Abstract
Background
Intramedullary spinal cord tumours are rare and account for about 2–4% of primary CNS tumours. Ependymomas and astrocytomas are most frequent. The aim of this study was to evaluate the long-term neurological outcome, quality of life (QoL), survival, need for additional treatment and frequency of neuropathic pain in a patient group treated at a tertiary university hospital.
Method
Retrospective descriptive study of 52 long-term survivors with intramedullary or filum tumours consenting to participate in this study. Fifty-six operations were performed in 48 patients. Clinical and radiological follow-up period was 113 and 117 months, respectively.
Results
Good neurological outcome (ASIA score D or E, modified McCormick grade 1 or 2) was achieved in 88%. We found two negative prognostic factors in regards of severe disability which were large craniocaudal tumour size (p = 0.004) and histologic verified astrocytomas (p = 0.002). SF-36 results showed significantly lower results on all five subdomains concerning physical function, whereas scores for mental health and role emotional showed no significant differences compared to Norwegian norms.
Ten patients including all astrocytoma patients, one primitive neuroectodermal tumour and three recurrent tumours of filum terminale had adjuvant therapy. None of the patients with intramedullary ependymoma had adjuvant therapy.
Neuropathic pain was present in 54% of patients at the last follow-up.
Conclusion
This series shows that good results can be obtained with surgery for intramedullary tumours, even without perioperative neurophysiological monitoring. Multicentre studies are needed for further evaluation of negative and positive prognostic factors to further improve outcome.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Intramedullary spinal cord tumours are rare and account for about 2–4% of primary CNS tumours [25, 27]. Ependymomas and astrocytomas are most frequent. In Manzano’s series, 50% were ependymomas and 12.5% astrocytomas [25], whereas in Raco’s series, 34% were ependymomas and 42% astrocytomas [27]. In adults, as age increases, the incidence of ependymomas increases, and the incidence of astrocytomas decreases [25]. In addition, some rare tumours like primitive neuroectodermal tumours (PNET), hemangioblastoma, cavernomas, seeding of medulloblastomas and glioblastomas may be found. The age-adjusted annual incidence rate of primary intraspinal tumours in Norway in approximately the same time period was 1.29 per 100,000, increasing to 1.48 per 100,000 in the last quarter of the study period. Spinal cord tumours constituted 31% and 41% of the tumours located in the cauda equine or the spinal nerves. Seventeen percent were ependymomas, and 3% were astrocytomas [35]. In our series, a substantial number of ependymomas were located in the filum terminale. Good quality contrast enhanced magnetic resonance imaging (MRI) can detect the tumour at an early stage, even before the tumour has given rise to disabling symptoms like severe ataxia and para/tetra-plegia. Most patients present with a minor sensory or motor deficit, sometimes in addition to neuropathic pain. The treatment for these tumours is surgical resection, but the fear of poor neurological outcome might have had a great impact on the decision-making as to find the best timing for surgery. However, several publications suggest that best outcome is achieved when surgery is performed early, before significant neurological impairment has developed [17, 19, 28, 36].
The aim of this study was to evaluate the long-term neurological outcome, quality of life (QoL), need for additional treatment and frequency of neuropathic pain in a patient group treated at a tertiary university hospital. We also evaluated the frozen section histopathological result, compared to the final diagnosis, and MRI findings during the follow-up period.
The study is approved by the regional ethical committee (2011/2429 IMSHUS).
Method
In this descriptive follow-up study, patients were identified using hospital records searching for the ICD-10 diagnosis D32.1, D33.4, D36.1, C70.1, C72.0 and C72.1 during the years 1991 through 2012. Seventy-eight patients met the requirements of an intramedullary tumour or cyst. Eighteen patients had died before study start. The 60 living patients were invited to participate in the study by mail, and the consent was achieved from 52 patients (87%) (Fig. 1).
Data were collected from patient files and from interviews at the last admission. The patients were examined by one of two neurologists evaluating the ASIA score and McCormick grade at last follow-up and estimating the ASIA score and McCormick grade before treatment based on the patient’s chart. Four patients did not want to come for a clinical follow-up, but consented to the use of the data from their hospital files in the study. Five patients died during the study period before the last study follow-up by a neurologist. Three patients who died due to the spinal tumour were scored according to the best postoperative status. The two patients who died of tumour-unrelated reasons were scored according to the latest clinical follow-up. Fourty-eight patients had a MRI of the spinal column at the last follow-up, and 45 patients filled out questionnaires for QoL (SF-36).
