Abstract
Purpose
The authors tried to assess the treatment outcomes depending on the extent of resection in axial chordomas and compare the outcome of two adjunctive therapies (external beam radiation therapy vs. stereotactic radiosurgery) following incomplete tumour resection in terms of local tumour control.
Patients and methods
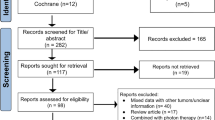
We retrospectively reviewed 30 consecutive patients with chordoma involving skull base, sacrum and mobile spine between 1993 and 2008. Their initial treatments had different extent of resection. Wide resection was performed for 12 (40%), subtotal resection and adjunctive radiotherapy/radiosurgery for 12 (40%), while six patients (20%) were solely treated with radiotherapy/radiosurgery. For these three groups, overall and progression-free survival rate were compared.
Results
The overall survival rate was 96% at 5 years and 67% at 10 years. Tumour progression-free survival (PFS) rates were 73% and 43% at 5 and 10 years, respectively.
Local tumour progression was seen in 67% in all patients, 58% in wide resection group, 67% in subtotal resection plus radiotherapy/radiosurgery group, and 75% in radiotherapy/radiosurgery group; however, this was not statistically significant (P = 0.69). Neither the extent of resection nor tumour location significantly influenced overall and progression-free survival (P > 0.05). With regard to the type of radiotherapy, tumour progression occurred in all lesions treated with external photon beam radiation therapy (EBRT) but only 38% of lesions treated with stereotactic radiosurgery (SRS) (P = 0.003).
Conclusions
Adjunctive radiotherapy/radiosurgery following subtotal resection showed comparable local control and survival to wide resection. SRS offered superior local tumour control compared to EBRT.




Similar content being viewed by others
References
Al-Mefty O, Borba LA (1997) Skull base chordomas: a management challenge. J Neurosurg 86:182–189
Ammirati M, Bernardo A (1999) Management of skull base chordoma. Crit Rev Neurosurg 9:63–69
Baratti D, Gronchi A, Pennacchioli E, Lozza L, Colecchia M, Fiore M, Santinami M (2003) Chordoma: natural history and results in 28 patients treated at a single institution. Ann Surg Oncol 10:291–296
Bergh P, Kindblom LG, Gunterberg B, Remotti F, Ryd W, Meis-Kindblom JM (2000) Prognostic factors in chordoma of the sacrum and mobile spine: a study of 39 patients. Cancer 88:2122–2134
Bjornsson J, Wold LE, Ebersold MJ, Laws ER (1993) Chordoma of the mobile spine. A clinicopathologic analysis of 40 patients. Cancer 71:735–740
Boriani S, Bandiera S, Biagini R, Bacchini P, Boriani L, Cappuccio M, Chevalley F, Gasbarrini A, Picci P, Weinstein JN (2006) Chordoma of the mobile spine: fifty years of experience. Spine (Phila Pa 1976) 31:493–503
Cheng EY, Ozerdemoglu RA, Transfeldt EE, Thompson RC (1999) Lumbosacral chordoma. Prognostic factors and treatment. Spine (Phila Pa 1976) 24:1639–1645
Chugh R, Tawbi H, Lucas DR, Biermann JS, Schuetze SM, Baker LH (2007) Chordoma: the nonsarcoma primary bone tumor. Oncologist 12:1344–1350
Dassoulas K, Schlesinger D, Yen CP, Sheehan J (2009) The role of gamma knife surgery in the treatment of skull base chordomas. J Neurooncol 94:243–248
Debus J, Schulz-Ertner D, Schad L, Essig M, Rhein B, Thillmann CO, Wannenmacher M (2000) Stereotactic fractionated radiotherapy for chordomas and chondrosarcomas of the skull base. Int J Radiat Oncol Biol Phys 47:591–596
Eriksson B, Gunterberg B, Kindblom LG (1981) Chordoma. A clinicopathologic and prognostic study of a Swedish national series. Acta Orthop Scand 52:49–58
Fagundes MA, Hug EB, Liebsch NJ, Daly W, Efird J, Munzenrider JE (1995) Radiation therapy for chordomas of the base of skull and cervical spine: patterns of failure and outcome after relapse. Int J Radiat Oncol Biol Phys 33:579–584
