Abstract
Background
Return to work (RTW) might be delayed in patients with complicated mild traumatic brain injury (MTBI), i.e., MTBI patients with associated traumatic intracranial lesions. However, the effect of different types of lesions on RTW has not studied before. We investigated whether traumatic intracranial lesions detected by CT and MRI are associated with return to work and post-concussion symptoms in patients with MTBI.
Methods
We prospectively followed up 113 adult patients with MTBI that underwent a brain MRI within 3–17 days after injury. Return to work was assessed with one-day accuracy up to one year after injury. Rivermead Post-Concussion Symptoms Questionnaire (RPQ) and Glasgow Outcome Scale Extended (GOS-E) were conducted one month after injury. A Kaplan–Meier log-rank analysis was performed to analyze the differences in RTW.
Results
Full RTW-% one year after injury was 98%. There were 38 patients with complicated MTBI, who had delayed median RTW compared to uncomplicated MTBI group (17 vs. 6 days), and more post-concussion symptoms (median RPQ 12.0 vs. 6.5). Further, RTW was more delayed in patients with multiple types of traumatic intracranial lesions visible in MRI (31 days, n = 19) and when lesions were detected in the primary CT (31 days, n = 24). There were no significant differences in GOS-E.
Conclusions
The imaging results that were most clearly associated with delayed RTW were positive primary CT and multiple types of lesions in MRI. RTW-% of patients with MTBI was excellent and a single intracranial lesion does not seem to be a predictive factor of disability to work.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Traumatic intracranial lesions in mild traumatic brain injury (MTBI) are common and are associated with use of antithrombotic drugs and advancing age [7, 13, 18, 20]. In MTBI, lesions are seldom life-threatening and patients with complicated MTBI rarely need follow-up scans or neurosurgical intervention [7, 13, 20, 28, 36]. Recovery after complicated MTBI is related to various factors such as lesion type and location, age, level of education and psychiatric profile [35, 39].
Return to work (RTW), an important outcome parameter, might be delayed in patients with complicated MTBI [14]. Still, MTBI is not considered a long-term risk factor for disability to work [2].
In most cases, full recovery is expected, even though a minority has been reported to experience persistent post-concussion symptoms [6, 12, 17, 33].
Types and locations of intracranial lesions in MTBI have been well characterized [13], but to our knowledge, the influence of lesions on RTW in MTBI has not been studied before. In addition, the influence of primary CT finding is of interest, since CT is primary imaging modality in emergency units. Nevertheless, up to one-third of patients with MTBI and a normal head CT have an abnormal MRI [41]. Thus, accounting for traumatic MRI findings is necessary to fully understand the relationship between MTBI and RTW.
The main objective of this study was to compare RTW in complicated and uncomplicated MTBI, and to assess the association between traumatic intracranial lesions and RTW. We hypothesized that overall RTW would be similar between the groups but that RTW would be delayed in MTBI patients with traumatic CT and/or MRI findings (i.e., complicated MTBI) in comparison with MTBI patients with negative CT and/or MRI findings (i.e., uncomplicated MTBI).
Methods
Patients
Our prospective cohort included 131 consecutive patients with MTBI (age 18–68 years) from the Traumatic Brain Injury Outpatient Clinic of Helsinki University Hospital. Patients from the catchment area of the Helsinki University Hospital, home to 2 million inhabitants, were prospectively recruited from 2015 to 2018 and evaluated in the clinic one month after injury.
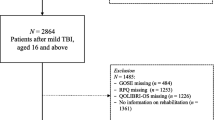
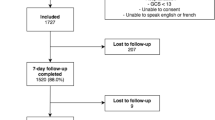
In this study, patients with alcohol or drug dependence were excluded. Dependence was defined according to the Diagnostic and Statistical Manual of Mental Disorders, 4th edition (DSM-IV) criteria. In addition, visual or auditory disability, previously diagnosed schizophrenia or schizoaffective disorder, contraindications for MRI imaging and not being a native Finnish speaker were also exclusion criteria. For this study, we also excluded patients who were not employed during the time of injury, and patients who underwent MRI imaging later than 17 days after injury. This resulted in a total of 113 patients diagnosed with MTBI.
All included patients gave their written consent. This study was additionally approved by the ethics committee of Helsinki University Hospital (code 105/13/03/01/2014).
MTBI classification
We used the World Health Organization (WHO) definition of MTBI [3]. These criteria include one or more of the following: 1) confusion or disorientation, loss of consciousness for 30 min or less, post-traumatic amnesia for less than 24 h and/or other transient neurological abnormalities such as focal signs, seizure and intracranial lesion not requiring surgery; and 2) Glasgow Coma Scale score of 13–15 after 30 min post-injury or later upon presentation for healthcare. These manifestations of MTBI could not be due to drugs, alcohol, medications, caused by other injuries or treatment for other injuries (e.g., systemic injuries, facial injuries or intubation), caused by other problems (e.g., psychological trauma, language barrier or coexisting medical conditions), or caused by penetrating craniocerebral injury. MTBI was defined as complicated when a traumatic intracranial abnormality was present in CT or MRI imaging. Isolated skull fractures were not considered as complicated MTBI.