Two experienced pathologists blinded for the primary diagnosis reviewed the histopathological samples. Individual assessments were compared and discrepancies were settled by joint review of the slides in question. The reviewed histopathological diagnoses were compared with the primary diagnosis.
Statistical analyses
Baseline characteristics with differences between subgroups were assessed with Student’s independent samples t-test or chi-squared tests. Statistical significance was p = 0.05. Normal distribution was checked (Q-Q plots), and central tendency presented as means, or medians if skewed. To get a better model-data fit, we dichotomized two variables: thoracic vs other (yes/no) and astrocytoma vs other (yes/no). Additionally, we produced a variable from the combination of ASIA score (A-C) and modified McCormick grade (3–4) to non-ambulant/severe difficulty walking (yes/no). SPSS for Windows version 23.0 was used for all analyses.
Results
Table 1 shows the population characteristics of the 52 study patients.
Four patients with intramedullary tumours having minor or no symptoms were observed clinically for 57–91 (mean 77) months and with MRI scans 68–95 (mean 86) months. These tumours did not grow during the observation period, and the patients had no treatment. Three asymptomatic patients had MRI due to other reasons, which incidentally detected cervical spine ependymomas. Two of these patients had follow-up MRI 6 months later, which showed tumour growth, and both were operated. The third asymptomatic patient had a large tumour in the cranio-cervical junction, and surgery was performed without an observation period.
Initially, all patients except two had estimated ASIA score D or E, and 45 patients had modified McCormick grade 1 or 2a (Tables 2 and 3).
Surgery was performed 0–70 (median 2) months after the diagnosis. Six patients had observation time of more than 8 months before surgery. All patients were operated in the prone position with laminectomy and midline opening of the dura and spinal cord. As judged by the surgeon, 17 of a total of 18 intramedullary ependymomas, one of a total of 6 astrocytomas and 12 of a total of 19 myxopapillary ependymomas of filum terminale were totally resected at the primary procedure. Two patients with astrocytomas and the patient with a cyst in conus medullaris had only biopsy, and one patient with a cystic ependymoma in the cervical spinal cord had biopsy and a very limited resection.
Fifty-six operations were performed in 48 patients. Seven patients had two procedures and one patient had a total of three procedures, two due to residual tumour (one astrocytoma and one myxopapillary ependymoma) and five due to tumour recurrences (three astrocytomas and two large myxopapillary ependymomas of filum terminale). Most of the procedures for pure intramedullary tumours (n = 21, 79%) were performed by the same surgeon (EM), whereas four different surgeons performed the other procedures.
Intraoperative electrophysiological monitoring was not available in this series, except for two patients where somatosensory evoked potentials (SEP) and motor evoked potentials (MEP) were used.
Neurological outcome
Tables 2 and 3 show the neurological outcome compared to the neurological function at first admission assessed by the ASIA score and McCormick grade. A good neurological outcome defined as ASIA score D + E or modified McCormick grade 1–2 was achieved in 88% and 85%, respectively, of intramedullary tumour patients and in all filum tumour patients. Eight patients changed from ASIA E to D due to sensory deficits. Two patients deteriorated from ASIA D + E to C, and only one patient lost more than one level in ASIA score (from C to A). According to McCormick grading, one patient with an intramedullary tumour deteriorated from grade 1 or 2 to grade 3 or 4. The 12 patients with preoperative ASIA grade E were all able to walk independently, whilst 3 out of 19 patients (15.8%) with ASIA grade D needed at least aid like crutches. At the last follow-up, four out of 33 patients (12.1%) with intramedullary tumours were dependent of walking support or wheelchair.
Only one patient (4.2%) with a tumour in the cervical cord needed walking support, whilst one-third of the patients with tumours in the thoracic cord or filum were dependent of walking aids postoperatively. Only one of the 18 patients with intramedullary ependymomas was dependent of postoperative walking support, whilst half of the six patients with astrocytomas were so (p = 0.002).
