Abstract
Introduction
Anterior temporal lobectomy (ATL) is a safe and well-validated procedure in the treatment of temporal lobe epilepsy (TLE), but is a challenging technique to master and still confers a risk of morbidity and mortality due to the complex anatomy of the mesial temporal lobe structures. Automated robotic 3D exoscopes have been developed to address limitations traditionally associated with microscopic visualization, allowing for ergonomic, high-definition 3D visualization with hands-free control of the robot. Given the potential advantages of using such a system for visualization of complex anatomy seen during mesial structure resection in ATL, this group sought to investigate impact on the percentage of hippocampal resection in both exoscope and microscope guided procedures.
Methods
We conducted a retrospective analysis of 20 consecutive patients undergoing standard ATL for treatment of medically refractory TLE at our institution. Using pre-operative and post-operative imaging, the coronal plane cuts in which either the head, body, or tail of the hippocampus appeared were counted. The number of cuts in which the hippocampus appeared were multiplied by slice thickness to estimate hippocampal length.
Results
Mean percentage of hippocampal resection was 61.1 (SD 13.1) and 76.5 (SD 6.5) for microscope and exoscope visualization, respectively (p = 0.0037).
Conclusion
Use of exoscope for mesial resection during ATL has provided good visualization for those in the operating room and the potential for a safe increase in hippocampal resection in our series. Further investigation of its applications should be evaluated to see if it will improve outcomes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Anterior temporal lobectomy (ATL) is a well-validated procedure in the treatment of temporal lobe epilepsy (TLE) cited to provide 60–80% seizure freedom [6, 21, 24]. While the procedure is already considered a safe and efficacious staple in the treatment of TLE, it is a challenging technique to master and still confers a risk of morbidity and mortality that cannot be ignored [1, 4]. At the root of the risk of the procedure lies the complex anatomy of the mesial temporal lobe structures. Most neurosurgical techniques emphasize resection with a posterior extent that reaches the quadrigeminal plate to ensure adequate resection [2, 3, 12, 16]. As such, it is important that innovations and technical advances that may improve safety during mesial temporal lobe resection are explored.
While surgeons have attempted to avoid additional morbidity with techniques such as selective amygdalohippocampectomy [23] or minimally invasive techniques such as laser interstitial thermotherapy (LITT) [8, 13], their efficacy falls short of standard ATL [6, 8, 11, 13, 24]. Parallel to innovating new techniques and approaches, history has demonstrated that advances in operative tools and technology have supported a robust improvement in outcomes, safety, and patient toleration of complex procedures. Examples of such advents include bipolar cautery [15], advances in general anesthesia [17], and improved visualization via the operative microscope [14, 22].
While the operating microscope has improved visualization as well as the ability to combine visual modalities such as augmented reality [9] or fluorescence [20], limitations still exist. Conventional neurosurgical microscopes are large, bulky; require manual movement for changing views; and may be a source of contamination as the eyepieces cannot remain sterile [5].
To address these limitations, automated robotic 3D exoscopes have been developed. These new tools allow for ergonomic, 3D visualization with hands-free control of the robot. Various groups have noted the improved ergonomics for both primary and assisting surgeons, comfort with the hands-free adjustments to the scope, and excellent stereopsis with use of high definition 3D imaging [7, 10, 19]. Given the potential advantages of using such a system for visualization of complex anatomy seen during mesial structure resection in ATL, this group sought to investigate impact on the percentage of hippocampal resection in both exoscope and microscope-guided procedures.
Methods
Patient selection, surgical technique
We conducted a retrospective analysis of 20 consecutive patients undergoing standard ATL for treatment of medically refractory TLE at our institution from April of 2019 to June of 2021. The study protocol was approved by the Institutional Review Board and the participants and any identifiable individuals consented to publication of their image. Of the 20 total cases, 10 of them utilized exoscope as opposed to conventional operative microscope. Two surgeons at a single institution both performed the microscopic and exoscope cases. Both surgeons had been trained to use the exoscope by the same educators and had a similar length of time using the device at approximately 12 months. Patients were included for analysis if they had preoperative and postoperative MRI brain to evaluate length of hippocampus and extent of resection. If patients underwent simultaneous implantation of neuromodulation device, modified approach or extended resection, or lacked any imaging they were excluded. Of note, there was a large gap in cases during the 2020 coronavirus pandemic.