Initial evaluation
Patients with MTBI were initially evaluated in the Helsinki University Hospital or city hospital emergency units. Initial GCS, loss of consciousness (LOC) and presence and length of post-traumatic amnesia (PTA) along with relevant clinical findings were documented by physicians in the emergency unit, who also determined the initial sick leave length. At this point, patients with more severe TBI were excluded. Primary CT was obtained from 106 (94%) of the patients. Acute symptoms, such as headache and vomiting, were screened and other injuries were determined by Abbreviated Injury Scale and Injury Severity Score, retrospectively from hospital records [5].
At one month after injury, patients were evaluated by a board certified neurologist in the Traumatic Brain Injury Outpatient Clinic of Helsinki University Hospital, using the Neurological Outcome Scale for TBI (NOS-TBI) [37]. Previous and current illnesses and medications were thoroughly assessed using hospital records and by conducting a structured interview. Successful RTW was verified and time to RTW was documented. When deemed requisite, sick leave was extended and additional appointments were arranged in addition to the research protocol.
Additionally, at one month after injury, the presence of post-traumatic symptoms was assessed using the Rivermead Post-Concussion Symptoms Questionnaire (RPQ), evaluating the frequency and severity of 16 post-concussion symptoms, including various physical, emotional and cognitive symptoms [19]. Overall recovery assessed using Glasgow Outcome Scale Extended (GOS-E) [16]. GOS-E is an assessment tool for functional recovery, a scale ranging from 1 (dead) to 8 (good recovery). A score of ≥ 6 is considered as a favorable outcome for patients with TBI [32]. Patients, who have fully returned to work, are generally considered GOS-E = 8, though some patients may work with minor post-concussion symptoms [31].
Return to work evaluation
Return to work was assessed retrospectively with one-day accuracy using clinic records, and patients’ successful RTW was later verified by a structured telephone interview at one year after injury, by the study author (AH). In Finland, the sick leaves are registered electronically thus considered reliable [34]. Thus, RTW could be documented with one-day accuracy. Full RTW was determined days from injury to the first day back to full-time work, with no further significant sick leave in the follow-up period. Patients were not considered to have fully returned to work until their possible partial labor period was over. Patients who were full-time (n = 13) or part-time (n = 6) students were also included in this study and their return to studies was comparable and included in RTW parameters.
RTW was assessed as a continuous variable (time to RTW) with one-day accuracy and dichotomously (RTW-%) with predetermined cutoff points: 14 days, 30 days, 90 days and one year after injury.
Brain imaging
All patients underwent brain MRI imaging within 3–17 days (mean 9.6, SD 3.2) after MTBI and all MRI scans were evaluated by a board certified neuroradiologist. Lesions were assessed systematically by using common data elements (CDE) for TBI neuroimaging [11].
All imaging was performed with 3 T Siemens Magnetom Verio (Siemens, Erlangen) scanner with a 32-channel head coil. The imaging protocol consisted of fast localizer, T1 sagittal localizer, axial FLAIR, coronal T2, 3D T2 SPACE, 3D T1 MPRAGE and 3D gradient-echo susceptibility weighted imaging (SWI) sequence.
Investigated traumatic intracranial lesions included extra-axial lesions (subdural hemorrhages [SDH], subarachnoid hemorrhages [SAH] and epidural hemorrhages [EDH]) and intraparenchymal lesions (cerebral contusions, traumatic microbleeds [TMBs] and other intracerebral hemorrhages [ICH]). Traumatic microbleeds were defined as a single or several small hemorrhagic lesion(s) in the white matter or grey–white interface, detected with SWI sequence. No non-hemorrhagic diffuse axonal injury lesions were detected. The presence of each of the lesions was assessed. The location or the number of lesions were not specified in this study.
Differentiation between the patients with uncomplicated MTBI (n = 75) and those with complicated MTBI (n = 38) was based on the findings in the conventional 3 T MRI, including SWI sequences. The latter group was furthermore divided into those who had only one type of traumatic intracranial lesion (n = 19), and those who had more than one type of traumatic intracranial lesions (n = 19). Additionally, we analyzed following subgroups separately: patients who did not undergo primary CT imaging (n = 7), patients with no traumatic intracranial lesions visible in the primary CT (n = 82) and patients with one or more traumatic intracranial lesions visible in the primary CT (n = 24). Figure 1 shows a flowchart on the relation between findings in CT and MRI.