For patients with filum ependymoma, the neurological function did not change significantly.
The risk factors for a severe outcome for pure intramedullary tumours were evaluated by computing a variable that combined both the ASIA score (A, B and C) in addition to McCormick grading (3 and 4) for better data fit. These patients were evaluated as severely disabled and needed either walking aid or wheel chair. In a univariate analysis, large craniocaudal tumour size and histologic verified astrocytomas were identified as negative prognostic factors, p = 0.004 and p = 0.002, respectively. Other clinically relevant factors as cystic tumours, symptom duration, location, contrast enhancement and time from diagnosis to surgery did not show any negative prognosis (Table 4). However, when including the prognostic factors with a statistical significance in a multivariate regression analysis, we could not find any significant risk factor.
Quality of life
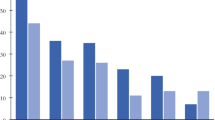
According to the SF-36 questionnaire, the 45 eligible patients scored significantly lower than the Norwegian norms on physical health summary score, but not on mental health summary score. Patients scored significantly lower on all five subdomains concerning physical health (physical function, role physical, bodily pain, general health and vitality), whereas scores for mental health and role emotional showed no significant differences (Fig. 2 and Table 5).
Histopathology
The histopathological diagnosis was assessed according to the WHO classification of tumours of the central nervous system version 2007, and the numbers of tumours are shown in Table 6. Two ependymomas were subependymoma WHO grade 1. Two astrocytomas were graded as WHO grade 2, two grade 2–3, one grade 4 and one uncertain. The other tumours were one ganglioglioneurocytoma, one PNET, one paraganglioma, one hemangioblastoma WHO grade 1 and one cyst in conus.
Two cases were given a new diagnosis based on the re-examination. One ependymoma WHO grade 2 was reviewed as a myxopapillary ependymoma WHO grade 1, and one myxopapillary ependymoma was classified as ependymoma WHO grade 2. For 7 cases, no slides were available for re-examination, and their primary diagnosis was kept.
Frozen section diagnosis was available in 35 cases and was identical to the final histopathological diagnosis in 22 cases (63%). A correct frozen section diagnosis was made in 19 out of 22 ependymomas (86%) and 3 out of 6 astrocytomas (50%). None of the four patients with “other” tumours had correct frozen section diagnosis.
MRI
In the preoperative evaluation of MRI scans, most of the results were reported as “tumour”, but not specified any more. In only ten cases, the tentative radiological diagnosis was consistent with the histopathological diagnosis.
The last MRI scan of the 48 treated patients at an average of 117 months (18–260) postoperatively showed that 14 patients had no visible tumour (Figs. 3 and 4), 30 patients had unchanged postoperative signs (Fig. 5), three patients had residual or remnant tumours (Fig. 6) and one patient had an increasing cyst and atrophy of the spinal cord.
Adjuvant treatment
Ten patients had adjuvant treatment. Five patients had postoperative radiotherapy after the primary surgery, and the other five patients had radiation in combination with chemotherapy for recurrent tumour. These ten patients were six astrocytoma patients, two recurrent and one residual myxopapillary ependymoma of filum terminale and one PNET. None of the patients with intramedullary ependymoma had adjuvant therapy.
Pain
Twenty-eight patients (54%) suffered from neuropathic pain at the last follow-up, 18 patients (54.5%) with tumours in the spinal cord and 10 patients (52.6%) with filum tumours. Unfortunately, the patients’ charts were insufficient regarding preoperative pain.
Survival
Five patients died during the study period, 60–247 months after the surgery. Three patients had astrocytoma grade 2, 3 and 4, respectively, and died due to the tumour. Two of them had more than one surgical procedure, and both had other malignant disease (glioblastoma multiforme and non-Hodgkin’s disease). Two patients with ependymoma of filum terminale, one with grade 1 and one with grade 2, had unknown cause of death.
