Abstract
Isolated lesions of the corpus callosum are rare and may represent permanent but also transient responses to various pathology termed “reversible splenial lesion syndrome” (RESLES) when in light of relevant clinical presentation. We present the first case of the RESLES after elective surgery for distant arteriovenous malformation (AVM), followed by a slight speech disturbance and MRI verified small, oval, well-circumscribed area of apparent cytotoxic edema in the center of the corpus callosum splenium, which completely resolved within 15 days. Surgery for AVM is followed by the complex adaptation to a new vascular pattern, RESLES might develop, and should be suspected.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Corpus callosum is the largest commissure interconnecting the frontal, parietal, temporal, occipital, insular, and limbic lobes and the basal ganglia of the two hemispheres [14]. Consisting of the 200–250 million densely bundled axonal projections and the major commissural pathway, it forms the largest commissural white matter bundle in the brain [3]. The splenium of the corpus callosum is the most posterior part containing the fibers projecting from the occipital-parietal and temporal cortex, and appears to play an important role in consciousness. The arterial supply is from both anterior and posterior basins, but due to its specific characteristics, it is vulnerable to specific pathological processes [1].
Butterfly lesions involving the corpus callosum and both cerebral hemispheres (usually associated with aggressive tumors, demyelination, and traumatic brain injury) are common, while the isolated lesions of the corpus callosum are rare and may represent permanent or transient responses to injury or other underlying pathology [9]. The former presents a specific lesion usually referred to recently introduced entity — the cytotoxic lesion of the corpus callosum, a secondary lesion associated with various entities including (but not limited to): drug therapy (antiepileptic drugs (AED) in particular), malignancy, infection, subarachnoid hemorrhage, metabolic disorders, and trauma [16]. However, its appearance is not limited to the splenium; therefore, the clinical-radiological condition associated with these lesions has been termed “reversible splenial lesion syndrome” (RESLES) [5]. Several pathophysiological theories have been proposed for the development of transient splenial lesions. Most notable ones related the RESLES to the arginine-vasopressin/antidiuretic hormone induced hyponatremia [17], and transient inflammatory response after sustained hypoosmolality [16].
The clinical presentation varies from the asymptomatic state, but usually involves prodromal fever as the most common symptom, or consciousness disorder, delirium, or seizures [1]. Focal symptoms were rarely reported [6, 10, 13].
Patients in the early postoperative phase are vulnerable to various metabolic and electrolyte disruptions, the RESLES in our patient occurred after the surgery for the parietal lobe arteriovenous malformation (AVM).
Case report
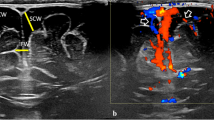
A 15-year-old male underwent elective surgery for the 12-mm lower left parietal lobe AVM. Angiography revealed three feeding arteries arising from the anterior parietal artery (frontal side, parietal side, medial side), and also the draining through the vein of Labbe, to the transverse sinus. No deep drainers were identified (Fig. 1).
Preoperative magnetic resonance imaging (MRI) revealed no other lesions apart from the mentioned AVM. The surgery was carried on under general anesthesia, with motor and somatosensory evoked potentials neuromonitoring. Prior to the surgery, an endovascular procedure was performed for the feeder occlusion with the use of Onyx (Medtronic, Minneapolis, Minnesota, USA) in the same act of general anesthesia.
The surgery went well, and the whole AVM was disconnected from the feeding vessels and the draining vain and extirped. The neuromonitoring and immediate postoperative computerized tomography (CT) were unremarkable and the patient woke up and started to recover as usual in the intensive care unit. Prophylactic AED administration started with 100 mg of lacosamide (days 0–7), followed by 2 mg of perampanel (days 3–16). The doses were increased to 200 mg of lacosamide daily from the day 8, and 4 mg of perampanel from the 17th postoperative day.
Moderate motor aphasia was noted on 1st postoperative day, which deteriorated within a few days. The MRI was performed on the 5th postoperative day, showing the state after the surgery, but also the area of altered signal in the center of the corpus callosum splenium. The small, oval, well-circumscribed lesion demonstrated the features of cytotoxic edema (hypointense on T1, hyperintense on T2, signs of restricted diffusion on diffusion weighted imaging (DWI)) (Fig. 2).
The MRI was performed on the 5th postoperative day, showing the state after the surgery, but also the area of altered signal in the center of the corpus callosum splenium. The small, oval, well-circumscribed lesion demonstrated the features of cytotoxic edema: signs of restricted diffusion: A Hyperintense on DWI b-1000; B Hypointense on DWI b-0 and hyperintense on T2 in C sagittal and D axial planes
The patient was evaluated for clinical signs of the splenial lesion, and the presence of only mild paresthesia in the right hand was noted. A fever of 39.3 °C was present on the 1st day after surgery, and remained in range of 38–39.3 °C for the next 3 days. On the 6th postoperative day, a single focal seizure in the form of hemifacial spasm occurred, however, without any consciousness disturbance or sequelae later on. Since the seizure never reoccurred, no diagnostic evaluation was ordered, and only an additional 1000 mg of levetiracetam was given for 2 days. Motor aphasia persisted for a week, and then dramatically improved two weeks later, and completely disappeared within a month.