Statistical analysis
Skewed distributions were reported as medians with interquartile range (IQR) and normally distributed values in mean and standard deviation (SD). Skewed data were compared between groups using a nonparametric Mann–Whitney U test. With multiple groups, we used Kruskal–Wallis H test. Categorical variables were compared using a two-sided χ2 test (Pearson Chi-square test). Spearman correlation test was used to compare correlation between two continuous variables. A Kaplan–Meier log-rank analysis was performed to investigate the time differences in RTW between groups. We considered p values < 0.05 as statistically significant.
IBM SPSS Statistics 25 (IBM Corp., Armonk, NY, USA) was used to perform the analyses.
Results
There were 38 patients with complicated MTBI and 24 patients had a traumatic lesion visible in the primary head CT scan. Nineteen patients had more than one type of lesion in MRI, for instance, traumatic microbleeds and SDH or SAH (Fig. 1). The most common mechanism of injury was ground level fall (28%), followed by bicycle accident (26%) and fall from heights (20%).
Median RTW was 9 days (IQR 4.0–30.0). Fifty-seven percent of the patients had full RTW before MRI. RTW-% was 92% at three months and 98% one year after injury. One patient was still working partially and only one patient had not returned to work at all at one year after injury. For all patients, RPQ showed a median of 8.0 points (IQR 3.0–15.0), while in GOS-E, 56% of patients had a good (GOS-E = 8) functional recovery.
There were 14 patients who had traumatic intracranial lesions detected only in the subacute MRI and not in CT (CT-/MRI + group). Of them, 11 had traumatic microbleeds, one had a cerebral contusion, one had SDH, and one patient had both SDH and SAH. Their median RTW was 9 days (IQR 5.0–15.0), median RPQ 10.0 points (IQR 3.5–15.0) and 62% of patients had a good (GOS-E = 8) functional recovery, at one month after injury.
Complicated vs. uncomplicated MTBI
Clinical characteristics of patients with uncomplicated vs. complicated MTBI are shown in Table 1. There was a significant difference between the groups in length of hospitalization (p = 0.012) and frequency and severity of extracranial injuries (p = 0.024).
Patients with complicated MTBI had significantly delayed RTW (n = 38, median 17 days, IQR 9.5–50.5) compared with patients with uncomplicated MTBI (n = 75, median 6 days, IQR 3.0–16.0; p < 0.001), as illustrated in Table 1 and Fig. 2. The most noticeable difference in RTW-% was at 14 days after injury (37% vs. 75%; p < 0.001). There were no significant differences at three months after injury, but RTW-% was lower in the complicated group one year after injury (95% vs. 100%, p = 0.045).
Patients with complicated MTBI (Table 1) suffered significantly more frequently from post-concussion symptoms (median RPQ 12.0, IQR 5.0–15.0) than patients with uncomplicated MTBI (median RPQ 6.5, IQR 2.0–13.3; p = 0.025). There was no significant difference in GOS-E results; 50% vs. 59% had a good functional recovery (GOS-E = 8).
Positive vs. negative primary CT
As illustrated in Fig. 3, MTBI patients with CT positive traumatic intracranial lesion(s) had delayed RTW (n = 24, median 31 days, IQR 15.3–56.5) compared to patients with negative primary CT (n = 82, median 7 days, IQR 4.0–16.0) and those who did not undergo CT imaging (n = 7, median 3 days, IQR 2.0–7.0; p < 0.001).
As shown in Table 2, MTBI patients with CT positive traumatic intracranial lesions had post-concussion symptoms more frequently (median RPQ 13.5, IQR 9.3–15.3) than patients with negative primary CT (median RPQ 7.0, IQR 2.0–14.5) and those who did not undergo CT imaging (median RPQ 4.0, IQR 0–10.0; p = 0.017). There were no significant differences in GOS-E (p = 0.186).
Influence of number and type of lesions
Patients with more than one type of traumatic intracranial lesion had delayed RTW (n = 19, median 31 days), compared to those with only one type of lesion (n = 19, median 10 days) and patients with uncomplicated MTBI (n = 75, median 6 days; p < 0.001), as illustrated in Fig. 4. There were no statistically significant differences in RPQ or GOS-E (Table 3).
Regarding specific lesions (Table 4), median RTW was delayed among patients with SDH (median 37.5 days, p < 0.001), SAH (median 33.5 days, p = 0.001) and cerebral contusions (median 50.0 days; p < 0.001), compared to patients without that specific lesion.
Cerebral contusions showed a borderline association with more frequent post-concussion symptoms (median RPQ 14.5, IQR 8.3–21.0; p = 0.049). Regarding other lesions, there were no significant differences in RPQ or GOS-E.