Discussion
Intramedullary spinal cord tumours are most challenging to treat, with a significant risk of developing neurological deficit independent of treatment strategy. The natural course of ependymomas and even low-grade astrocytomas is usually rather benign with slowly progression of neurological symptoms over years, which traditionally has favoured a wait-and-see strategy or a conservative strategy with biopsy and spinal and dural decompression, and eventually adjuvant radiotherapy and/or chemotherapy [10, 11, 30]. However, during the last two or three decades, advances in surgical resection techniques with ultrasound aspirators, as well as improved electrophysiological monitoring and interpretation of signals, have advocated a more surgical approach to these tumours [8, 10, 11, 30]. Surgery has been shown to be least hazardous when the neurological deficit is minimal or non-existent [9,10,11, 17, 23, 24].
In this series, seven patients with tumours indicating ependymomas had no or minor symptoms. Six patients had serial follow-up MRI scans showing non-growing intramedullary tumours in four of them. They had no treatment, which is in accordance with the recommendations of Harrop et al. [17]. However, Aghakhani et al. [1] recommend that surgery should be considered carefully also to ependymoma patients with no or minor symptoms. Two of our asymptomatic patients had growing tumours, and one patient had a large tumour in the craniocervical junction, and these three patients were operated.
Most attention has been paid to preservation of motor function after intramedullary tumour resection. However, merely opening of the spinal cord to expose the tumour can lead to dorsal column dysfunction (DCD) including numbness and painful dysesthesias, as well as reduced joint sensibility, causing gait disturbances even with intact motor function. This might be the most debilitating postoperative morbidity experienced by these patients. New or increased DCD was seen in 43.6% of Manzano’s patients [25] and 67% in Halvorsen’s series [16].
The most important factor for achieving complete resection is finding of a cleavage plane between the tumour and spinal cord [13, 14]. In contrast to intramedullary astrocytomas, intramedullary ependymomas are usually well defined with a cleavage plane between the tumour and the spinal cord, making a complete resection possible. This is also the goal in ependymoma surgery. However, in a meta-analysis of grade 2 ependymomas, Sun et al. [31] did not find any significant difference in progression free survival and overall survival between age, sex, tumour length, total resection group, subtotal resection group and biopsy and decompression group or chemotherapy. Therefore, surgical resection must be safe in respect of neurological function. It is usually preferable to leave a small area of residual tumour rather than to risk neurological morbidity [30].
The neurological outcome in this series is encouraging and comparable with other series [1, 5, 15]. For intramedullary tumours, some deterioration mostly due to sensory deficits is expected, whilst the neurological outcome for filum tumours most often is equal to the preoperative status.
Not surprisingly, SF-36 showed a significant reduction in physical health subdomains, but not in mental health domains, compared to Norwegian norms. Similar results have been found in patients with spinal cord injury [4]. In spite of neurological deficits, the patients seem to cope with their disability. However, one patient who was quite satisfied with the treatment reported very low scores on physical health domains in spite of minor symptoms and no residual tumour.
Neuropathic pain is a common complaint both pre- and postoperatively in patients with intramedullary tumours. The pain fibres are crossing in the spinal cord and have a close relationship to the central canal where the origin of ependymomas is located. In our series, postoperative neuropathic pain occurred in 54% of the patients as compared to 22% in Klekamp’s series [19]. Surprisingly, postoperative neuropathic pain was equally frequent in patients with intramedullary tumours and myxopapillary tumours of the filum terminale.
In Klekamp’s series, syringomyelia and preoperative presence of neuropathic pain appeared to be the strongest predictors of a postoperative neuropathic pain syndrome, and pain was more common in type A tumours (displacing tumours) compared to type B tumours (infiltrative tumours).
A correct frozen section histopathological answer will facilitate the decision-making for the surgeon, as astrocytomas usually grow infiltrating and do not have a clear surgical plane making complete resection possible. Due to the inherent inaccuracy of attempted frozen section diagnosis at the time of operation, Cooper and Epstein [9] meant that frozen section diagnosis was not helpful. In contrast to this, Hongo et al. [18] found that the frozen section and/or dissection plane helps guide neurosurgeons. The frozen-section diagnosis and final permanent-section diagnosis in their series agreed in 23 (72%) of 32 cases of ependymoma, and in 12 (71%) of 17 cases of astrocytoma. Our results were 86% and 50%, respectively.
