Abstract
Purpose
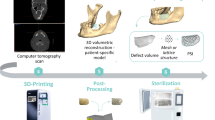
Financial restrictions limit the options for hermetically precise, patient-specific cranial implants (PSCIs) after decompressive hemicraniectomy (DHC) in low-income countries. Use of image segmentation, modeling software, and 3D printers has lowered costs associated with PSCIs. However, requirements of time and technical expertise have prevented widespread utilization. Our objective was to create a fully automated software algorithm that is able to generate a virtual model (.STL) of a negative of an implant using CT imaging following DHC.
Methods
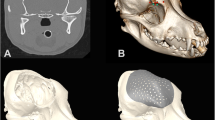
A freeware algorithm (CranialRebuild) was constructed with the following capabilities: (1) after the upload of digital imaging and communications in medicine files, the normal side is analyzed in reference to the side of DHC, (2) Boolean subtraction is used to obtain a virtual image of the desired implant, and (3) a two-piece virtual model (.STL) of the PSCI mold is generated. In four cadaveric specimens, a standard DHC was performed. Post-DHC CT imaging was used to obtain a .STL of the negative of the implant, which was then printed using poly-lactic acid (PLA). Methylmethacrylate cement was used to generate a PSCI from the mold. The PSCIs were implanted into the index specimens; cosmesis was subjectively evaluated using a 5-point Likert scale.
Results
Two specimens were graded as 4/5, indicating that minor post-processing modification was needed for optimal cosmesis. Two specimens were graded as 3/5, indicating that optimal cosmesis could be obtained following moderate post-processing modification.
Conclusions
CranialRebuild can be used to create hermetically precise PSCIs at a fraction of the price of third-party vendors. Validation of this technology has significant implications for the accessibility of customized cranial implants worldwide.



Similar content being viewed by others
Data Availability
All data from the study is reported in Table 2.
References
Ashraf M, Choudhary N, Kamboh UA, Raza MA, Sultan KA, Ghulam N, Hussain SS, Ashraf N (2022) Early experience with patient-specific low-cost 3D-printed polymethylmethacrylate cranioplasty implants in a lower-middle-income-country: technical note and economic analysis. Surg Neurol Int 13:270
Bhaskar IP, Yusheng L, Zheng M, Lee GYF (2011) Autogenous skull flaps stored frozen for more than 6 months: do they remain viable? J Clin Neurosci 18(12):1690–1693
Binhammer A, Jakubowski J, Antonyshyn O, Binhammer P (2020) Comparative cost-effectiveness of cranioplasty implants. Plast Surg (Oakv) 28(1):29–39
ChemAnalyst (2022) Polylactic acid price trend and forecast https://www.chemanalyst.com/Pricing-data/polylactic-acid-1275 Accessed June 2022
ChemAnalyst (2022) Polycarbonate price trend and forecast https://www.chemanalyst.com/Pricing-data/polycarbonate-47 Accessed June 2022
Cheng CH, Lee HC, Chen CC, Cho DY, Lin HL (2014) Cryopreservation versus subcutaneous preservation of autologous bone flaps for cranioplasty: comparison of the surgical site infection and bone resorption rates. Clin Neurol Neurosurg 124:85–89
Eufinger H, Wehmoller M (1998) Individual prefabricated titanium implants in reconstructive craniofacial surgery: clinical and technical aspects of the first 22 cases. Plast Reconstr Surg 102(2):300–308
Eufinger H, Wehmoller M, Machtens E, Heuser L, Harders A, Kruse D (1995) Reconstruction of craniofacial bone defects with individual alloplastic implants based on CAD/CAM-manipulated CT-data. J Craniomaxillofac Surg 23(3):175–181
Hatamleh M (2021) Contemporary review on craniectomy and cranioplasty; part 1. J Craniofac Surg 33(3):838–841
Honeybul S, Ho KM (2012) How "successful" is calvarial reconstruction using frozen autologous bone? Plast Reconstr Surg 130(5):1110–1117
Hutchinson PJ, Kolias AG, Timofeev IS, Corteen EA, Czosnyka M, Timothy J, Anderson I, Bulters DO, Belli A, Eynon A, Wadley J, Mendelow D et al (2016) Trial of decompressive craniectomy for traumatic intracranial hypertension. N Engl J Med 375(12):1119–1130
Morales-Gomez JA, Garcia-Estrada E, Leos-Bortoni JE, Delgado-Brito M, Flores-Huerta LE, De La Cruz-Arriaga AA, Torres-Diaz LJ, Martinez-Ponce de Leon AR (2019) Cranioplasty with a low-cost customized polymethylmethacrylate implant using a desktop 3D printer. J Neurosurg 130(5):1721–1727
Piazza M, Grady MS (2017) Cranioplasty. Neurosurg Clin N Am 28(2):257–265
Schon SN, Skalicky N, Sharma N, Zumofen DW, Thieringer FM (2021) 3D-printer-assisted patient-specific polymethyl methacrylate cranioplasty: a case series of 16 consecutive patients. World Neurosurg 148:e356–e362
Solomon D, Xu A, Cheng JC, Forbes JA (2021) Cost effectiveness in cranioplasty: investigational 3D-printed method for patient-specific cranial implant. Congress Quarterly 22(1):12
Tan ETW, Ling JM, Dinesh SK (2016) The feasibility of producing patient-specific acrylic cranioplasty implants with a low-cost 3D printer. J Neurosurg 124(5):1531–1537
Venugopal V, Ghalsasi O, McConaha M, Xu A, Forbes J, Anand S (2021) Image processing-based method for automatic design of patient-specific cranial implant for additive manufacturing. Proc Manuf 53:375–386
Acknowledgements
We would like to acknowledge and thank Kathleen Smith for her contributions to the preparation of this work.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Alice Xu, Vysakh Venugopal, Manish Raj, Yara Alfawares, Abhijith Matur, Joshua Cheng, Ethan Kosco, Matthew McConaha, Omkar Ghalsasi, Demiah Lockett, Gabriella Bal, Norberto Andaluz, Laura Ngwenya, Sam Anand, and Jonathan Forbes. The first draft of the manuscript was written by Alice Xu, Vysakh Venugopal, Manish Raj, Yara Alfawares, and Abhijith Matur, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the Institutional Review Board of the University of Cincinnati College of Medicine, and all specimens were obtained using the standard informed consent protocol in place at the University of Cincinnati Body Donation Program. The protocols used were approved by the institutional Human Research Protection Program. We certify that the study was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
The authors affirm that human research participants provided informed consent for the publication of the images in Fig. 3.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
(MP4 24446 kb)
(MP4 54244 kb)
(MP4 68711 kb)
(MP4 68803 kb)
(MP4 80182 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, A., Venugopal, V., Aryal, M.R. et al. Toward global availability of low-cost, patient-specific cranial implants: creation and validation of automated CranialRebuild freeware application. Acta Neurochir 165, 2219–2224 (2023). https://doi.org/10.1007/s00701-023-05663-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-023-05663-x