One year after injury, two patients with three types of traumatic intracranial lesions, had not fully returned to work (Tables 1, 2, 3 and 4). RTW-% at one year was significantly lower in the following groups: those with CT positive lesions (92%, p = 0.023), those with more than one type of intracranial lesion (90%, p = 0.006) and those with SDH (88%, p < 0.001), cerebral contusions (82%, p < 0.001) or other intracranial hemorrhages (75%, p < 0.001), compared to other patients with MTBI.
There was a significant but weak positive correlation between RTW and RPQ (0.256, p = 0.011). Fully recovered patients (GOS-E = 8) returned to work faster (median RTW 6.0 days, IQR 3.0–11.8) than patients with impaired recovery (GOS-E < 8) (median RTW 17.5 days, IQR 5.3–58.8, p = 0.001).
Discussion
In this well-characterized group of patients with MTBI, the imaging results that were most clearly associated with delayed RTW were positive primary CT and finding of more than one type of lesion in MRI. The difference is most noticeable in the first weeks after injury but tend to even out after three months, though a minority of patients have impaired recovery after this. Delayed RTW may result from prolonged symptoms and slower recovery, but also from primary care physicians’ tendency to give longer sick leave when intracranial lesion is visible in the primary CT scan. Nevertheless, RTW-% of patients with MTBI was excellent and a single intracranial lesion does not seem to be a predictive factor of disability to work.
According to meta-analysis by Bloom and colleagues, RTW after MTBI generally varies between 13 and 93 days, and approximately 89% of patients return to work in one year after injury [1]. Our patients returned to work reasonably fast, with a median of 9 days and 98% were fully returned to work by the end of the year. Similar results were found by Wäljas and colleagues, with 97% RTW rate one year after injury [34].
Delayed RTW in patients with complicated MTBI has been previously documented [14, 34], but there is also documentation on that the prognosis does not differ much between uncomplicated and complicated MTBI [2, 6, 17]. Iverson and colleagues found that MTBI patients with intracranial lesions had significantly delayed RTW (36 vs. 6 days) compared to patients with uncomplicated MTBI [14]. Our results (median RTW 17 vs. 6 days) are in line with these findings.
In our study, CT positive trauma lesions seemed to be strongly associated with delayed RTW (median time to RTW 31 days, n = 24). Patients with positive CT findings had more post-concussion symptoms at one month after injury, likely explaining the delayed RTW in this group. Conversely, the 14 patients who had traumatic intracranial lesions detected only in the subacute MRI and not in CT, had a shorter RTW than those with acute positive CT findings, similar to patients with uncomplicated MTBI. Since the majority of the patients in our cohort returned to work before MRI (57%) and most of the patients had successful RTW quickly after imaging, it is unlikely that MRI results had a major role in RTW. In our opinion, subacute MRI might provide additional information when targeted to a subgroup of patients with expected difficulties in RTW.
Furthermore, in a study of 378 patients with MTBI by Skandsen and colleagues, not being triaged for CT was associated with a reduced risk of post-concussion symptoms at three months after injury [27]. In our study, we found accordingly that patients with MTBI, who did not undergo CT imaging, were the fastest to return to work (median 3 days, n = 7) and reported post-concussion symptoms least frequently. These patients most likely do not meet the head CT criteria and are generally less symptomatic in the emergency units, thus not requiring longer sick leaves [8].
In our cohort, two patients had not fully returned to work at one year post-injury. One of them was still working partially at this point, and only one patient had not returned to work at all. Both patients had three types of traumatic intracranial lesions, indicating an overall more severe injury. Half of the patients with complicated MTBI had multiple different lesions, and their RTW parameters were worse. After excluding patients with multiple types of lesions, only patients with CT positive lesions had delayed RTW, up to one month after injury. No other RTW parameters showed significant differences at any time point.
The effect of multiple traumatic lesions on outcome in MTBI patients is largely unknown. In a large retrospective cohort by Isokuortti and colleagues, 6.4% of patients with MTBI had more than one type of traumatic intracranial lesion in CT imaging [13]. In a prospective observational study by Sharifuddin and colleagues, having multiple traumatic intracranial lesions (144 of 279 complicated MTBI patients) was an independent risk factor for worse repeat CT, which often lead to a neurosurgical intervention [26]. Patients with multiple traumatic intracranial lesions would highly likely benefit from additional care and adequate information and follow-up.
Regarding specific traumatic intracranial lesions in MTBI, their prevalence was comparable to previous studies [8, 13, 23, 36]. Lesions visible in CT (SDH, SAH and cerebral contusions) were associated with delayed RTW, but only cerebral contusions were associated with persistent post-concussion symptoms at one month after injury. However, the significance was weak in our study. This is consistent with previous studies, some of which have found cerebral contusions as a risk factor for persistent post-concussion symptoms and impaired recovery [30, 39, 41], although some studies point to the opposite [42]. Other lesions have not been considered a clinically significant risk factor from this point of view [7, 15].
