Abstract
Background
Implantation of 1,3-bis (2-chloroethyl)-1-nitrosourea (BCNU) wafer for malignant glioma is not recommended in the case of surgical opening of the ventricular system during microsurgical tumor resection because the wafer material may dislocate from the resection cavity into the ventricular system and cause obstructive hydrocephalus. TachoSil is an adhesive collagen fleece used in different surgical disciplines that provides an air- and liquid-tight seal closing communications between the ventricular system and the resection cavity after tumor removal.
Methods
Occlusion of ventricular defects with TachoSil after microsurgical glioma resection was performed in two patients with newly diagnosed and seven patients with recurrent malignant glioma prior to BCNU wafer implantation into the resection cavity. Early postoperative cranial computed tomography (CCT)/MRI and follow-up MRI at 3 months’ intervals were performed with a median follow-up of 10.4 months.
Results
The collagen fleece was identified as a linear structure hypodense/hypointense to white matter on postoperative CT/MRI separating the resection cavity from the ventricular lumen in all cases. In no case did early CCT/MRI or follow-up MRI reveal wafer material within the ventricular system. In no case did signs of obstructive hydrocephalus occur.
Conclusion
Sealing of the ventricular system using a fibrinogen-coated collagen fleece effectively separates the resection cavity from the ventricular system and allows implantation of BCNU wafers into the resection cavity. No morphological evidence for wafer material dislocation into the ventricular system or obstruction of CSF pathways was found in nine patients who received 41 follow-up MRI over 10.4 months of follow-up.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Surgical opening of the ventricular system frequently occurs during the resection of brain tumors located near the ventricles and is usually well tolerated [6]. Therefore, a need to close such openings rarely occurs. In terms of alteration of the pattern of dissemination along the ependyma and occurrence of tumor dissemination along the CSF pathways by surgical entry of the ventricular system during glioma resection, the literature remains controversial [5]. Whether surgical occlusion of ventricular openings in the setting of glioma or metastasis surgery prevents CSF dissemination of tumors has not been systematically studied.
However, opening of the ventricles limits the use of 1,3-bis (2-chloroethyl)-1-nitrosourea (BCNU) wafer implantation into the resection cavity for local chemotherapy of malignant gliomas [19]. BCNU wafer implantation is not recommended in such situations because the wafer material may dislocate into the ventricular system and cause acute occlusive hydrocephalus [4]. Furthermore, high concentrations of BCNU at the implantation site in direct communication with the ventricular system may cause self-limiting ventriculitis associated with a transient hydrocephalus [8]. Because three clinical phase III trials have demonstrated a survival benefit for patients implanted with BNCU wafers for both newly diagnosed malignant glioma [21–23] and recurrent malignant gliomas [3] and several recent reports have now indicated a safe integration of BCNU wafer treatment into multimodal treatment strategies with promising preliminary survival data [1, 2, 12, 13, 15], we have considered occlusion of larger surgical ventricular openings prior to wafer implantation using an adhesive collagen fleece (TachoSil®, Nycomed GmbH, Linz, Austria).
An important property of TachoSil® is to activate the coagulation cascade by recapitulating the final steps of physiological blood clotting in order to seal the corresponding tissue it is applied to. TachoSil® consists of an equine-derived collagen type I matrix covered with human fibrinogen and thrombin. In contact with fluid, TachoSil® adheres to the surface of a wound by thrombin-mediated transformation of fibrin into fibrin monomer. Fibrin monomers spontaneously polymerize into a fibrin clot, which cross-links for stabilization mediated by endogenous factor XIII [18]. Coagulation occurs within the collagen matrix and the wound within 5 min, which leads to conglutination of the collagen matrix and the wound surface, resulting in an air- and liquid-tight seal [18]. The adhesive matrix resists tension force and, because of its elasticity, adapts the movements of the substrate. The latter has been demonstrated in experimental lung surgery and by the use of TachoSil® in occlusion of air leaks after lobectomy in lung tissue [10, 11]. TachoSil® absorbs within 12 weeks by phagocytosis and fibrinolysis and is replaced by endogenous granulation tissue. This collagen fleece has been widely used in vascular, lung, hepatic, urological, and gynecological surgeries [18].
