < 0.008) and thyrotropic (p = 0.057) axis. The rate of new hormone deficiencies after surgery varies widely in the literature between 6% and up to 43% [1, 5, 17, 18, 27]. The aggressiveness of the resection of the cyst wall appears to have a crucial influence on the rate of pituitary dysfunction [1]. Patients in this study were treated with a rather conservative surgical approach with cyst fenestration or augmented cyst wall resection as previously described [21]. The rate of surgical complications was lower in the secondary surgery group, indicating the safety of the surgical procedure after the initial WWS. However, the reason for the lower complication rate remains unclear as there was no difference in demographics, symptoms, cyst configuration, or surgical technique between primary and secondary surgery patients.
The timing of surgery also appears to have no influence on the occurrence of postoperative pituitary insufficiency, as the rates were identical in the primary and secondary surgery groups. An improvement in endocrine function was achieved in 15.5% of patients with primary surgery, which is consistent with previously reported rates [1]. In contrast, no improvement in endocrine function was observed in patients who underwent surgery after initial WWS. The comparison of postoperative pituitary recovery did not differ significantly. However, this aspect should be further investigated in future studies.
Surgery led to a significant improvement of visual field deficits in the primary surgery as well as in the secondary surgery group. Visual acuity also improved after surgery without reaching a statistically significant result (28.8% with mild or severe visual deficit vs. 19.2%; p = 0.2). Visual impairment was rare in patients with WWS, with only about 2% of patients having visual field deficits or blurred vision that did not change over time. In these patients, there was no obvious relation between visual disturbances and RCC.
Mixed intensity of RCC on MRI at T1 and T2 was previously described by Eymann et al. [8]. In our cohort, T1 hyperintensity was typical for RCC treated by primary or secondary surgery and was not observed in patients with continued WWS until the end of FU. Accordingly, univariate and multivariate analysis confirmed T1 hyperintensity as a risk factor for secondary surgery after initial WWS.
In addition, hyperprolactinemia or corticotropic pituitary insufficiency might be an early sign of raised intrasellar pressure and lead to secondary cyst progression.
Interestingly, Kinoshita et al. [12] described 6 of 229 patients who developed new symptoms during FU. Apart from two patients with new visual disturbances, there was one patient with new corticotropic insufficiency and one patient with new hyperprolactinemia. This observation seems to be consistent with our results. However, the results need to be verified in a larger cohort of patients in order to enhance the results of the multivariate analysis.
Conclusion
This study shows the benign natural clinical course of RCC, which is supportive of WWS in RCC without compression of the optic apparatus. However, regular surveillance is warranted as almost 25% of patients with WWS required surgery for cyst progression. T1 hyperintense cysts on MRI, hyperprolactinemia, and hypocortisolism were significant indicators of cyst progression. Delayed surgery after an initial WWS does not lead to a worse outcome than in patients undergoing primary surgery.
Data availability
The original contributions presented in this study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author.
Code availability
Not applicable.