Ferraresi V, Nuzzo C, Zoccali C, Marandino F, Vidiri A, Salducca N, Zeuli M, Giannarelli D, Cognetti F, Biagini R (2010) Chordoma: clinical characteristics, management and prognosis of a case series of 25 patients. BMC Cancer 10:22
Gay E, Sekhar LN, Rubinstein E, Wright DC, Sen C, Janecka IP, Snyderman CH (1995) Chordomas and chondrosarcomas of the cranial base: results and follow-up of 60 patients. Neurosurgery 36:887–896, discussion 896-7
Glosser G, McManus P, Munzenrider J, Austin-Seymour M, Fullerton B, Adams J, Urie MM (1997) Neuropsychological function in adults after high dose fractionated radiation therapy of skull base tumors. Int J Radiat Oncol Biol Phys 38:231–239
Hasegawa T, Ishii D, Kida Y, Yoshimoto M, Koike J, Iizuka H (2007) Gamma Knife surgery for skull base chordomas and chondrosarcomas. J Neurosurg 107:752–757
Ito E, Saito K, Okada T, Nagatani T, Nagasaka T (2010) Long-term control of clival chordoma with initial aggressive surgical resection and gamma knife radiosurgery for recurrence. Acta Neurochir (Wien) 152:57–67, discussion 67
Jawad MU, Scully SP (2010) Surgery significantly improves survival in patients with chordoma. Spine (Phila Pa 1976) 35:117–123
Jemal A, Siegel R, Xu J, Ward E (2010) Cancer statistics, 2010. CA Cancer J Clin 60:277–300
Jian BJ, Bloch OG, Yang I, Han SJ, Aranda D, Tihan T, Parsa AT (2010) Adjuvant radiation therapy and chondroid chordoma subtype are associated with a lower tumor recurrence rate of cranial chordoma. J Neurooncol 98:101–108
Krishnan S, Foote RL, Brown PD, Pollock BE, Link MJ, Garces YI (2005) Radiosurgery for cranial base chordomas and chondrosarcomas. Neurosurgery 56:777–784, discussion 777-84
Liu AL, Wang ZC, Sun SB, Wang MH, Luo B, Liu P (2008) Gamma knife radiosurgery for residual skull base chordomas. Neurol Res 30:557–561
Martin JJ, Niranjan A, Kondziolka D, Flickinger JC, Lozanne KA, Lunsford LD (2007) Radiosurgery for chordomas and chondrosarcomas of the skull base. J Neurosurg 107:758–764
Miller RC, Foote RL, Coffey RJ, Gorman DA, Earle JD, Schomberg PJ, Kline RW (1997) The role of stereotactic radiosurgery in the treatment of malignant skull base tumors. Int J Radiat Oncol Biol Phys 39:977–981
Muthukumar N, Kondziolka D, Lunsford LD, Flickinger JC (1998) Stereotactic radiosurgery for chordoma and chondrosarcoma: further experiences. Int J Radiat Oncol Biol Phys 41:387–392
Ozaki T, Hillmann A, Winkelmann W (1997) Surgical treatment of sacrococcygeal chordoma. J Surg Oncol 64:274–279
Park L, Delaney TF, Liebsch NJ, Hornicek FJ, Goldberg S, Mankin H, Rosenberg AE, Rosenthal DI, Suit HD (2006) Sacral chordomas: impact of high-dose proton/photon-beam radiation therapy combined with or without surgery for primary versus recurrent tumor. Int J Radiat Oncol Biol Phys 65:1514–1521
Tzortzidis F, Elahi F, Wright D, Natarajan SK, Sekhar LN (2006) Patient outcome at long-term follow-up after aggressive microsurgical resection of cranial base chordomas. Neurosurgery 59:230–237, discussion 230-7
Wu Z, Zhang J, Zhang L, Jia G, Tang J, Wang L, Wang Z (2010) Prognostic factors for long-term outcome of patients with surgical resection of skull base chordomas-106 cases review in one institution. Neurosurg Rev 33:451–456
Yonemoto T, Tatezaki S, Takenouchi T, Ishii T, Satoh T, Moriya H (1999) The surgical management of sacrococcygeal chordoma. Cancer 85:878–883
York JE, Kaczaraj A, Abi-Said D, Fuller GN, Skibber JM, Janjan NA, Gokaslan ZL (1999) Sacral chordoma: 40-year experience at a major cancer center. Neurosurgery 44:74–79, discussion 79–80
Acknowledgement
We thank Ahmed Kamal Abul-Magd, MD, for assisting in statistical analyses.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Comments
Despite the fact that the overall number of patients is not negligible, it encompasses varied locations, degrees of resection and corresponding combinations of adjuvant strategies which breaks the total into numerous fragmentary groups rendering statistical value somewhat questionable.