None of the parameters affected functional recovery (GOS-E) of the patients. This is also consistent with previous literature and general recovery presentation of patients with MTBI, with other risk factors than traumatic intracranial lesions being more significant for long-term recovery [15, 25, 27, 30, 38].
However, a recent large study by Yuh and colleagues found that MTBI patients with SDH, SAH and/or cerebral contusions had impaired (GOS-E < 8) and unfavorable (GOS-E < 5) recovery up to 1 year after injury [40]. Similar to our results, these particular lesions often coincide. It is noteworthy that the recovery in terms of GOS-E was remarkably worse in the study by Yuh (only 47% of MTBI patients had GOS-E = 8 one year post-injury; respectively, in our cohort, 56% of patients had complete recovery at one month after injury). On the other hand, their patients with MTBI were often (31%) admitted to the intensive care unit even without traumatic intracranial lesions in CT imaging, suggesting a higher degree of injury severity. Patients in our cohort were only monitored up to 24–48 h (median 2 days, IQR 1.0–2.0) in case of complicated MTBI. In general, our patients were quite healthy and recovered well regardless of any examined parameters, although a very small subpopulation (n = 2) had impaired recovery.
The presence of even minor traumatic lesion causes inevitable, though mostly unwarranted concern in many patients, as well as in health care professionals [24]. Even though the mentioned lesions are small and clinically related to good prognosis, their presence per se might affect the threshold for recommending sick-leaves thus economically affecting both employer and society [22]. Well-timed RTW, however, promotes the subjective feeling of independence and success, as previously stated by Esbjörnsson and colleagues [10]. Prognosis of MTBI is good nevertheless and early reassuring and educational information is considered beneficial for MTBI recovery [9].
It has been well documented that RTW after MTBI is also affected by extracranial co-trauma, age, occupational factors (such as pre-injury unemployment, lower level of education, limited job independence and decision-making latitude), pre-injury substance and alcohol abuse, and post-concussion symptoms such as nausea or vomiting on hospital admission, fatigue and severe head/bodily pain [2, 5, 15, 29, 34, 38]. In addition, various psychological and psychosocial factors, such as mental health problems, acute psychological stress and perceived injustice, affect outcome after MTBI [4, 21, 35].
Our study has multiple strengths: RTW evaluation was precise with one-day accuracy up to one year after injury. In addition, our comprehensive imaging included both CT and 3 T MRI results and we were able to compare them. The study group was well characterized, including extensive neuropsychological and neuropsychiatric examinations. Finally, due to our particular role as a specialized outpatient clinic for patients with TBI, we are closely able to follow patients’ recovery and return to work after their discharge from emergency units.
We also recognize some limitations: first of all, the number of patients was quite low and did not allow analysis of subgroups with isolated or combined specific lesions or their localization. For instance, due to the low sample size of the CT-/MRI + subgroup, the role of subacute MRI in RTW remains unclear. Significant differences might have been achieved in a larger cohort study. Secondly, the majority of patients had favorable outcome nevertheless. Both of these aspects result in decreased likelihood of achieving statistical significance in any of the examined parameters. Finally, in this study, we focus solely on traumatic intracranial lesions, even though we recognize that outcome after MTBI is multifactorial.
In general, one intracranial lesion in MTBI does not seem to be a long-term predictor of disability to work. RTW after MTBI is undoubtedly multifactorial and dependent of other factors than the lesion itself. This imposes the necessity of further research in order to improve and individualize the care of MTBI patients.