Neurosurgical applications of TachoSil® (earlier marketed under the name TachoComb®) have mainly focused on the repair of dural defects [14, 17]. We have used TachoSil® in a prospective case collection study of nine patients for occlusion of surgical openings of the ventricular system during surgery for malignant gliomas scheduled for implantation of BCNU wafers into the tumor resection cavity. Postoperative imaging demonstrated occlusion of the ventricular openings and no evidence of dislocation of wafer material into the ventricular system over a median follow-up of 10.4 months. In no case did postoperative imaging revealed a hydrocephalus, and in no case did clinical signs suggest a complication associated with the CSF pathways.
Methods
Patients and surgical procedure
From August 2005 to July 2010, 19 patients with newly diagnosed malignant gliomas and 24 patients treated for recurrent malignant gliomas at our institution were treated by microsurgical tumor resection followed by implantation of BCNU wafers. In nine patients (21%), the location of tumors required wide opening of the ventricles to allow gross total resection of tumors, resulting in fenestrations of the ventricular system larger than 10 mm. The diameter of BCNU wafers measures 14 mm. Because eroding wafers decrease in size and the geometry of resection cavities frequently requires implantation of wafer fragments, openings of the ventricular system larger than 10 mm were not considered safe for wafer implantation. All patients gave written consent to the surgical procedure, including tumor resection with opening of the ventricular system followed by occlusion using a collagen fleece and local BCNU wafer implantation. In one patient with newly diagnosed and eight patients with recurrent malignant gliomas, surgical openings of the ventricular system were closed using TachoSil® (Table 1). The collagen fleece was cut to cover the entire defect and overlap with the adjacent resection edge by at least 5 mm. The fleece was applied wet and pressed to the tissue surface using dry surgical cotton for 1 min; then, the cotton was removed and the resection cavity was kept dry for 5 min. The resection cavity was rinsed to ensure hemostasis and the BCNU wafers implanted (Fig. 1). The dura and wound were closed and perioperative antibiotics and dexamethasone schedule was given adhering to a protocol specifically designed to avoid wound healing abnormalities and to minimize other risks associated with local chemotherapy using BCNU wafers [7].
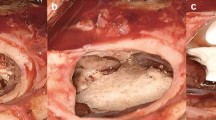
Occlusion of surgical opening of the ventricular system after resection of a recurrent malignant glioma (patient 7) using a fibrinogen-coated collagen fleece (TachoSil®) prior to implantation of BCNU wafers (Gliadel). a Opening of the right frontal horn of the ventricle. b Occlusion of the ventricular fenestration by TachoSil®. c Implantation of seven BCNU-containing wafers (Gliadel)
Postoperative imaging and follow-up
As routine protocol at our institution 4 h postoperatively, all patients received a cranial computed tomography (CCT) to rule out early perioperative complications. Within 48 h, an MRI scan was obtained and prior to discharge a CCT or MRI performed at postoperative day 10 according to a follow-up protocol developed for patients receiving BCNU wafer chemotherapy. After discharge, patients were scheduled for visits at the neurosurgical–neurooncology outpatient clinic at postoperative days 14, 21, 42, and 65 for wound inspection, evaluation of blood parameters, and clinical status. Three months postoperatively, a follow-up MRI was done and repeated at 3-month intervals [7].
Results
Intraoperative pliability and versatility
After contact with irrigation fluid, the collagen fleece became pliable and easily adapted to the surface contour of the resection cavity. After modeling and gently pressing the fleece against the tissue with dry surgical cotton, in all cases, the fleece firmly adhered to the wound. In no case was air or CSF leakage from the ventricular system into the resection cavity observed for the remaining time of hemostasis and wafer implantation until dural closure.
