Abstract
Juniperus thurifera is an important component of woodland communities of dry sites within the West Mediterranean region and is characterised by a strongly disjunctive geographic range. Two subspecies were recognised, subsp. thurifera in Europe and subsp. africana in Africa. The aim of the study was the comparison of phenetic diversity to the pattern of AFLP geographic differentiation of the species described in the literature. The examination of phenetic diversity was based on the biometrical analysis of 17 populations using 12 morphological characters of cone and seed. The differences among populations were analysed using Student’s t test, analysis of discrimination, UPGMA agglomeration and hierarchical analysis of variance. The majority of morphological characters differentiated at a statistically significant level between populations and between J. thurifera subsp. thurifera and subsp. africana. Three groups of populations were detected using multivariate statistical analyses. The first, well separated, is subsp. africana, while the following two concern subsp. thurifera. The morphological differentiation of populations appeared similar to that described on the AFLP. The Gibraltar Straight appeared to be the most important barrier.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Juniperus thurifera L. is a tree-like juniper of the section Sabina (Mill.) Spach. (Farjon 1993, 2005; Adams 2011), occurring in the western Mediterranean region (Fig. 1), where it grows in the continental climate conditions of the mountain systems (Achhal et al. 1980; Fromard and Gauquelin 1993; Romo and Boratyński 2005; Gastón González 2006). The present range of J. thurifera covers several, more or less isolated, areas in the Iberian Peninsula, Pyrenees, Alps and Corsica in Europe; and the Middle, High and Anti-Atlas and Aurès in North Africa (Jalas and Suominen 1973; Barbero et al. 1988; Lathuillière 1994; Gamisans et al. 1994; Gauquelin et al. 1999; Quézel and Médail 2003; Farjon 2005; Romo and Boratyński 2005).
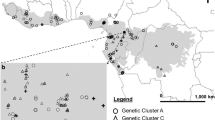
Distribution of Juniperus thurifera (compiled from: Jalas and Suominen 1973; Romo and Boratyński 2005; Gastón González 2006) and location of sampled populations (acronyms as in Table 1): subsp. thurifera: filled triangle Iberian–Corsican group, filled circle Alpine–Iberian Group, filled square subsp. africana: morphological boundaries (a–d) obtained with Monmoniers’s maximum difference algorithm of Euclidean distances among populations revealed by Barrier: a between African and European, b between Corsican and Iberian, c between Iberian and north-Iberian–Alpine, and d between north and southern Middle Atlas populations; the thickness of barriers is related to its importance
The genetic variation of J. thurifera, tested on the random amplified polymorphic DNA (RAPD) by Jiménez et al. (2003), suggests that the species migrated from the European mountain systems southward and westward towards Corsica, the Iberian Peninsula and then the Atlas Mountains in N Africa. The African populations, the most isolated from the others, appeared to be the most distant genetically and morphologically (Adams et al. 2003; Jiménez et al. 2003; Romo and Boratyński 2007; Terrab et al. 2008). The morphological differences between European and African individuals of J. thurifera were the reason for the distinguishing of subsp. africana (Maire) Romo et Boratyński, which differs from J. thurifera subsp. thurifera by its smaller cone diameter, lower number of seeds per cone and shorter leaf scales (Romo and Boratyński 2007). Differences between subsp. thurifera and africana have also been found in the biochemical composition (Gauquelin et al. 1988, 1999; Lebreton 1990; Akkad et al. 2001) and genetic structure (Adams et al. 2001, 2003; Jiménez et al. 2003; Terrab et al. 2008). The latter study revealed strong differences between European and African populations of J. thurifera, and confirmed subsp. africana as a separate taxon. The analysis of European populations showed two different groups, the first including populations from the Alps, Pyrenees, northern part of the Iberian Peninsula and Corsica, and the second overlapping those from the central part of the Iberian Peninsula (Terrab et al. 2008). The biometrical examinations of four European populations (Mazur et al. 2005; Marcysiak et al. 2007) revealed the great differences among them, especially between one from the Alps and three from the Iberian Peninsula (Mazur et al. 2005). Taking into account the last result we hypothesised that J. thurifera’s phenetic diversity pattern should be similar to AFLP geographic differentiation and is mainly a consequence of the history of the species. J. thurifera is well adapted to the dry environments and occurs on the sites inaccessible or only weakly accessible for other trees (Quézel and Médail 2003). The adaptation by selection as an important source of phenotypic differentiation of that species cannot be excluded however, but probably had similar influence in various parts of its geographic range (Terrab et al. 2008).