Abbreviations
- CT:
-
Computer tomography
- GCS:
-
Glasgow Coma Scale
- GOS-E:
-
Glasgow Outcome Scale Extended
- IQR:
-
Interquartile range
- LOC:
-
Loss of consciousness
- MRI:
-
Magnetic resonance imaging
- MTBI:
-
Mild traumatic brain injury
- PTA:
-
Post-traumatic amnesia
- RPQ:
-
Rivermead Post-Concussion Symptoms Questionnaire
- RTW:
-
Return to work
- SAH:
-
Subarachnoid hemorrhage
- SDH:
-
Subdural hemorrhage
- TBI:
-
Traumatic brain injury
- WHO:
-
World Health Organization
References
Bloom B, Thomas S, Ahrensberg JM, Weaver R, Fowler A, Bestwick J et al (2018) A systematic review and meta-analysis of return to work after mild Traumatic brain injury. Brain Inj 32(13–14):1623–1636
Cancelliere C, Kristman VL, Cassidy JD, Hincapié CA, Côté P, Boyle E et al (2014) Systematic Review of Return to Work After Mild Traumatic Brain Injury: Results of the International Collaboration on Mild Traumatic Brain Injury Prognosis. Arch Phys Med Rehabil 95(3 Suppl):S201–S209
Carroll LJ, Cassidy JD, Holm L, Kraus J, Coronado VG (2004) WHO Collaborating Centre Task Force on Mild Traumatic Brain Injury. Methodological issues and research recommendations for mild traumatic brain injury: the WHO Collaborating Centre Task Force on Mild Traumatic Brain Injury. J Rehabil Med (43 Suppl):113–25. https://doi.org/10.1080/16501960410023877
Carroll LJ, Cassidy JD, Peloso PM, Borg J, von Holst H, Paniak C et al. Prognosis for mild traumatic brain injury: results of the WHO Collaborating Centre Task Force on Mild Traumatic Brain Injury. J Rehabil Med. 2004;(43 Suppl):84–105. https://doi.org/10.1080/16501960410023859
Chien DK, Hwang HF, Lin MR (2017) Injury severity measures for predicting return-to-work after a traumatic brain injury. Accid Anal Prev 98:101–107. https://doi.org/10.1016/j.aap.2016.09.025
de Koning ME, Scheenen ME, van der Horn HJ, Timmerman ME, Hageman G, Roks G et al (2017) Prediction of work resumption and sustainability up to 1 year after mild traumatic brain injury. Neurology 89(18):1908–1914
Ditty BJ, Omar NB, Foreman PM, Patel DM, Pritchard PR, Okor MO (2015) The nonsurgical nature of patients with subarachnoid or intraparenchymal hemorrhage associated with mild traumatic brain injury. J Neurosurg 123(3):649–653. https://doi.org/10.3171/2014.10.JNS132713
Easter JS, Haukoos JS, Meehan WP, Novack V, Edlow JA. Will Neuroimaging Reveal a Severe Intracranial Injury in This Adult With Minor Head Trauma?: The Rational Clinical Examination Systematic Review [published correction appears in JAMA. 2017 May 16;317(19):2021]. JAMA. 2015;314(24):2672–2681. https://doi.org/10.1001/jama.2015.16316
Eliyahu L, Kirkland S, Campbell S, Rowe BH (2016) The Effectiveness of Early Educational Interventions in the Emergency Department to Reduce Incidence or Severity of Postconcussion Syndrome Following a Concussion: A Systematic Review. Acad Emerg Med 23(5):531–542. https://doi.org/10.1111/acem.12924
Esbjörnsson E, Skoglund T, Sunnerhagen KS (2013) Fatigue, psychosocial adaptation and quality of life one year after traumatic brain injury and suspected traumatic axonal injury; evaluations of patients and relatives: a pilot study. J Rehabil Med 45(8):771–777. https://doi.org/10.2340/16501977-1170
Haacke EM, Duhaime AC, Gean AD, Riedy G, Wintermark M, Mukherjee P et al (2010) Common data elements in radiologic imaging of traumatic brain injury. J Magn Reson Imaging 32(3):516–543
Hsu HH, Lai WH, Yu HT, Xiao SH, Tsai YH, Wang KC et al (2021) Long-Term Presentation of Postconcussion Symptoms and Associated Factors: Analysis of Latent Class Modeling. Arch Clin Neuropsychol 36(1):62–73. https://doi.org/10.1093/arclin/acaa063 (PMID: 32839820)
Isokuortti H, Iverson GL, Silverberg ND, Kataja A, Brander A, Öhman J et al (2018) Characterizing the type and location of intracranial abnormalities in mild traumatic brain injury. J Neurosurg 129(6):1588–1597. https://doi.org/10.3171/2017.7.JNS17615 (PMID: 29328003)
Iverson GL, Lange RT, Wäljas M, Liimatainen S, Dastidar P, Hartikainen KM et al (2012) Outcome from Complicated versus Uncomplicated Mild Traumatic Brain Injury. Rehabil Res Pract 2012:415740. https://doi.org/10.1155/2012/415740
Jacobs B, Beems T, Stulemeijer M, van Vugt AB, van der Vliet TM, Borm GF et al (2010) Outcome prediction in mild traumatic brain injury: age and clinical variables are stronger predictors than CT abnormalities. J Neurotrauma 27(4):655–668. https://doi.org/10.1089/neu.2009.1059 (PMID: 20035619)
Jennett B, Snoek J, Bond MR, Brooks N (1981) Disability after severe head injury: observations on the use of the Glasgow Outcome Scale. J Neurol Neurosurg Psychiatry 44(4):285–293
Karr JE, Iverson GL, Williams MW, Huang SJ, Yang CC (2020) Complicated versus uncomplicated mild traumatic brain injuries: A comparison of psychological, cognitive, and post-concussion symptom outcomes. J Clin Exp Neuropsychol 42(10):1049–1058. https://doi.org/10.1080/13803395.2020.1841118
Kim B, Jeong H, Kim J, Kim T, Kim K, Lee H et al (2018) Incidence and risk factors of delayed intracranial hemorrhage in the emergency department. Am J Emerg Med 36(2):271–276. https://doi.org/10.1016/j.ajem.2017.08.009 (Epub 2017 Aug 4 PMID: 28811212)
King NS, Crawford S, Wenden FJ, Moss NE, Wade DT (1995) The Rivermead Post Concussion Symptoms Questionnaire: a measure of symptoms commonly experienced after head injury and its reliability. J Neurol 242(9):587–592
Moore MM, Pasquale MD, Badellino M (2012) Impact of age and anticoagulation: need for neurosurgical intervention in trauma patients with mild traumatic brain injury. J Trauma Acute Care Surg 73(1):126–130. https://doi.org/10.1097/TA.0b013e31824b01af (PMID: 22710784)
Mäki K, Nybo T, Hietanen M, Huovinen A, Marinkovic I, Isokuortti H, et al. Perceived Injustice After Mild Traumatic Brain Injury. J Head Trauma Rehabil. 2021 Jun 15. https://doi.org/10.1097/HTR.0000000000000698.