Postoperative imaging
Four-hour postoperative CT scans in all patients showed residual air and the BCNU wafers appearing as hyperdense linear structures within the resection cavity. The collagen fleece could be identified as a linear structure hypodense to white matter separating the resection cavity from the ventricular lumen (Fig. 2a). On a 24-h postoperative MRI (Fig. 2b, c), as previously described, the BCNU wafers in all patients appeared as hypointense structures; after administration of gadolinium contrast material, no enhancement of the wafer structure or adjacent brain was observed [8, 9, 16]. The collagen fleece appeared hypointense on T1-weighted MRI between the ventricular lumen and the resection cavity. In all cases, tri-planar MRI demonstrated a separation of the resection cavity from the ventricular system by the collagen fleece. In no case did early CCT or MRI reveal wafer material within the ventricular system or signs of obstructive hydrocephalus (Table 2). In no case was contrast enhancement of the ependyma as a sign of aseptic ventriculitis observed [8].
Appearance of collagen fleece (TachoSil®) and BCNU wafers (Gliadel) on postoperative imaging in a case of recurrent malignant glioma. a Four-hour postoperative CCT: The collagen fleece is occluding the entire ventricular defect; BCNU wafers are detectable within the resection cavity. b Postoperative (24-h) MRI T2w: Hypointense collagen fleece separates the tumor cavity from the ventricle. c T1w + GD, there is no enhancement of the wafer structure or the adjacent brain and the collagen fleece appears hypointense; otherwise, there were no signs of enhancement of the ependyma
On 3 months’ follow-up MRI, the resection cavity after BCNU wafer implantation typically showed a ring-shaped contrast enhancement around the resection cavity with varying degrees of perifocal edema [7]. This phenomenon has been well documented in the literature. Contrast enhancement will usually resolve within 3–6 months, but may persist for up to 10 months. Wafer remnants can be identified in the resection cavity on MRI for 4–6 months [9, 16]. Expectedly, this appearance of the resection cavity and dynamics of contrast enhancement were also confirmed in this study. On 3 months’ postoperative MRI, the collagen fleece appears replaced by a granulation tissue with spongy structure, hypointense on T1- and hyperintense on T2-weighted MRI (Fig. 3c, d).
Occlusion of ventricular opening with collagen fleece (TachoSil®) followed by BCNU wafer (Gliadel) implantation in a case of multifocal glioblastoma. Reoperation was done for a rapidly progressive left frontal focus under chemotherapy. Twenty-four-hour MRI (a) and 10-day postoperative CCT (b) demonstrating the hypodense collagen fleece separating the resection cavity from the ventricular system in which the wafer material can be identified. c Three-month postoperative MRI T1w + GD. Ring-shaped contrast enhancement of the resection cavity was suspected as early local progression, causing mass effect. Residues of collagen fleece appear hypointense, occluding the left frontal horn of the ventricle. d Three-month postoperative T2w MRI. Spongy tissue replaces the collagen fleece (yellow arrow). Wafer remnants can be identified in the resection cavity. Despite mass effect of the cystic cavity, there are no signs of communication through the ventricular defect occluded with the collagen fleece 3 months after surgery
None of the 41 follow-up MRIs in nine patients followed for a median of 10.4 months (range, 2–24.6 months) revealed signs of wafer material dislocated to the ventricular system at any time point.
Clinical course and outcome
The postoperative recovery of all patients was uneventful, and the median postoperative hospitalization was 11.5 days. Wound healing abnormalities occurred in one patient without necessity of surgical intervention. No patient developed clinical signs of acute or subacute hydrocephalus in the perioperative phase; no patient required treatment for hydrocephalus during the entire follow-up. For seven patients, follow-up MRI demonstrated signs of tumor progression, and at the time of data summary, four patients had died. Clinical deterioration occurred in five patients and in all cases was associated with MRI-documented progression of tumors. In no case was MRI-documented tumor progression associated with morphological signs of communication of the resection cavity with the previously collagen fleece-sealed ventricular system, and in no case was dislocation of wafer material from the cavity or necrotic tumor focus observed despite mass effect (compare Fig. 3c, d).