Laboratory analyses revealed only slight hyponatremia of minimum 133 milliequivalents per liter (mEq/L), which was present for 5 days, starting from the fifth postoperative day, when the lesions were already clearly demarcated.
Follow-up MRI performed on 7th postoperative day confirmed the previous finding, while the second follow-up MRI, performed on the 15th day, showed complete regression of the lesion (Fig. 3), and it was followed by the symptoms decrease. MRI with tractography was performed 4 weeks after surgery, and revealed no disturbance of the tectum tracts at the site of the lesion, while confirming the splenial lesion vanish (Fig. 4).
Discussion
The case is the first RESLES after the AVM surgery in the English language literature, and the first case to report such complication after the surgery for distant AVM. Usually reported clinical presentation including the fever and seizure was noted. The underlying pathophysiological mechanism of the lesion occurrence was not found, but it may likely be the slight hyponatremia.
Our patient is a 15-year-old male teenager. Previously, these lesions were found to be relatively common among the patients aged 40–94, undergoing surgery for ruptured intracranial aneurysm [18], while in the group of patients with epilepsy, it ranged from 20 to 55 [7]. Starkey et al. reported these lesions in association with various diseases from the age of 0, and in several patients of young age [16]. Focal high signal intensity mimicking these lesions in the splenium of the corpus callosum is a common finding in elderly patients, and may compromise the diagnosis[8].
One paper published in Japanese described a similar lesion after AVM surgery, but with an AVM seated nearby to the splenium, which could be directly associated with the reported splenium lesion [19]. The present case involved the surgery for the cortical AVM in the parietal region, which was completely unrelated to the blood supply of the splenium; however, the blood flow disturbance after the exclusion of the AVM may have played a role in the development of the lesion.
Many of the cases will remain asymptomatic, or at least not worse due to the occurrence of the lesion [18], although a wide variety of symptoms may be observed primarily in forms of: headache, dizziness, blurred vision, vertigo, amnesia, dysmetria, and paresthesia [4, 12]. Ultimately, various levels of the consciousness disturbance of acute or subacute onset may occur with or without seizure or prodromal syndrome (including confusion, visual hallucination, ataxia, signs of interhemispheric disconnection, and dysarthria), and may last for up to 2 weeks [20].
Our patient came for elective surgery, and developed headache, and a seizure, as well as mild paresthesia in his right hand. Due to the complexity of the surgical treatment, which included preoperative endovascular embolization, surgical exclusion and extraction of the nidus, and additional endovascular angiographies performed during the 14 h under general anesthesia, complemented by the sudden blood flow alterations, the symptoms may not be clearly attributed to the RESLES as a sole etiology.
The etiology and the underlying pathophysiology for these lesions are somewhat known, infection, and antiepileptic drugs withdrawal are usual causes [5], and it is relatively common among patients with subarachnoid hemorrhage [18], when the cytotoxic mechanism appears to be the most appropriate, and may be due to the hyponatremia [17], and transient inflammatory response following sustained hypoosmolality [16]. However, the development of motor aphasia may be attributed to the non-convulsive status epilepticus, therefore suggesting the correlation between the RESLES and the seizures, being the symptom or the origin of the splenial lesion in our patient as previously suggested [7]. This remains as a hypothesis, since we failed to confirm the status with relevant diagnostic evaluation.
On the other hand, previously reported a case of the RESLES following the AVM surgery considered demyelination, as a definite rather than reversible process, which we aimed to exclude with the intact tracts visualization [19]. Ischemic origin was revived during the coronavirus disease 2019 pandemic, in a series of splenial lesions associated with the disease; however, these were not followed for the reversibility of the lesions and may be real proper infarctions [15], although it is considered exceptionally rare due to the dual arterial supply, rich collateralization, and emboli resistant posterior pericallosal artery[2, 11].
Conclusion
Arteriovenous malformations surgery is followed by the complex brain adaptation process to a new vascular pattern, therefore demanding extremely careful observation in the postoperative course. The occurrence of the RESLES after elective surgery is extremely rare. Although it is usually a benign entity, it may lead to the development of focal or general consciousness disturbances compromising the initial recovery. Pediatric population is not spared, and postoperative MRI should be performed especially in cases with clinical suspicion of a splenium lesion.