Norup A, Kruse M, Soendergaard PL, Rasmussen KW, Biering-Sørensen F. Socioeconomic Consequences of Traumatic Brain Injury: A Danish Nationwide Register-Based Study [published online ahead of print, 2020 Sep 17]. J Neurotrauma. 2020;https://doi.org/10.1089/neu.2020.7064.
Quigley MR, Chew BG, Swartz CE, Wilberger JE (2013) The clinical significance of isolated traumatic subarachnoid hemorrhage. J Trauma Acute Care Surg 74(2):581–584. https://doi.org/10.1097/TA.0b013e31827d6088
Rohacek M, Albrecht M, Kleim B, Zimmermann H, Exadaktylos A (2012) Reasons for ordering computed tomography scans of the head in patients with minor brain injury. Injury 43(9):1415–1418. https://doi.org/10.1016/j.injury.2012.01.001
Scholten AC, Haagsma JA, Andriessen TM, Vos PE, Steyerberg EW, van Beeck EF et al (2015) Health-related quality of life after mild, moderate and severe traumatic brain injury: patterns and predictors of suboptimal functioning during the first year after injury. Injury 46(4):616–624. https://doi.org/10.1016/j.injury.2014.10.064 (Epub 2014 Nov 4 PMID: 25476014)
Sharifuddin A, Adnan J, Ghani AR, Abdullah JM (2012) The role of repeat head computed tomography in the management of mild traumatic brain injury patients with a positive initial head CT. Med J Malaysia 67(3):305–308 (PMID: 23082423)
Skandsen T, Stenberg J, Follestad T, Karaliute M, Saksvik SB, Einarsen CE et al (2021) Personal Factors Associated With Postconcussion Symptoms 3 Months After Mild Traumatic Brain Injury. Arch Phys Med Rehabil 102(6):1102–1112. https://doi.org/10.1016/j.apmr.2020.10.106 (Epub 2020 Oct 27 PMID: 33127352)
Stippler M, Smith C, McLean AR, Carlson A, Morley S, Murray-Krezan C et al (2012) Utility of routine follow-up head CT scanning after mild traumatic brain injury: a systematic review of the literature. Emerg Med J 29(7):528–532. https://doi.org/10.1136/emermed-2011-200162 (Epub 2012 Feb 3 PMID: 22307924)
Stulemeijer M, van der Werf SP, Jacobs B, Biert J, van Vugt AB, Brauer JM et al (2006) Impact of additional extracranial injuries on outcome after mild traumatic brain injury. J Neurotrauma 23(10):1561–1569. https://doi.org/10.1089/neu.2006.23.1561 (PMID: 17020490)
Su BY, Guo NW, Chen NC, Lin SS, Chuang MT, Liao YC et al (2017) Brain contusion as the main risk factor of memory or emotional complaints in chronic complicated mild traumatic brain injury. Brain Inj 31(5):601–606. https://doi.org/10.1080/02699052.2016.1267800 (Epub 2017 Mar 28 PMID: 28350185)
Theadom A, Barker-Collo S, Jones K, Kahan M, Te Ao B, McPherson K et al (2017) Work Limitations 4 Years After Mild Traumatic Brain Injury: A Cohort Study. Arch Phys Med Rehabil 98(8):1560–1566. https://doi.org/10.1016/j.apmr.2017.01.010 (Epub 2017 Feb 8 PMID: 28188778)
van Eijck M, van der Naalt J, de Jongh M, Schoonman G, Oldenbeuving A, Peluso J et al (2018) Patients with Diffuse Axonal Injury Can Recover to a Favorable Long-Term Functional and Quality of Life Outcome. J Neurotrauma 35(20):2357–2364. https://doi.org/10.1089/neu.2018.5650 (Epub 2018 Jul 2 PMID: 29774826)
Voormolen DC, Haagsma JA, Polinder S, Maas AIR, Steyerberg EW, Vuleković P et al (2019) Post-Concussion Symptoms in Complicated vs. Uncomplicated Mild Traumatic Brain Injury Patients at Three and Six Months Post-Injury: Results from the CENTER-TBI Study. J Clin Med 8;8(11):1921
Wäljas M, Iverson GL, Lange RT, Liimatainen S, Hartikainen KM, Dastidar P et al (2014) Return to work following mild traumatic brain injury. J Head Trauma Rehabil 29(5):443–50. https://doi.org/10.1097/HTR.0000000000000002