Data was collected on use of exoscope versus microscope, laterality of resection, index temporal resection versus revision, hippocampal length on preoperative imaging, hippocampal residual on postoperative imaging, and presence of significant stroke on post-operative MRI. Significant stroke was defined as infarct on MRI imaging correlating with motor deficit or change from preoperative status. This definition was adopted to avoid erroneous inclusion of “marginal ischemia” and overestimation of morbidity associated with the procedure.
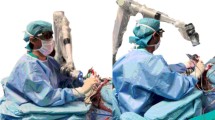
Each patient underwent standard surgical technique for ATL. The surgeon would complete neocortical and amygdala resection under direct visualization with loupes. At this time, the exoscope or microscope would be brought into the surgical field for visualization of the hippocampal resection. Throughout the entirety of the study, the Synaptive Modus V exoscope was used. Figure 1 demonstrates the operating room configuration for use of the exoscope. The exoscope employs a stereotactic 3D visualization microscope mounted on a robotic arm that may be manually moved or adjusted via optical tracking on the device. This allows the surgeon to obtain many optimal working angles using optical tracking on custom instruments while being able to view the ergonomically placed screen during resection with an assistant. Demonstration of the excellent visualization provided by the exoscope with ergonomic benefit during right hippocampal resection is shown in Video 1. After completion of hippocampal resection, standard hemostasis and closure ensued prior to transfer for monitoring postoperatively in the intensive care unit.
Hippocampal resection measurements
Each patient’s preoperative MRI was reviewed. Using axial and sagittal views as references, the coronal plane cuts in which either the head, body, or tail of the hippocampus appeared were counted. For this study, the posterior border of the hippocampus was defined by the plane of the quadrigeminal plate on coronal MRI sequences. The number of cuts in which the hippocampus appeared were multiplied by slice thickness (1 mm on all preoperative imaging) to determine an estimate of preoperative hippocampal length.
On post-operative MRI, a similar technique was used. The number of cuts in which any remaining hippocampus appeared were multiplied by slice thickness (1.5 mm on postoperative imaging) to determine an estimate of residual hippocampal length.
For each patient, percent resection of hippocampus was calculated using [1-(residual length/preoperative length)] × 100 and recorded for analysis.
Statistical analysis
Data were collected and transferred for analysis to SPSS 28.0 for Windows (Chicago, Illinois, USA) and Microsoft Excel 2016. Data on resection percentage are presented as mean and standard deviation. Analysis included a two-sided independent T-test (p-values less than 0.05 were considered statistically significant) to determine if hippocampal resection differed between exoscope and microscope use for visualization. A Shapiro–Wilk test was used to determine the normality of variables; variables with a p-value less than 0.05 were considered not normally distributed.
Results
Mean percentage of hippocampal resection was 61.1 (SD 13.1) and 76.5 (SD 6.5) for microscope and exoscope visualization, respectively. The difference was statistically different (p = 0.0037) in favor of exoscope for more complete hippocampal resection. Mean length of resection of hippocampus was 27.0 mm and 21.4 mm in the exoscope and microscope groups, respectively. These measurements also included revision surgery patients. Comparison of laterality and revision status are shown in Table 1. Only 1 patient out of the population of 20 experienced significant infarct (5%). This was one of the microscope-assisted cases in which the patient suffered mild hemiparesis (4/5) concordant with the site of the ATL. Internal capsule diffusion restriction was visualized on postoperative MRI.
Discussion
Our institution’s experience with exoscope-assisted ATL has been largely positive. Anecdotally, the exoscope improves surgeon ergonomics, while allowing for a co-surgeon to assist. Normally using a microscope makes the latter much more difficult because the co-surgeon is normally positioned at an angle that makes reaching the operative anatomy during mesial resection challenging. With consistent operating room configurations (Fig. 2), set up and positioning of the exoscope has not delayed the case as familiarity at our institution has improved.
From a surgical visualization standpoint, the 3D imaging provides a very clear picture (as seen in Video 1) while providing comfortable positioning. This allows the surgeon to operate comfortably with good visualization as long as needed to obtain a satisfactory resection. The authors hypothesize that this may be useful during hippocampal resection as improved visualization down a more optimal angle allows for more posterior hippocampus to be seen and resected safely without the need for more aggressive retraction. In addition, the authors also feel that the setup and working angles allow for surgical assistants to be more effective in maintaining retraction and good working angles for the primary surgeon to maximize resection. While the authors acknowledge that maximal hippocampal resection may have diminishing returns in the very posterior aspects and may not improve seizure freedom rate [18, 25], traditional teaching of ATL demands that a surgeon be able to complete a hippocampal resection approaching the quadrigeminal plate in order to be considered adequate [2, 3, 16]. In our study, percentage resection using the exoscope was increased compared to when microscopic visualization was utilized, thus maximizing the surgical effect on seizure outcome.
