Abstract
Background
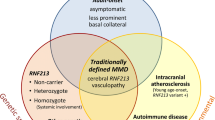
The etiology and genetic susceptibility of Moyamoya angiopathy (MMA) (Moyamoya disease, Moyamoya syndrome and unilateral type of MMA) still remain unclear. In Asian patient cohorts several HLA markers were described to be associated with MMA, but in Caucasians very little is known about genetic susceptibility of this angiopathy.
Method
We analysed DNA of 33 Caucasian patients with MMA for HLA-A, HLA-B, HLA-DRB1, and HLA-DQB1 markers, respectively. HLA frequencies of all 33 patients with MMA were compared with HLA-frequencies of Caucasian controls. Additionally, subgroup analysis of 22 patients with Moyamoya disease (MMD) and 11 patients with unilateral type of MMA was performed.
Findings
Significant association was observed for HLA-DRB1*03 and HLA-DRB1*13 in all 33 patients (P c < 0.001 and P c < 0.001, respectively). Moreover, HLA-A*02 (P c = 0.009); HLA-B*08 (P c = 0.009), and HLA-DQB1*03 (P c = 0.003) frequencies were higher in all patients with MMA when compared with the controls. In addition, in 22 patients with MMD a higher frequency of HLA-DRB1*03 (P c < 0.001) was observed when compared with controls.
Conclusions
The results of this study indicate a putative association of HLA markers with MMA in Caucasian patients. Further studies are needed to elucidate the role of human MHC in the pathogenesis of this angiopathy.
Similar content being viewed by others
References
Achrol AS, Guzman R, Lee M, Steinberg GK (2009) Pathophysiology and genetic factors in moyamoya disease. Neurosurg Focus 26:E4
Andreone V, Ciarmiello A, Fusco C, Ambrosanio G, Florio C, Linfante I (1999) Moyamoya disease in Italian monozygotic twins. Neurology 53:1332–1335
Aoyagi M, Ogami K, Matsushima Y, Shikata M, Yamamoto M, Yamamoto K (1995) Human leukocyte antigen in patients with moyamoya disease. Stroke 26:415–417
Bax M, van Heemst J, Huizinga TW, Toes RE (2011) Genetics of rheumatoid arthritis: what have we learned? Immunogenetics 63:459–466
Chessman D, Kostenko L, Lethborg T, Purcell AW, Williamson NA, Chen Z, Kjer-Nielsen L, Mifsud NA, Tait BD, Holdsworth R, Almeida CA, Nolan D, Macdonald WA, Archbold JK, Kellerher AD, Marriott D, Mallal S, Bharadwaj M, Rossjohn J, McCluskey J (2008) Human leukocyte antigen class I-restricted activation of CD8+ T cells provides the immunogenetic basis of a systemic drug hypersensitivity. Immunity 28:822–832
Fukui M (1997) Guidelines for the diagnosis and treatment of spontaneous occlusion of the circle of Willis (‘moyamoya’ disease). Research Committee on Spontaneous Occlusion of the Circle of Willis (Moyamoya Disease) of the Ministry of Health and Welfare, Japan. Clin Neurol Neurosurg 99(Suppl 2):S238–S240
Goyal MS, Hallemeier CL, Zipfel GJ, Rich KM, Grubb RL Jr, Chicoine MR, Moran CJ, Cross DT 3rd, Dacey RG Jr, Derdeyn CP (2010) Clinical features and outcome in North American adults with idiopathic basal arterial occlusive disease without moyamoya collaterals. Neurosurgery 67:278–285
Guo DC, Papke CL, Tran-Fadulu V, Regalado ES, Avidan N, Johnson RJ, Kim DH, Pannu H, Willing MC, Sparks E, Pyeritz RE, Singh MN, Dalman RL, Grotta JC, Marian AJ, Boerwinkle EA, Frazier LQ, LeMaire SA, Coselli JS, Estrera AL, Safi HJ, Veeraraghavan S, Muzny DM, Wheeler DA, Willerson JT, Yu RK, Shete SS, Scherer SE, Raman CS, Buja LM, Milewicz DM (2009) Mutations in smooth muscle alpha-actin (ACTA2) cause coronary artery disease, stroke, and Moyamoya disease, along with thoracic aortic disease. Am J Hum Genet 84:617–627