Wäljas M, Lange RT, Hakulinen U, Huhtala H, Dastidar P, Hartikainen K et al (2014) Biopsychosocial outcome after uncomplicated mild traumatic brain injury. J Neurotrauma 31(1):108–124. https://doi.org/10.1089/neu.2013.2941 (Epub 2013 Nov 12 PMID: 23978227)
Washington CW, Grubb RL Jr (2012) Are routine repeat imaging and intensive care unit admission necessary in mild traumatic brain injury? J Neurosurg 116(3):549–557. https://doi.org/10.3171/2011.11.JNS111092
Wilde EA, McCauley SR, Kelly TM, Levin HS, Pedroza C, Clifton GL et al (2010) Feasibility of the Neurological Outcome Scale for Traumatic Brain Injury (NOS-TBI) in adults. J Neurotrauma 27(6):975–981
Willemse-van Son AH, Ribbers GM, Verhagen AP, Stam HJ (2007) Prognostic factors of long-term functioning and productivity after traumatic brain injury: a systematic review of prospective cohort studies. Clin Rehabil 21(11):1024–1037. https://doi.org/10.1177/0269215507077603
Yue JK, Winkler EA, Puffer RC, Deng H, Phelps RRL, Wagle S et al (2018) Temporal lobe contusions on computed tomography are associated with impaired 6-month functional recovery after mild traumatic brain injury: a TRACK-TBI study. Neurol Res 40(11):972–981. https://doi.org/10.1080/01616412.2018.1505416
Yuh EL, Jain S, Sun X, Pisica D, Harris MH, Taylor SR et al (2021) Pathological Computed Tomography Features Associated With Adverse Outcomes After Mild Traumatic Brain Injury: A TRACK-TBI Study With External Validation in CENTER-TBI. JAMA Neurol 78(9):1137–1148. https://doi.org/10.1001/jamaneurol.2021.2120.PMID:34279565;PMCID:PMC8290344
Yuh EL, Mukherjee P, Lingsma HF, Yue JK, Ferguson AR, Gordon WA et al (2013) Magnetic resonance imaging improves 3-month outcome prediction in mild traumatic brain injury. Ann Neurol. 73(2):224–35. https://doi.org/10.1002/ana.23783
Zare MA, Ahmadi K, Zadegan SA, Farsi D, Rahimi-Movaghar V. Effects of brain contusion on mild traumatic brain-injured patients [published correction appears in Int J Neurosci. 2013 Oct;123(10):744]. Int J Neurosci. 2013;123(1):65–69. https://doi.org/10.3109/00207454.2012.728653
Acknowledgements
The authors thank Simo Vanni and Aarne Ylinen (MD, PhD) for their contribution to this article. Written permissions were obtained.
Funding
Open Access funding provided by University of Helsinki including Helsinki University Central Hospital. This study was supported by grants from Dorothea, Jarl Walter and Karl Walter Perklén’s foundation, Päivikki and Sakari Sohlberg’s foundation, the Maire Taponen foundation, and the Helsinki University Hospital (HUS) Neurocenter.
The authors disclose no other financial or non-financial interests.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation and data collection were performed by all authors. Analysis were performed by Antti Huovinen. The first draft of the manuscript was written by Antti Huovinen and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript, and they all meet the ICMJE requirements for authorship.
Corresponding author
Ethics declarations
Ethics approval
All included patients gave their written informed consent. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee of Helsinki University Hospital (code 105/13/03/01/2014) and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Author statement
This manuscript was written as part of Dr. Huovinen’s doctoral thesis.
This article is part of the Topical Collection on Brain trauma
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Huovinen, A., Marinkovic, I., Isokuortti, H. et al. Return to work after mild traumatic brain injury: association with positive CT and MRI findings. Acta Neurochir 164, 1707–1717 (2022). https://doi.org/10.1007/s00701-022-05244-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-022-05244-4