References
Aho CJ, Liu C, Zelman V, Couldwell WT, Weiss MH (2005) Surgical outcomes in 118 patients with Rathke cleft cysts. J Neurosurg 102:189–193. https://doi.org/10.3171/jns.2005.102.2.0189
Barkhoudarian G, Palejwala SK, Ansari S, Eisenberg AA, Huang X, Griffiths CF, Cohan P, Rettinger S, Lavin N, Kelly DF (2019) Rathke’s cleft cysts: a 6-year experience of surgery vs. observation with comparative volumetric analysis. Pituitary 22:362–371. https://doi.org/10.1007/s11102-019-00962-y
Benveniste RJ, King WA, Walsh J, Lee JS, Naidich TP, Post KD (2004) Surgery for Rathke cleft cysts: technical considerations and outcomes. J Neurosurg 101:577–584. https://doi.org/10.3171/jns.2004.101.4.0577
Byun WM, Kim OL, Kim D (2000) MR imaging findings of Rathke’s cleft cysts: significance of intracystic nodules. AJNR Am J Neuroradiol 21:485–488
Castle-Kirszbaum M, Kam J, Wang YY, King J, Fryer K, Goldschlager T (2022) Surgical outcomes and quality of life in Rathke’s cleft cysts undergoing endoscopic transsphenoidal resection: a multicentre study and systematic review of the literature. Pituitary 25:285–295. https://doi.org/10.1007/s11102-021-01197-6
Chong GYC, Tan KCB, Lau EYF, Lai AYT, Man KKY, Chan TM, Leung WKW, Leung JYY (2022) A study on clinical outcomes of Rathke’s cleft cyst in patients managed conservatively. Pituitary 25:258–266. https://doi.org/10.1007/s11102-021-01194-9
Culver SA, Grober Y, Ornan DA, Patrie JT, Oldfield EH, Jane JA Jr, Thorner MO (2015) A case for conservative management: characterizing the natural history of radiographically diagnosed Rathke cleft cysts. J Clin Endocrinol Metab 100:3943–3948. https://doi.org/10.1210/jc.2015-2604
Eymann R, Kiefer M (2018) Rathke-Zyste, Kraniopharyngeom und Kolloidzyste. Radiologe 58:646–652. https://doi.org/10.1007/s00117-018-0398-0
Fukui I, Hayashi Y, Kita D, Sasagawa Y, Oishi M, Tachibana O, Nakada M (2017) Significant improvement in chronic persistent headaches caused by small Rathke cleft cysts after transsphenoidal surgery. World Neurosurg 99:362–368. https://doi.org/10.1016/j.wneu.2016.12.111
Iqbal J, Kanaan I, Al Homsi M (1999) Non-neoplastic cystic lesions of the sellar region presentation, diagnosis and management of eight cases and review of the literature. Acta Neurochir (Wien) 141:389–397; discussion 397–388. https://doi.org/10.1007/s007010050315
Kim JE, Kim JH, Kim OL, Paek SH, Kim DG, Chi JG, Jung HW (2004) Surgical treatment of symptomatic Rathke cleft cysts: clinical features and results with special attention to recurrence. J Neurosurg 100:33–40. https://doi.org/10.3171/jns.2004.100.1.0033
Kinoshita Y, Taguchi A, Yamasaki F, Tominaga A, Arita K, Horie N (2023) Natural course of Rathke’s cleft cysts and risk factors for progression. J Neurosurg 138:1426–1432. https://doi.org/10.3171/2022.7.JNS22716
Kinoshita Y, Tominaga A, Usui S, Arita K, Sakoguchi T, Sugiyama K, Kurisu K (2016) The long-term recurrence of Rathke’s cleft cysts as predicted by histology but not by surgical procedure. J Neurosurg 125:1002–1007. https://doi.org/10.3171/2015.8.JNS151282
Kumar M, Dutta D, Shivaprasad KS, Jain R, Sen A, Biswas D, Mukhopadhyay M, Ghosh S, Mukhopadhyay S, Chowdhury S (2013) Diabetes insipidus as a presenting manifestation of Rathke’s cleft cyst. Indian J Endocrinol Metab 17:S127-129. https://doi.org/10.4103/2230-8210.119529
Mendelson ZS, Husain Q, Elmoursi S, Svider PF, Eloy JA, Liu JK (2014) Rathke’s cleft cyst recurrence after transsphenoidal surgery: a meta-analysis of 1151 cases. J Clin Neurosci 21:378–385. https://doi.org/10.1016/j.jocn.2013.07.008
Nishioka H, Haraoka J, Izawa H, Ikeda Y (2006) Headaches associated with Rathke’s cleft cyst. Headache 46:1580–1586. https://doi.org/10.1111/j.1526-4610.2006.00539.x