Han H, Pyo CW, Yoo DS, Huh PW, Cho KS, Kim DS (2003) Associations of Moyamoya patients with HLA class I and class II alleles in the Korean population. J Korean Med Sci 18:876–880
Heinemann FM (2009) HLA genotyping and antibody characterization using the Luminex multiplex technology. Transfus Med Hemother 36:273–278
Herve D, Touraine P, Verloes A, Miskinyte S, Krivosic V, Logeart D, Alili N, Laredo JD, Gaudric A, Houdart E, Metzger JP, Tournier-Lasserve E, Woimant F (2010) A hereditary moyamoya syndrome with multisystemic manifestations. Neurology 75:259–264
Hong SH, Wang KC, Kim SK, Cho BK, Park MH (2009) Association of HLA-DR and -DQ genes with familial Moyamoya disease in Koreans. J Korean Neurosurg Soc 46:558–563
Houkin K, Abe H, Yoshimoto T, Takahashi A (1996) Is "unilateral" moyamoya disease different from moyamoya disease? J Neurosurg 85:772–776
Ikeda H, Sasaki T, Yoshimoto T, Fukui M, Arinami T (1999) Mapping of a familial moyamoya disease gene to chromosome 3p24.2-p26. Am J Hum Genet 64:533–537
Inoue TK, Ikezaki K, Sasazuki T, Matsushima T, Fukui M (2000) Linkage analysis of moyamoya disease on chromosome 6. J Child Neurol 15:179–182
Inoue TK, Ikezaki K, Sasazuki T, Ono T, Kamikawaji N, Matsushima T, Fukui M (1997) DNA typing of HLA in the patients with moyamoya disease. Jpn J Hum Genet 42:507–515
Kamada F, Aoki Y, Narisawa A, Abe Y, Komatsuzaki S, Kikuchi A, Kanno J, Niihori T, Ono M, Ishii N, Owada Y, Fujimura M, Mashimo Y, Suzuki Y, Hata A, Tsuchiya S, Tominaga T, Matsubara Y, Kure S (2011) A genome-wide association study identifies RNF213 as the first Moyamoya disease gene. J Hum Genet 56:34–40
Khan N, Schuknecht B, Boltshauser E, Capone A, Buck A, Imhof HG, Yonekawa Y (2003) Moyamoya disease and Moyamoya syndrome: experience in Europe; choice of revascularisation procedures. Acta Neurochir (Wien) 145:1061–1071, discussion 1071
Kim SJ, Heo KG, Shin HY, Bang OY, Kim GM, Chung CS, Kim KH, Jeon P, Kim JS, Hong SC, Lee KH (2010) Association of thyroid autoantibodies with moyamoya-type cerebrovascular disease: a prospective study. Stroke 41:173–176
Kitahara T, Okumura K, Semba A, Yamaura A, Makino H (1982) Genetic and immunologic analysis on moya-moya. J Neurol Neurosurg Psychiatry 45:1048–1052
Kraemer M, Berlit P (2010) Primary central nervous system vasculitis and moyamoya disease: similarities and differences. J Neurol 257:816–819
Kraemer M, Berlit P, Diesner F, Khan N (2011) What is the expert’s option on antiplatelet therapy in moyamoya disease? Results of a worldwide Survey. Eur J Neurol. doi:10.1111/j.1468-1331.2011.03481.x
Kraemer M, Heienbrok W, Berlit P (2008) Moyamoya disease in Europeans. Stroke 39:3193–3200
Kraemer MHF, Horn P, Venker C, Berlit P, Krischek B, Khan N (2011) Inheritance of Moyamoya disease in a Caucasian familiy. Eur J Neurol. doi:10.1111/j.1468-1331.2011.03536.x
Kramer M, Berlit P (2010) [Moyamoya disease - a rare vasculopathy in Europeans]. Fortschr Neurol Psychiatr 78:542–550, quiz 551
Krischek B, Kasuya H, Khan N, Tatagiba M, Roder C, Kraemer M (2011) Genetic and clinical characteristics of Moyamoya disease in Europeans. Acta neurochirurgica Supplement 112
Li H, Zhang ZS, Dong ZN, Ma MJ, Yang WZ, Han C, Du MM, Liu YX, Yang H, Liu W, Duan L, Cao WC (2011) Increased thyroid function and elevated thyroid autoantibodies in pediatric patients with moyamoya disease: a case–control study. Stroke 42:1138–1139