It is a well-known fact that chordomas may behave quite differently despite undergoing similar treatment protocols. In fact, even after radical resections some tumours have the ability to just keep coming back again from apparently nothing, forming a group of more aggressive behaviour. These tumours may only be distinguished from the pathological standpoint by an elevated cell turnover patented by the Ki-67 or MIB-1, or else by a pattern of higher malignancy (references 1 and 2). No information regarding these particularities is given for the series.
The authors have found no statistical difference between patients treated with different degree of resection, location of tumour or progression of the disease. In fact the only difference acknowledged seems to lie with the time for recurrence between patients treated with EBRT or with GK/CK following surgical resection. However the follow-up for the EBRT group is significantly longer than that for GK/CK (91 versus 62 months). It would be interesting to know the graphic distribution of signalled recurrences along the X-line coordinate for time. If most recurrences, as it likely is, are detected more consistently as the time expands from the date of surgery perhaps the difference found by the authors will likely be attenuated once the follow-up for GK/CK is taken to a mean similar to the one for EBRT.
1. Skull base chordomas: correlation of tumour doubling time with age, mitosis and Ki-67 proliferation index. Holton JL, Steel T, Luxsuwong M, Crockard HA, Revesz T. Neuropathol Appl Neurobiol. 2000 Dec; 26(6):497–503
2. Skull base and Nonskull base chrodomas. Clinicopathologic and immunohistochemical study with special reference to nuclear pleomorphism and proliferative ability. Naka T, Boltze C, Samii A, Herold C, Ostertag H, Iwamoto Y, Oda Y, Tsuneyoshi M, Kuester D, Roessner A. Cancer Nov 1, 2003/Vol 98/Numb 9
Manuel Cunha e Sa
Almada, Portugal
Comments
This is a well-written and well-researched paper regarding the outcome of different management protocols for chordomas, comparing the usual combinations of microsurgery, fractionated radiotherapy and stereotactic radiosurgery, including patients treated with radiosurgery alone.
Chordomas are particularly difficult tumours to manage not only because of their usual late presentation, locally aggressive tendency for recurrence and traditionally acknowledged radio resistance, but also because fewer surgeons and indeed few departments see sufficient numbers to formulate their own policy. Therefore this paper is of particular interest.
The authors have found that wide resection alone or resection with radiotherapy was less effective than radiation treatment alone (although this was not statistically significant). Interestingly the extent of resection was not a relevant factor, underlining the importance of radiation in achieving our aim. Local tumour progression was not prevented by external beam radiotherapy in any patient whereas stereotactic radiosurgery achieved this in 62%. On the face of it, these statements would lead to a chosen policy of using radiosurgery but it has to be acknowledged that many lesions are not suitable for focused radiation by virtue of their large size at presentation. It seems sensible however not to pursue aggressive removal and use microsurgery as an enabling procedure before using radiosurgery. It is also important that in keeping with observations about other benign and low-level malignant pathologies fractionation may not offer an advantage as long as the tumour is geometrically suitable for single fraction treatment.
Andras Kemeny
Sheffield, UK
Rights and permissions
About this article
Cite this article
Eid, A.S., Chang, UK., Lee, SY. et al. The treatment outcome depending on the extent of resection in skull base and spinal chordomas. Acta Neurochir 153, 509–516 (2011). https://doi.org/10.1007/s00701-010-0928-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-010-0928-7