When examining the two groups, it is important to note that this difference outcome was achieved despite similar rates of revision versus index procedures. In addition, several more left sided resections were completed in the exoscope group. Given this trend, one would expect the increased posterior resection to be more difficult to achieve in the exoscope group as the posterior resection corridor may be limited by neocortical structures.
From a safety perspective, no adverse events related to using the exoscope were noted. One mild motor deficit was noted as a sequalae of the microscopic ATL group corresponding to a capsular infarct. While sample size is not large enough to rigorously examine adverse events during surgery, the exoscope group appears to be at least as safe as the conventional microscope.
Limitations
The authors acknowledge the limitations of a small, retrospective study. The relatively small number of patients in the cohort limits definitive comparisons between microscope and exoscope-assisted resection of the hippocampus. In addition, the cases selected for microscope versus exoscope were not randomized and thus may contain inherent bias. However, given the recent development of this new technology, the authors felt the indication, experience, and video footage would serve as a useful reference for other neurosurgeons interested in preliminary evaluation of the technology.
Given the small, largely technical evaluation of the exoscope’s effect on mesial resection, seizure outcomes and other clinical outcomes were not investigated in this study. More rigorous testing of the exoscope may include prospective analysis and further insight into clinical outcomes. In addition, application of exoscope visualization for other procedures or techniques should be investigated to find where this technology is most effective. Certainly, continued evaluation and application in neurosurgery must be practically adoptable to justify the large investment this technology represents for an institution.
Conclusion
Use of exoscope for mesial resection during ATL has provided good visualization for those in the operating room including surgeon, co-surgeon, and residents or students observing the procedure, comfortable ergonomics, and the potential for a safe increase in hippocampal resection in our series. Further investigation of its applications in both epilepsy and other neurosurgical procedures should be evaluated to see if it will improve outcomes.
References
Ahmedov ML, Korkmaz TS, Kemerdere R, Yeni SN, Tanriverdi T (2018) Surgical and neurological complications in temporal lobe epilepsy surgery in modern era. Surg Neurol Int 9:134
Alonso Vanegas MA, Lew SM, Morino M, Sarmento SA (2017) Microsurgical techniques in temporal lobe epilepsy. Epilepsia 58:10–18
Al-Otaibi F, Baeesa SS, Parrent AG, Girvin JP, Steven D (2012) Surgical techniques for the treatment of temporal lobe epilepsy. Epilepsy Res Treat. https://doi.org/10.1155/2012/374848
Brotis AG, Giannis T, Kapsalaki E, Dardiotis E, Fountas KN (2019) Complications after anterior temporal lobectomy for medically intractable epilepsy: a systematic review and meta-analysis. Stereotact Funct Neurosurg 97(2):69–82
Damodaran O, Lee J, Lee G (2013) Microscope in modern spinal surgery: advantages, ergonomics and limitations. ANZ J Surg 83(4):211–214
Engel J, McDermott MP, Wiebe S et al (2012) Early surgical therapy for drug-resistant temporal lobe epilepsy: a randomized trial. JAMA 307(9):922–930
Fiani B, Jarrah R, Griepp DW, Adukuzhiyil J (2021) The role of 3D exoscope systems in neurosurgery: an optical innovation. Cureus. https://doi.org/10.7759/cureus.15878
Gross RE, Stern MA, Willie JT, Fasano RE, Saindane AM, Soares BP, Pedersen NP, Drane DL (2018) Stereotactic laser amygdalohippocampotomy for mesial temporal lobe epilepsy. Ann Neurol 83(3):575–587
Guha D, Alotaibi NM, Nguyen N, Gupta S, McFaul C, Yang VXD (2017) Augmented reality in neurosurgery: a review of current concepts and emerging applications. Can J Neurol Sci 44(3):235–245