Liu W, Morito D, Takashima S, Mineharu Y, Kobayashi H, Hitomi T, Hashikata H, Matsuura N, Yamazaki S, Toyoda A, Kikuta K, Takagi Y, Harada KH, Fujiyama A, Herzig R, Krischek B, Zou L, Kim JE, Kitakaze M, Miyamoto S, Nagata K, Hashimoto N, Koizumi A (2011) Identification of RNF213 as a susceptibility gene for moyamoya disease and its possible role in vascular development. PLoS One 6:e22542
Maiers M, Gragert L, Klitz W (2007) High-resolution HLA alleles and haplotypes in the United States population. Hum Immunol 68:779–788
Mallal S, Nolan D, Witt C, Masel G, Martin AM, Moore C, Sayer D, Castley A, Mamotte C, Maxwell D, James I, Christiansen FT (2002) Association between presence of HLA-B*5701, HLA-DR7, and HLA-DQ3 and hypersensitivity to HIV-1 reverse-transcriptase inhibitor abacavir. Lancet 359:727–732
Martorana D, Vaglio A, Greco P, Zanetti A, Moroni G, Salvarani C, Savi M, Buzio C, Neri TM (2006) Chronic periaortitis and HLA-DRB1*03: another clue to an autoimmune origin. Arthritis Rheum 55:126–130
Milewicz DM, Ostergaard JR, Ala-Kokko LM, Khan N, Grange DK, Mendoza-Londono R, Bradley TJ, Olney AH, Ades L, Maher JF, Guo D, Buja LM, Kim D, Hyland JC, Regalado ES (2010) De novo ACTA2 mutation causes a novel syndrome of multisystemic smooth muscle dysfunction. Am J Med Genet A 152A:2437–2443
Mineharu Y, Liu W, Inoue K, Matsuura N, Inoue S, Takenaka K, Ikeda H, Houkin K, Takagi Y, Kikuta K, Nozaki K, Hashimoto N, Koizumi A (2008) Autosomal dominant moyamoya disease maps to chromosome 17q25.3. Neurology 70:2357–2363
Mineharu Y, Takenaka K, Yamakawa H, Inoue K, Ikeda H, Kikuta KI, Takagi Y, Nozaki K, Hashimoto N, Koizumi A (2006) Inheritance pattern of familial moyamoya disease: autosomal dominant mode and genomic imprinting. J Neurol Neurosurg Psychiatry 77:1025–1029
Miskinyte S, Butler MG, Herve D, Sarret C, Nicolino M, Petralia JD, Bergametti F, Arnould M, Pham VN, Gore AV, Spengos K, Gazal S, Woimant F, Steinberg GK, Weinstein BM, Tournier-Lasserve E (2011) Loss of BRCC3 deubiquitinating enzyme leads to abnormal angiogenesis and is associated with syndromic moyamoya. Am J Hum Genet 88:718–728
Muller CR, Ehninger G, Goldmann SF (2003) Gene and haplotype frequencies for the loci hLA-A, hLA-B, and hLA-DR based on over 13,000 german blood donors. Hum Immunol 64:137–151
Oudshoorn M, Horn PA, Tilanus M, Yu N (2007) Typing of potential and selected donors for transplant: methodology and resolution. Tissue Antigens 69(Suppl 1):10–12
Park MH, Hong SH, Wang K-C (2010) HLA studies in Moyamoya disease. In: Cho BK, Tominaga T (eds) Moyamoya disease update. Springer, New York, pp 54–60
Parums DV, Brown DL, Mitchinson MJ (1990) Serum antibodies to oxidized low-density lipoprotein and ceroid in chronic periaortitis. Arch Pathol Lab Med 114:383–387
Parums DV, Dunn DC, Dixon AK, Mitchinson MJ (1990) Characterization of inflammatory cells in a patient with chronic periaortitis. Am J Cardiovasc Pathol 3:121–129
Roder C, Nayak NR, Khan N, Tatagiba M, Inoue I, Krischek B (2010) Genetics of moyamoya disease. J Hum Genet 55:711–716
Roder C, Peters V, Kasuya H, Nishizawa T, Takehara Y, Berg D, Schulte C, Khan N, Tatagiba M, Krischek B (2010) Polymorphisms in TGFB1 and PDGFRB are associated with Moyamoya disease in European patients. Acta Neurochir (Wien) 152:2153–2160
Roder C, Peters V, Kasuya H, Nishizawa T, Takehara Y, Berg D, Schulte C, Khan N, Tatagiba M, Krischek B (2011) Common genetic polymorphisms in moyamoya and atherosclerotic disease in Europeans. Childs Nerv Syst 27:245–252