Petersson M, Berinder K, Eden Engstrom B, Tsatsaris E, Ekman B, Wahlberg J, Burman P, Borg H, Siesjo P, Dahlqvist P, Akerman AK, Ragnarsson O, Olsson M, Forander P, Bensing S, Hoybye C (2022) Natural history and surgical outcome of Rathke’s cleft cysts-a study from the Swedish Pituitary Registry. Clin Endocrinol (Oxf) 96:54–61. https://doi.org/10.1111/cen.14622
Ratha V, Patil S, Karmarkar VS, Shah NJ, Deopujari CE (2017) Surgical management of Rathke cleft cysts. World Neurosurg 107:276–284. https://doi.org/10.1016/j.wneu.2017.07.164
Saeger W, Ludecke DK, Buchfelder M, Fahlbusch R, Quabbe HJ, Petersenn S (2007) Pathohistological classification of pituitary tumors: 10 years of experience with the German Pituitary Tumor Registry. Eur J Endocrinol 156:203–216. https://doi.org/10.1530/eje.1.02326
Sala E, Moore JM, Amorin A, Carosi G, Martinez H Jr, Harsh GR, Arosio M, Mantovani G, Katznelson L (2018) Natural history of Rathke’s cleft cysts: a retrospective analysis of a two centres experience. Clin Endocrinol (Oxf) 89:178–186. https://doi.org/10.1111/cen.13744
Schmutzer M, Thorsteinsdottir J, Weller J, Rachinger W, Schichor C, Thon N, Ueberschaer M (2023) Nuclear translocation of beta catenin in patients with Rathke cleft cysts-clinical and imaging characteristics and risk of recurrence. Acta Neurochir (Wien). https://doi.org/10.1007/s00701-023-05733-0
Shepard MJ, Elzoghby MA, Kiehna EN, Payne SC, Jane JA (2018) Presentation and outcomes in surgically and conservatively managed pediatric Rathke cleft cysts. J Neurosurg Pediatr 21:308–314. https://doi.org/10.3171/2017.9.PEDS17400
Teramoto A, Hirakawa K, Sanno N, Osamura Y (1994) Incidental pituitary lesions in 1,000 unselected autopsy specimens. Radiology 193:161–164. https://doi.org/10.1148/radiology.193.1.8090885
Trifanescu R, Ansorge O, Wass JA, Grossman AB, Karavitaki N (2012) Rathke’s cleft cysts. Clin Endocrinol (Oxf) 76:151–160. https://doi.org/10.1111/j.1365-2265.2011.04235.x
Truong LUF, Bazin C, Gomis P, Decoudier B, Delemer B, Litre CF (2021) Surgery versus conservative care for Rathke’s cleft cyst. Neurochirurgie 67:104–111. https://doi.org/10.1016/j.neuchi.2020.12.010
Wang S, Nie Q, Wu Z, Zhang J, Wei L (2020) MRI and pathological features of Rathke cleft cysts in the sellar region. Exp Ther Med 19:611–618. https://doi.org/10.3892/etm.2019.8272
Wedemeyer MA, Lin M, Fredrickson VL, Arakelyan A, Bradley D, Donoho DA, Hurth KM, Weiss MH, Carmichael JD, Zada G (2019) Recurrent Rathke’s cleft cysts: incidence and surgical management in a tertiary pituitary center over 2 decades. Oper Neurosurg (Hagerstown) 16:675–684. https://doi.org/10.1093/ons/opy258
Zada G (2011) Rathke cleft cysts: a review of clinical and surgical management. Neurosurg Focus 31:E1. https://doi.org/10.3171/2011.5.FOCUS1183
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Data collection and analysis were performed by Michael Schmutzer-Sondergeld. The first draft of the manuscript was written by Michael Schmutzer-Sondergeld and Moritz Ueberschaer, and all authors commented on the previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. This study was approved by the institutional review board of the Ludwig-Maximilians-University Munich (reference number 21–0271).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Schmutzer-Sondergeld, M., Weller, J., Thorsteinsdottir, J. et al. Long-term outcome of surgically treated and conservatively managed Rathke cleft cysts. Acta Neurochir 166, 159 (2024). https://doi.org/10.1007/s00701-024-06052-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00701-024-06052-8