Several papers report of the usefulness of electrophysiological monitoring in improving neurological outcome [7, 8, 20,21,22, 26], but none of the studies are RCTs comparing surgery with or without electrophysiologic monitoring. Perioperative electrophysiological monitoring using somatosensory evoked potentials (SEP), motor evoked potentials (MEP) and D-wave recording is now regarded mandatory in surgery for intramedullary spinal cord tumours. Electrophysiological monitoring is a tool to help the surgeon, but cannot replace the skill of the surgeon. In this series, perioperative neurophysiological monitoring was not used, except for two patients. Nevertheless, our results show that good outcome can be achieved also without perioperative monitoring.
Some of the ependymoma patients where tumours were totally resected as judged by the surgeon had follow-up MRI that showed some contrast enhancement that did not change over years (Fig. 5). This might be very small remnants, or contrast enhancement in the tumour bed due to damage of blood-cord barrier. Nevertheless, this finding carries a good prognosis. Even known small remnants seem to be unchanged over many years [1].
Even if it is well accepted that volume matters, our fairly good outcomes are achieved in a small volume centre. However, most of the patients are handled by one experienced senior neurosurgeon, which may reduce the possible negative effect of few procedures.
The role of postoperative radiotherapy for astrocytomas seems well established, but totally resected ependymomas do not need radiotherapy [2, 6, 11, 23, 30, 33]. For recurrent tumours or growing remnants, surgery should be offered [6, 30]. For residual or recurrent high-grade (WHO grades 3 and 4) tumours, radiotherapy is recommended, and chemotherapy is also a treatment option [17]. Myxopapillary ependymoma (MPE) has a generally benign clinical course [2]. However, MPE of the paediatric age group is reported to be more aggressive with tendency for more dissemination (up to 80%) [3, 12].
Using a univariate analysis, we found that large craniocaudal tumour extension and astrocytoma were negative prognostic factors. However, using multivariate regression analysis, these could not be confirmed. This is probably due to the small data sample, as others have found large tumour extension as negative prognostic factor [15, 34].
The weakness of this study is the retrospective design and the small patient population.
Due to the infrequent number of patients with intramedullary spinal cord tumours, to our knowledge, no prospective studies are published, and the numbers of patients in the existing retrospective papers are usually rather small. The need for multi-centre prospective studies is therefore obvious.
Conclusion
This series show that good results are achievable with surgery for intramedullary tumours, even without perioperative neurophysiological monitoring. We recommend that patients with intramedullary tumours without neurological symptoms should undergo a wait-and-see follow-up with serial MRI scans, and surgical resection if tumour growth is detected. For patients with tumour-related symptoms, surgical resection should be recommended, unless there is contra-indications. However, dorsal column dysfunction is likely to occur, and patient’s information in this regard should be emphasised prior to surgery. During recent years, the use of multimodal neurophysiological monitoring has shown encouraging results regarding neurological outcome and is now considered mandatory in surgical treatment of intramedullary tumours.