References
Blaauw J, Meiners LC (2020) The splenium of the corpus callosum: embryology, anatomy, function and imaging with pathophysiological hypothesis. Neuroradiology 62:563–585. https://doi.org/10.1007/s00234-019-02357-z
Chrysikopoulos H, Andreou J, Roussakis A, Pappas J (1997) Infarction of the corpus callosum: computed tomography and magnetic resonance imaging. Eur J Radiol 25:2–8. https://doi.org/10.1016/s0720-048x(96)01155-2
Fitsiori A, Nguyen D, Karentzos A, Delavelle J, Vargas MI (2011) The corpus callosum: white matter or terra incognita. Br J Radiol 84:5–18. https://doi.org/10.1259/bjr/21946513
Gao X, Feng Q, Arif S, Liaqat J, Li B, Jiang K (2020) Clinical analysis of reversible splenial lesion syndrome in Chinese adults: a retrospective study of 11 cases. Medicine (Baltimore) 99:e22052. https://doi.org/10.1097/MD.0000000000022052
Garcia-Monco JC, Cortina IE, Ferreira E, Martinez A, Ruiz L, Cabrera A, Beldarrain MG (2011) Reversible splenial lesion syndrome (RESLES): what’s in a name? J Neuroimaging 21:e1-14. https://doi.org/10.1111/j.1552-6569.2008.00279.x
Gellman SR, Ng YT (2018) Transient corpus callosal lesion presenting with alien hand syndrome. Pediatr Neurol 89:66–67. https://doi.org/10.1016/j.pediatrneurol.2018.08.014
Groppel G, Gallmetzer P, Prayer D, Serles W, Baumgartner C (2009) Focal lesions in the splenium of the corpus callosum in patients with epilepsy. Epilepsia 50:1354–1360. https://doi.org/10.1111/j.1528-1167.2008.01800.x
Guo G, Wu R, Ter Brugge K, Mikulis DJ (2006) Focal lesion in splenium of corpus callosum on FLAIR MRI: common findings in aged patients. Neuroradiol J 19:301–305. https://doi.org/10.1177/197140090601900305
Ho ML, Moonis G, Ginat DT, Eisenberg RL (2013) Lesions of the corpus callosum. AJR Am J Roentgenol 200:W1-16. https://doi.org/10.2214/AJR.11.8080
Katsuki M, Kato H, Niizuma H, Nakagawa Y, Tsunoda M (2021) Homonymous hemianopsia due to the infarction in the splenium of the corpus callosum. Cureus 13:e19574. https://doi.org/10.7759/cureus.19574
Li S, Sun X, Bai YM, Qin HM, Wu XM, Zhang X, Jolkkonen J, Boltze J, Wang SP (2015) Infarction of the corpus callosum: a retrospective clinical investigation. PLoS One 10:e0120409. https://doi.org/10.1371/journal.pone.0120409
Lu PL, Hodes JF, Zheng X, Hu XY (2020) Reversible splenial lesion syndrome with some novel causes and clinical manifestations. Intern Med 59:2471–2480. https://doi.org/10.2169/internalmedicine.4516-20
Miyakawa Y, Fuchigami T, Aoki M, Mine Y, Suzuki J, Urakami T, Takahashi S (2018) Agraphia with reversible splenial corpus callosum lesion caused by hypoglycemia. Brain Dev 40:592–595. https://doi.org/10.1016/j.braindev.2018.03.003
Shah A, Jhawar S, Goel A, Goel A (2021) Corpus callosum and its connections: a fiber dissection study. World Neurosurg 151:e1024–e1035. https://doi.org/10.1016/j.wneu.2021.05.047
Sparr SA, Bieri PL (2020) Infarction of the splenium of the corpus callosum in the age of COVID-19: a snapshot in time. Stroke 51:e223–e226. https://doi.org/10.1161/STROKEAHA.120.030434
Starkey J, Kobayashi N, Numaguchi Y, Moritani T (2017) Cytotoxic lesions of the corpus callosum that show restricted diffusion: mechanisms, causes, and manifestations. Radiographics 37:562–576. https://doi.org/10.1148/rg.2017160085
Takanashi J, Shiihara T, Hasegawa T, Takayanagi M, Hara M, Okumura A, Mizuguchi M (2015) Clinically mild encephalitis with a reversible splenial lesion (MERS) after mumps vaccination. J Neurol Sci 349:226–228. https://doi.org/10.1016/j.jns.2014.12.019
Toi H, Yagi K, Matsubara S, Hara K, Uno M (2021) Clinical features of cytotoxic lesions of the corpus callosum associated with aneurysmal subarachnoid hemorrhage. AJNR Am J Neuroradiol 42:1046–1051. https://doi.org/10.3174/ajnr.A7055
Uchida K, Shirakawa M, Sakamoto D, Ando K, Ishikura R, Maeno K, Arita N (2013) A case of reversible splenial lesion after surgical resection of arteriovenous malformation. Brain Nerve 65:212–213
Zhang S, Ma Y, Feng J (2015) Clinicoradiological spectrum of reversible splenial lesion syndrome (RESLES) in adults: a retrospective study of a rare entity. Medicine (Baltimore) 94:e512. https://doi.org/10.1097/MD.0000000000000512
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from the patient’s family.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sato, H., Lepić, M., Suzuki, K. et al. Reversible splenial lesion after surgery for distant arteriovenous malformation: a case report. Acta Neurochir 165, 1603–1607 (2023). https://doi.org/10.1007/s00701-023-05573-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-023-05573-y