Herlan S, Marquardt JS, Hirt B, Tatagiba M, Ebner FH (2019) 3D Exoscope system in neurosurgery—comparison of a standard operating microscope with a new 3D exoscope in the Cadaver Lab. Oper Neurosurg 17(5):518–524
Hu W-H, Zhang C, Zhang K, Meng F-G, Chen N, Zhang J-G (2013) Selective amygdalohippocampectomy versus anterior temporal lobectomy in the management of mesial temporal lobe epilepsy: a meta-analysis of comparative studies: a systematic review. J Neurosurg 119(5):1089–1097
Joo EY, Han HJ, Lee EK et al (2005) Resection extent versus postoperative outcomes of seizure and memory in mesial temporal lobe epilepsy. Seizure 14(8):541–551
Kang JY, Wu C, Tracy J, Lorenzo M, Evans J, Nei M, Skidmore C, Mintzer S, Sharan AD, Sperling MR (2016) Laser interstitial thermal therapy for medically intractable mesial temporal lobe epilepsy. Epilepsia 57(2):325–334
Kriss TC, Kriss VM (1998) History of the operating microscope: from magnifying glass to microneurosurgery. Neurosurgery 42(4):899–907 (discussion 907-908)
Malis LI (2006) Electrosurgery and bipolar technology. Oper Neurosurg 58(suppl_1):ONS-1
Muzumdar D, Patil M, Goel A, Ravat S, Sawant N, Shah U (2016) Mesial temporal lobe epilepsy — an overview of surgical techniques. Int J Surg 36:411–419
Ormond DR, Hadjipanayis CG (2014) The history of neurosurgery and its relation to the development and refinement of the frontotemporal craniotomy. Neurosurg Focus 36(4):E12
Schramm J, Lehmann TN, Zentner J, Mueller CA, Scorzin J, Fimmers R, Meencke HJ, Schulze-Bonhage A, Elger CE (2011) Randomized controlled trial of 2.5-cm versus 3.5-cm mesial temporal resection—part 2: volumetric resection extent and subgroup analyses. Acta Neurochir (Wien) 153(2):221–228
Siller S, Zoellner C, Fuetsch M, Trabold R, Tonn JC, Zausinger S (2020) A high-definition 3D exoscope as an alternative to the operating microscope in spinal microsurgery. J Neurosurg Spine: 1–10. https://doi.org/10.3171/2020.4.SPINE20374
Stummer W, Novotny A, Stepp H, Goetz C, Bise K, Reulen HJ (2000) Fluorescence-guided resection of glioblastoma multiforme by using 5-aminolevulinic acid-induced porphyrins: a prospective study in 52 consecutive patients. J Neurosurg 93(6):1003–1013
Tanriverdi T, Olivier A, Poulin N, Andermann F, Dubeau F (2008) Long-term seizure outcome after mesial temporal lobe epilepsy surgery: corticalamygdalohippocampectomy versus selective amygdalohippocampectomy. J Neurosurg 108(3):517–524
Uluç K, Kujoth GC, Başkaya MK (2009) Operating microscopes: past, present, and future. Neurosurg Focus 27(3):E4
Wendling A-S, Hirsch E, Wisniewski I et al (2013) Selective amygdalohippocampectomy versus standard temporal lobectomy in patients with mesial temporal lobe epilepsy and unilateral hippocampal sclerosis. Epilepsy Res 104(1–2):94–104
Wiebe S, Blume WT, Girvin JP, Eliasziw M, Effectiveness and efficiency of surgery for temporal lobe epilepsy study group (2001) a randomized, controlled trial of surgery for temporal-lobe epilepsy. N Engl J Med 345(5):311–318
Wu C, Jermakowicz WJ, Chakravorti S et al (2019) Effects of surgical targeting in laser interstitial thermal therapy for mesial temporal lobe epilepsy: a multicenter study of 234 patients. Epilepsia 60(6):1171–1183
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
The study protocol was approved by the Institutional Review Board, and the participants and any identifiable individual patients consented to all procedures described.
Informed consent
Patients consented to all described.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Functional Neurosurgery—Epilepsy
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (MP4 69485 KB)
Supplementary file2 (MP4 67466 KB)
Supplementary file3 (MP4 69600 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hines, K., Hughes, L.P., Franco, D. et al. Exoscope improves visualization and extent of hippocampal resection in temporal lobectomy. Acta Neurochir 165, 259–263 (2023). https://doi.org/10.1007/s00701-022-05405-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-022-05405-5