References
Aghakhani N, David P, Parker F, Lacroix C, Benoudiba F, Tadie M (2008) Intramedullary spinal ependymomas: analysis of a consecutive series of 82 adult cases with particular attention to patients with no preoperative neurological deficit. Neurosurgery 62(6):1279–85. https://doi.org/10.1227/01.neu.0000333299.26566.15 (discussion 1285-6)
Al-Habib A, Al-Radi OO, Shannon P, Al-Ahmadi H, Petrenko Y, Fehlings MG (2011) Myxopapillary ependymoma: correlation of clinical and imaging features with surgical resectability in a series with long-term follow-up. Spinal Cord 49(10):1073–1078. https://doi.org/10.1038/sc.2011.67
Bagley CA, Kothbauer KF, Wilson S, Bookland MJ, Epstein FJ, Jallo GI (2007) Resection of myxopapillary ependymomas in children. J Neurosurg 106(4 Suppl):261–267
Boakye M, Leigh BC, Skelly AC (2012) Quality of life in persons with spinal cord injury: comparisons with other populations. J Neurosurg Spine 17(1Suppl):29–37. https://doi.org/10.3171/2012.6.AOSPINE1252
Boström A, Kanther N-C, Grote A, Boström J (2014) Management and outcome in adult intramedullary spinal cord tumours: a 20-year single institution experience. BMC Res Notes 7:908
Chamberlain MC (2002) Salvage chemotherapy for recurrent spinal cord ependymoma. Cancer 95(5):997–1002
Chang SH, Park YG, Kim DH, Yoon SY (2016) Monitoring of motor and somatosensory evoked potentials during spine surgery: intraoperative changes and postoperative outcomes. Ann Rehabil Med 40(#):470–480
Cheng JS, Ivan ME, Stapleton CJ, Quinones-HinoJosa A, Gupta N, Auguste KI (2014) Intraoperative changes in transcranial motor evoked potentials and somatosensory evoked potentials predicting outcome in children with intramedullary spinal cord tumors. J Neurosurg Pediatr 13(6):591–599
Cooper PR, Epstein F (1985) Radical resection of intramedullary spinal cord tumors in adults. J Neurosurg 63:492–499
Epstein FJ, Farmer J-P, Freed D (1992) Adult intramedullary astrocytomas of the spinal cord. J Neurosurg 77:355–359
Epstein FJ, Farmer J-P, Freed D (1993) Adult intramedullary spinal cord ependymomas: the result of surgery in 38 patients. J Neurosurg 79:204–209
Fassett DR, Pingree J, Kestle JR (2005) The high incidence of tumor dissemination in myxopapillary ependymoma in pediatric patients. Report of five cases and review of the literature. J Neurosurg 102(1 Suppl):59–64
Fehlings MG, Mercier D (2009) Factors predicting the resectability of intramedullary spinal cord tumors and the progressionfree survival following microsurgical treatment. J Neurosurg Spine 11(5):588–9. https://doi.org/10.3171/2009.6.SPINE09360 (discussion 590)
Garcés-Ambrossi GL, McGirt MJ, Mehta VA, Sciubba DM, Witham TF, Bydon A, Wolinksy JP, Jallo GI, Gokaslan ZL (2009) Factors associated with progression-free survival and long-term neurological outcome after resection of intramedullary spinal cord tumors: analysis of 101 consecutive cases. J Neurosurg Spine 11:591–599
Gembruch O, Chihi M, Haarmann M, Parlak A, Oppong MD, Rauschenbach L, Michel A, Jabbarli R, Ahmadipour Y, Sure U, Dammann P, Özkan N (2021) Surgical outcome and prognostic factors in spinal cord ependymoma: a single-center, long-term follow-up study. Ther Adv Neurol Disord 14:1–14
Halvorsen CM, Kolstad F, Hald J, Johannesen TB, Krossnes BK, Langmoen IA, Lied B, Rønning P, Skaar S, Spetalen S, Helseth E (2010) Long-term outcome after resection of intraspinal ependymomas: report of 86 consecutive cases. Neurosurgery 67(6):1622–1631
Harrop JS, Ganju A, Groff M, Bilsky M (2009) Primary intramedullary tumors of the spinal cord. Spine 34(22S):S69–S77
Hongo H, Takai K, Komori T, Taniguchi M (2019) Intramedullary spinal cord ependymoma and astrocytoma: intraoperative frozen-section diagnosis, extent of resection, and outcomes. J Neurosurg Spine 30:133–139
Klekamp J (2013) Treatment of intramedullary tumors: analysis of surgical morbidity and long-term results. J Neurosurg Spine 19:12–26
Kobayashi S, Matsuyama Y, Shinomiya K, Kawabata S, Ando M, Kanchiku T, Saito T, Takahashi M, Ito Z, Muramoto A, Fujiwara Y, Kida K, Yamada K, Wada K, Yamamoto N, Satomi K, Tani T (2014) A new alarm point of transcranial electric stimulation motor evoked potentials for intraoperative spinal cord monitoring: a prospective multicenter study from the Spinal Cord Monitoring Working Group of the Japanese Society for Spine Surgery and Related Research. J Neurosurg Spine 20:102–107