The aim of the present study was (1) to determine morphological dissimilarities between populations by performing a biometrical comparison of morphological characters of female cones, seeds and shoots, and (2) to evaluate to what extend the multivariate biometrical analysis would resemble the pattern of variation and differentiation described by Jiménez et al. (2003) and Terrab et al. (2008) based on RAPDs and AFLP analyses, respectively.
Materials and methods
Plant material and measuring procedures
The material for the study was based on 17 populations of J. thurifera (Table 1), which cover almost the entire species area of natural distribution (Fig. 1). Populations were represented by 16–36 individuals (27 in average) except for MO_02 (see Table 1), where only eight individuals have been sampled due to lack of cones on more junipers in that locality. The 441 individuals were biometrically examined, each represented by 10 adult cones and 10 young shoots with leaves, all collected from the sunny part of the crown. However, a few individuals were represented by smaller numbers, because of lack of sufficient number of cones or destruction of shoots. The cone scales (CSN) were counted after cone soaking. The seeds were taken off the soaked cones and dried after measuring. The thickness of the ultimate lateral branchlet with leaves (ST) was measured following methods described by Marcysiak et al. (2007). The measurement was performed on dry material. The populations were compared on the basis of six characters of cones and seeds, two of shoots and leaves, and four proportions (Table 2).
Data analysis
All data were standardised before analysis to avoid the possible influence of variation resulting from various types of used characters. The STATISTICA (StatSoft) procedures were performed. The frequency distribution of measured character values was verified using the Shapiro–Wilk’s test to assess the symmetry and unimodality and, consequently, the possibility of conducting the multivariate statistical analyses (Sokal and Rohlf 1997; Zar 1999). The homoscedasticity of data was checked using Brown–Forsythe test as implemented by STATISTICA (StatSoft) to assess the possibility of use of parametric statistic tests (Sokal and Rohlf 1997).
The main statistics such as arithmetic means, standard deviations and variation coefficient were calculated for each population in order to determine the range of their variation. The interactions between characters were checked with Pearson’s correlation coefficient to detect possible redundant variables. A stepwise discrimination analysis was performed to: (1) definitively eliminate the possible redundant variables, (2) identify the discrimination power of particular characters, and (3) determine the intra- and inter-populational variation (Tabachnik and Fidell 2007; Sokal and Rohlf 1997).
The significance of differences between mathematic means of characters for two groups of populations of subsp. thurifera and populations of subspecies africana were verified using t test for independent samples. The Kruskal–Wallis test for the traits with biased distribution was performed to attest the statistical significance of the differences (Sokal and Rohlf 1997; Zar 1999).
Dendrograms of the Euclidean distances of nearest neighbourhood among populations were constructed according to Ward’s method, to verify their affinities and taxonomic relations, revealed in discrimination analysis (Sokal and Rohlf 1997). Dendrograms on the Mahalanobis distances was analysed to examine the similarities among populations and taxa (Sokal and Rohlf 1997). STATISTICA software (StatSoft) was used in all the above-mentioned analyses.
To evaluate the correlation between geographic and morphological multicharacter differences among populations, a Mantel test (Mantel 1967) was performed on the matrices of Euclidean and Mahalanobis distances and the geographic distances (Manni et al. 2004), using GenAlEx software. Geographic distances were counted on the geographic coordinates using Map Info 9.5 (Pitney Bowes). To define the barriers within the network of populations, the Monmonier’s algorithm applied on a Delanaunay triangulation was used, as implemented by software BARRIER 2.2 (Manni et al. 2004).
The hierarchical analysis of variance was performed to determine which characters differentiated between subspecies and between three groups of populations detected by the discrimination analysis (Sokal and Rohlf 1997), as implemented by JMP software (SAS Institute).
Results
Variation and correlation of characters
The characters examined, except for CSN and SN, have a unimodal and normal or very close to normal frequency distribution, which enables us to use multivariate statistical methods of comparison and further analyses. The CSN and SN data were transformed into % frequencies and arcsined before the further, multivariate analyses.
The original data of all characters except CL, CD and CL/CD have a non homoscedastic character, which prevented direct utilisation of parametric analyses, but after standardisation have a homogenous level of variation.
The average values of characters were specific for every population at some degree, but with frequency distributions overlapping in groups of populations (Table 2). The values of variation coefficient of the measured characters differed slightly among three groups of populations, while SN was very variable, showing high differences (Table 2). The most stable appeared to be CL and SL.