Discussion
While generally surgical entry into the ventricular system does not require reconstruction, an open communication of the resection site and the ventricles may pose a significant problem for strategies that rely on the direct delivery of pharmaceuticals to the resection cavity. The distribution of compounds delivered by direct infusion into the resection cavity or through catheters placed into tissue surrounding the resection site as used in convection-enhanced delivery may be difficult, if not impossible, to predict in the case of open communication of the resection site with the ventricular system. For local chemotherapy using biodegradable wafers, communication of the implantation site and the ventricular system has been an exclusion criterion in the pivotal phase III trials because of concerns that the wafer material may dislocate into the ventricular system, causing obstruction of the CSF pathways [3, 21, 23]. However, it was suggested that openings of the ventricle significantly smaller than the diameter of a wafer do not preclude the use of BCNU wafers [20]. Gallego et al. [4] reported three cases of fatal hydrocephalus in patients treated with BCNU wafers after wide opening of the ventricular system 1–5 months after implantation. In all three cases, the wafers had been fixed to the wall of the resection site using fibrin glue, but the ventricular system itself remained open. Imaging in all three cases failed to directly visualize dislocation of the wafer material causing obstruction of the CSF pathways, but because the stability of fibrin glue in situ remains within days, it is unlikely that it can prevent wafers from dislocation over the course of wafer biodegradation. MRI imaging has well demonstrated that wafer remnants may be found within the resection cavity 4–6 months after implantation, suggesting that any strategy not permanently closing large communications between the resection site and the ventricular system may be dangerous in this setting. However, dislocation of wafers may not be the only mechanism resulting in hydrocephalus. We have recently reported a case of transient hydrocephalus in a patient who received BCNU wafer implantation into a resection cavity with a sub-wafer diameter communication to the ventricular system [8]. Based on the observation of the transient nature of the hydrocephalus manifesting 10 days post-implantation, resolving spontaneously by postoperative day 20, we have suggested that high concentrations of BCNU entering the ventricular system may cause an aseptic ventriculitis and subsequent self-limiting hydrocephalus [8]. In standard implantation settings with resection cavities communicating with the subarachnoid space, a similar phenomenon is frequently observed. Aseptic meningitis with clinical signs of meningitis, CSF pleocytosis in the absence of an infectious agent, and a self-limiting clinical course occurred in approximately 5% of patients treated with BCNU wafers and has been suggested to be a result of high intrathecal BCNU concentrations [13]. These observations strongly argue for a robust and permanent occlusion of communications of the resection site with the ventricular system if local chemotherapy is considered.
Conclusion
Surgical entry of the ventricular system during resection of malignant brain tumors has limited implantation of BCNU wafers for local chemotherapy. Postoperative imaging showed that sealing of the ventricular system using a fibrinogen-coated collagen fleece separates the resection cavity from the ventricular system with no evidence of wafer dislocation into the ventricular system on long-term follow-up MRI. The implantation of the collagen fleece was well tolerated in this small series of patients with no evidence of implant-related adverse events or adverse events associated with the CSF pathways. Based on our experience of occlusion of nine ventricular openings larger than 10 mm in nine patients followed over a mean follow-up of 10.4 months, we conclude that occlusion of these defects using TachoSil is safe. Early postoperative imaging and routine 3-month MRI follow-up in a total of 41 MRI studies have not revealed any evidence of early or late wafer dislocation, obstructive hydrocephalus, or any evidence of transient or permanent hydrocephalus in patients where the ventricular defect had been entirely covered with the collagen fleece.