Roder C, Peters V, Kasuya H, Nishizawa T, Wakita S, Berg D, Schulte C, Khan N, Tatagiba M, Krischek B (2011) Analysis of ACTA2 in European Moyamoya disease patients. Eur J Paediatr Neurol 15:117–122
Sakurai K, Horiuchi Y, Ikeda H, Ikezaki K, Yoshimoto T, Fukui M, Arinami T (2004) A novel susceptibility locus for moyamoya disease on chromosome 8q23. J Hum Genet 49:278–281
Takagi Y (2010) Vascular smooth muscle cell-related molecules and cells. In: Cho BK, Tominaga T (eds) Moyamoya disease update. Spinger, Heidelberg, pp 69–72
Takagi Y, Kikuta K, Nozaki K, Hashimoto N (2007) Histological features of middle cerebral arteries from patients treated for Moyamoya disease. Neurol Med Chir (Tokyo) 47:1–4
Yamamoto M, Aoyagi M, Tajima S, Wachi H, Fukai N, Matsushima Y, Yamamoto K (1997) Increase in elastin gene expression and protein synthesis in arterial smooth muscle cells derived from patients with Moyamoya disease. Stroke 28:1733–1738
Yamauchi T, Tada M, Houkin K, Tanaka T, Nakamura Y, Kuroda S, Abe H, Inoue T, Ikezaki K, Matsushima T, Fukui M (2000) Linkage of familial moyamoya disease (spontaneous occlusion of the circle of Willis) to chromosome 17q25. Stroke 31:930–935
Yonekawa Y, Fandimo J, Hug M, Wiesli M, Fujioka M, Khan N (2010) Moyamoya angiopathy in Europe. In: Cho BK, Tominaga T (eds) Moyamoya disease update. Springer, New York
Yonekawa Y, Khan N (2003) Moyamoya disease. Adv Neurol 92:113–118
Yonekawa Y, Ogata N, Kaku Y, Taub E, Imhof HG (1997) Moyamoya disease in Europe, past and present status. Clin Neurol Neurosurg 99(Suppl 2):S58–S60
Acknowledgements
The authors are thankful to Mr. J.C. Schwitalla, Ms. B. Lyss and Mr. B. Lappenkueper-Schaetz (Centre for Clinical Trials and Studies, Alfried-Krupp-Hospital, Essen, Germany) and to Ms. Sylke Hannemann for important help. Moreover, we thank our patients for participating in this study.
Conflicts of interest
M.K. has received grants, travel expenses and honoraria for lectures and/or educational activities from Bayer Schering Pharma, Biogen Idec, Merck Serono, Novartis Pharma and Teva Pharmaceutical Industries. Nevertheless, the study design and results are not influenced by commercial interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
Comment
Kraemer et al. analysed the DNA of 33 Caucasian patients with Moyamoya angiopathy for HLA markers. Significant association was observed for HLA-DRB1*03 and HLADRB1*13 in all 33 patients with Moyamoya angiopathy. Moreover, HLA-A*02; HLA-B*08, and HLADQB1*03 frequencies were higher in all patients with Moyamoya angiopathy when compared with the controls. The results of this study indicate a putative association of HLA markers with Moyamoya angiopathy in Caucasian patients. As discussed in this manuscript, in Asian patient cohorts several HLA markers were already reported to be associated with moyamoya disease (MMD), but in Caucasians very little is known about genetic susceptibility of MMD. This study shows several HLA markers which are associated with MMD. Frequency of MMD in Asian and Caucasians are different. This study has an important role in qualifying the genetic mechanism of MMD in Caucasian patients.
Yasushi Takagi
Kyoto, Japan
Rights and permissions
About this article
Cite this article
Kraemer, M., Horn, P.A., Roder, C. et al. Analysis of human leucocyte antigen genes in Caucasian patients with idiopathic Moyamoya angiopathy. Acta Neurochir 154, 445–454 (2012). https://doi.org/10.1007/s00701-011-1261-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-011-1261-5