Korn A, Halevi D, Lidar Z, Biron T, Ekstein P, Constantini S (2015) Intraoperative neurophysiological monitoring during resection of intradural extramedullary spinal cord tumors: experience with 100 cases. Acta Neurochir 157:819–830
Kothbauer KF, Deletis V, Epstein FJ (1998) Motor-evoked potential monitoring for intramedullary spinal cord tumor surgery: correlation of clinical and neurophysiological data in a series of 100 consecutive procedures. Neurosurg Focus 4(5):e1. https://doi.org/10.3171/foc.1998.4.5.4
Lee SH, Chung CK, Kim CH, Yoon SH, Hyun SJ, Kim KJ, Kim ES, Eoh W, Kim HJ (2013) Long-term outcomes of surgical resection with or without adjuvant radiation therapy for treatment of spinal ependymoma: a retrospective multicenter study by the Korea Spinal Oncology Research Group. Neuro Oncol 15(7):921–929
Lee SM, Cho YE, Kwon YM (2014) Neurological outcome after surgical treatment of intramedullary spinal cord tumors. Korean J Spine 11(3):121–126
Manzano G, Green BA, Vanni S, Levi AD (2008) Contemporary management of adult intramedullary spinal tumors - pathology and neurological outcomes related to surgical resection. Spinal Cord 46:540–546
Park J-H, Hyun S-J (2015) Intraoperative neurophysiological monitoring in spinal surgery. World J Clin Cases 3(9):765–773
Raco A, Esposito V, Lenzi J, Piccirilli M, Delfini R, Cantore G (2005) Long-term follow-up of intramedullary spinal cord tumors: a series of 202 cases. Neurosurgery 56(5):972–81 (discussion 972-81)
Samii M, Klekamp J (1994) Surgical results of 100 intramedullary tumors in relation to accompanying syringomyelia. Neurosurgery 35:865–873
Samuel N, Tetreault L, Santaguida C, Nater A, Moayeri N, Massicotte EM, Fehlings MG (2016) Clinical and pathological outcomes after resection of intramedullary spinal cord tumors: a single-institution case series. Neurosurg Focus 41(2):E8. https://doi.org/10.3171/2016.5.FOCUS16147
Shrivastava RK, Epstein FJ, Perin NI, Post KD, Jallo GI (2005) Intramedullary spinal cord tumors in patients older than 50 years of age: management and outcome analysis. J Neurosurg Spine 2:249–255
Sun XY, Kong C, Lu SB, Sun SY, Guo MC, Ding JZ (2018) Survival outcomes and prognostic factors of patients with intramedullary grade II ependymomas after surgical treatments. J Clin Neurosci 57:136–142
Sutter M, Eggspuehler A, Muller A, Dvorak J (2007) Multimodal intraoperative monitoring: an overview and proposal of methodology based on 1017 cases. Eur Spine J 16(Suppl 2):S153–S161
Volpp PB, Han K, Kagan AR, Tome M (2007) Outcomes in treatment for intradural spinal cord ependymomas. Int J Radiat Oncol Biol Phys 69(4):1199–1204
Wang X, Gao J, Wang T et al (2018) The long-term outcome after resection of upper cervical spinal cord tumors: report of 51 consecutive cases. Sci Rep 8:14831. https://doi.org/10.1038/s41598-018-33263-8
Weber C, Gulati S, Jakola A, Habiba S, Nygaard ØP, Johannesen TB, Solheim O (2014) Incidence rates and surgery of primary intraspinal tumors in the era of modern neuroimaging. A National Population-Based Study. Spine 39(16):E967–E973. https://doi.org/10.1097/BRS.0000000000000412
Woodworth GF, Chaichana K, McGirt MJ, Sciubba DM, Jallo GI, Gokaslan Z, Wolinsky J-P, Witham TF (2007) Predictors of ambulant function after surgical resection of intramedullary spinal cord tumors. Neurosurgery 61(1):99–106
Funding
Open access funding provided by University of Bergen (incl Haukeland University Hospital)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in this study involving human participants were in accordance with ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Tumor
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Myrseth, E., Habiba, S., Rekand, T. et al. Intramedullary spinal cord and filum tumours—long-term outcome: single institution case series. Acta Neurochir 164, 3047–3056 (2022). https://doi.org/10.1007/s00701-022-05350-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-022-05350-3