References
Affronti ML, Heery CR, Herndon JE, Rich JN, Reardon DA, Desjardins A, Vredenburgh JJ, Friedman AH, Bigner DD, Friedman HS (2009) Overall survival of newly diagnosed glioblastoma patients receiving carmustine wafers followed by radiation and concurrent temozolomide plus rotational multiagent chemotherapy. Cancer 115(15):3501–3511
Bock HC, Puchner MJ, Lohmann F, Schütze M, Koll S, Ketter R, Buchalla R, Rainov N, Kantelhardt SR, Rohde V, Giese A (2010) First line treatment of malignant glioma with carmustine implants followed by concomitant radiochemotherapy: a multicenter experience. Neurosurg Rev 33(4):441–449
Brem H, Piantadosi S, Burger PC, Walker M, Selker R, Vick NA, Black K, Sisti M, Brem S, Mohr G (1995) Placebo-controlled trial of safety and efficacy of intraoperative controlled delivery by biodegradable polymers of chemotherapy for recurrent gliomas. The Polymer-Brain Tumor Treatment Group. Lancet 345(8956):1008–1012
Gallego JM, Barcia JA, Barcia-Mariño C (2007) Fatal outcome related to carmustine implants in glioblastoma multiforme. Acta Neurochir 149(3):261–265
Giese A, Westphal M (1996) Glioma invasion in the central nervous system. Neurosurgery 39(2):235–250, discussion 250–252
Giese A, Bjerkvig R, Berens ME, Westphal M (2003) Cost of migration: invasion of malignant gliomas and implications for treatment. J Clin Oncol 21:1624–1636
Giese A, Bock HC, Kantelhardt SR, Rohde V (2010) Risk management in the treatment of malignant gliomas with BCNU wafer implants. Cen Eur Neurosurg 71(4):199–206. doi:s-1242775/s-0029-1242775
Giese A, Bock HC, Rohde V (2010) Transient hydrocephalus following carmustine wafer implantation for treatment of malignant glioma: case illustration and a review of the literature. Cen Eur Neurosurg (in press)
Hammoud DA, Belden JC, Ho AC, Dal Pan GJ, Herskovits EH, Hilt DC, Brem H, Pomper MG (2003) The surgical bed after BCNU polymer wafer placement for recurrent glioma: serial assessment on CT and MR imaging. AJR 180(5):469–475
Izbicki JR, Kreusser T, Meier M, Prenzel KL, Knoefel WT, Passlick B, Kuntz G, Schiele U, Thetter O (1994) Fibrin-glue-coated collagen fleece in lung surgery—experimental comparison with infrared coagulation and clinical experience. Thorac Cardiovasc Surg 42:306–309
Lang G, Csekeö A, Stamatis G, Lampl L, Hagman L, Marta GM, Mueller MR, Klepetko W (2004) Efficacy and safety of topical application of human fibrinogen/thrombin-coated collagen patch (TachoComb) for treatment of air leakage after standard lobectomy. Eur J Cardiothorac Surg 25(2):60–66
LaRocca R, Glisson S, Hargis J, Petruska D, Villanueva W, Morassutti D, Horne D, Amin-Zimmermann F (2006) High-grade glioma treated with surgery; carmustine wafer; postoperative radiation; and procarbazine, lomustine, and vincristine chemotherapy. Neurosurg 15(3):167–171
Menei P, Metellus P, Parot-Schinkel E, Loiseau H, Capelle L, Jacquet G, Neuro-oncology Club of the French Society of Neurosurgery (2010) Biodegradable carmustine wafers (Gliadel) alone or in combination with chemoradiotherapy: the French experience. Ann Surg Oncol 17:1740–1746
Nistor RF, Chiari FM, Maier H, Hehl K (1997) The fixed combination of collagen with components of fibrin adhesive—a new hemostypic agent in skull base procedures. Skull Base Surg 7:23–30
Pan E, Mitchell SB, Tsai JS (2008) A retrospective study of the safety of BCNU wafers with concurrent temozolomide and radiotherapy and adjuvant temozolomide for newly diagnosed glioblastoma patients. J Neurooncol 88:353–357
Prager JM, Grenier Y, Cozzens JW, Chiowanich P, Gorey MT, Meyer JR (2000) Serial CT and MR imaging of carmustine wafers. AJNR 21(1):119–123
Reddy M, Scho A, Reddy B, Saringer W, Weigel G, Matula C (2002) A clinical study of a fibrinogen-based collagen fleece for dural repair in neurosurgery. Acta Neurochir 144:265–269
Rickenbacher A, Breitenstein S, Lesurtel M, Frilling A (2009) Efficacy of TachoSil a fibrin-based haemostat in different fields of surgery—a systematic review. Expert Opin Biol Ther 9(7):897–907
Sabel M, Giese A (2008) Safety profile of carmustine wafers in malignant glioma: a review of controlled trials and a decade of clinical experience. Curr Med Res Opin 24(11):3239–3257
Sampath P, Brem H (1998) Implantable slow-release chemotherapeutic polymers for the treatment of malignant brain tumors. Cancer Control 5(2):130–137
Valtonen S, Timonen U, Toivanen P, Kalimo H, Kivipelto L, Heiskanen O, Unsgaard G, Kuurne T (1997) Interstitial chemotherapy with carmustine-loaded polymers for high-grade gliomas: a randomized double-blind study. Neurosurgery 41(1):44–49
Westphal M, Ram Z, Riddle V, Hilt D, Bortey E, Executive Committee of the Gliadel Study Group (2006) Gliadel wafer in initial surgery for malignant glioma: long-term follow-up of a multicenter controlled trial. Acta Neurochir 148(3):269–275
Westphal M, Hilt DC, Bortey E, Delavault P, Olivares R, Warnke PC, Whittle IR, Jääskeläinen J, Ram Z (2003) A phase 3 trial of local chemotherapy with biodegradable carmustine (BCNU) wafers (Gliadel wafers) in patients with primary malignant glioma. Neuro Oncol 5(2):79–88
Conflicts of interest
None.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Comment
Bock et al. report their experience about the implantation of BCNU wafers after high-grade glioma removal when a communication larger than 10 mm is finally detected between surgical and ventricular cavities. They implanted BCNU wafers on the cavity walls after covering the defect with an adhesive collagen fleece (TachoSil). No post-implantation cases of obstructive hydrocephalus occurred. No other complications related to the CSF pathway were recorded. The issue is remarkable and the authors’ effort is appreciable considering the worldwide use of BCNU wafers and the frequent wide opening of ventricles required to achieve gross total tumor removal. In our experience, a piece of gelatine sponge fixed with fibrin glue then covered with oxidized cellulose was suitable to manage ventricular defects smaller than 10 mm in diameter (Della Puppa A et al, World Neurosurgery, in press). The 10-mm cutoff was defined because the BCNU wafer’s diameter is actually 14 mm. Patients affected by large ventricular defects, as in the presented series, have been not considered up to now appropriate for BCNU wafer implantation. This condition was considered a critical issue by the manufacturer and also in the relevant literature (Attenello FJ et al., Ann Surg Oncol, 2008). In this sense, the authors’ experience could even extend in future inclusion criteria for BCNU wafer implantation. Finally, in order to achieve a better management of patients treated with carmustine wafers, we would like to emphasize the importance of additional short-term follow-up MRI imaging after surgery (Della Puppa A et al., Acta Neurochir, 2010).
Domenico d’Avella
Alessandro Della Puppa
Padova, Italy
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Bock, H.C., Cohnen, J., Keric, N. et al. Occlusion of surgical opening of the ventricular system with fibrinogen-coated collagen fleece: a case collection study. Acta Neurochir 153, 533–539 (2011). https://doi.org/10.1007/s00701-010-0923-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-010-0923-z