Correlations between measured characters were slightly different for each sample of J. thurifera, but generally, the dimensional characters of cones and seeds (CL, CD, SL and SW) positively correlate to each other at a statistically significant level (p ≤ 0.01) in all samples. The SN also positively correlated to CL and CD (p ≤ 0.01). The characters of cones and seeds are correlated at a much lower level with characters of needles and branchlets (LN and ST).
Multivariate differentiation of populations
All characters except those of SW and SL/SW had a statistically significant value for discrimination among populations of J. thurifera (Table 2). The greatest discriminating power had the ratio of CL/CD and LN, with a partial Wilks lambda values of about 0.35 and 0.45, respectively (Table 2). The first three discrimination variables covered 86.3 % of the total variation. On the plane of the two first discrimination variables U 1 and U 2, responsible for about 79 % of the variation, compared populations formed three well-separated agglomerations (Fig. 2A1), visible also on the plane between U 1 and U 3 (Fig. 2A2). The populations from the Middle, High and Anti-Atlas in Africa form the most distinct group, determined by the first variable, which in the greatest degree depends on CSN, SN, ratio CD/SN and CL/CD. The African populations, however, did not form a compact group, but were differentiated along the variables U 2 and U 3 (Fig. 2A3).
Results of discrimination analysis for Juniperus thurifera: a according to the discrimination variables U 1, U 2, U 3 for populations; b for individuals according to discrimination variables U 1 and U 2 (acronyms of populations as in Table 1)
The two remaining groups of populations are also determined in the first place by U 1, and one of them has a compact, the other a dispersed character influenced by U 2 and U 3 (Fig. 2A3). The isolation of four Alpine–Iberian populations (FR_01, SP_02, SP_06 and SP_07), which form a second group on the discrimination scatter plot, is very high. The third group is made up of Iberian–Corsican populations (FR_02, FR_03, SP_01, SP_03-05 and SP_08) and occupies the position between the Moroccan and Alpine–Iberian populations (Fig. 2A1). These two groups differ from each other at a statistically significant level in all respects of except for the CL and SL/SW. Also, both groups of populations representing subsp. thurifera differed from subsp. africana statistically significantly as regards of the whole set of characters except ST, which in Iberian–Corsican populations and African populations was almost the same (Table 2). The Iberian–Corsican populations have mean values of characters predominantly intermediate between subsp. africana and Alpine–Iberian populations (e.g. characters CD, CSN, SL, SW).
The individuals of each particular group of populations hardly penetrated others (Fig. 2b). The proper ordination of individuals into particular populations was nearly 65 % on average, and varied from 37.5 % in population SP_07 to 88.2 % in SP_02. Ordination to each of the three distinguished groups of populations, however, has generally a much higher success rate: 99.3 % for Moroccan, 98.9 % for the second (FR_01, SP_02, SP_06 and SP_07) and 97.8 % for the third (FR_02, FR_03, SP_01, SP_03–05 and SP_08). Such a high level of the fitness of ordination of individuals to the three distinguished groups of populations indicates the very high probability of proper inclusion of practically every single individual (Fig. 2b). The distribution of individuals on the plane of the two first discrimination variables, which account for 100 % of variation, shows that the first of them distinguishes completely the Alpine–Iberian group from all the others. The Central Iberian and Corsican group are distinguished by both discrimination variables; however, the first is responsible for a three times higher percentage of the total variation. The first discrimination variable relies on the full set of variables, but mostly from CSN, SN, CD/SN, CD, SN/CSN, and the second discrimination variable correlated additionally with ST.
On the graph showing results of the cluster analysis on the basis of the closest Euclidean distances, the populations also formed three separate groups. Those separated at the highest level are from Africa, then two subgroups are formed in an identical way to that of the discrimination analysis (Fig. 3). The shortest Mahalanobis distances connected samples directly at a similar level to that found in the cluster on the Euclidean distances and discrimination analyses (data not shown).
Dendrogram constructed by Ward method of cluster analysis on the closest Euclidean distances between samples of Juniperus thurifera (acronyms as in Table 1)
Differences between subspecies and populations in particular characters
The mean values of characters calculated separately from data of European and African individuals, comprehended as representing J. thurifera subsp. thurifera and subsp. africana, respectively, all differed at a statistically significant level (Table 2). In spite of that, the ranges of distribution of particular character values of subsp. thurifera overlapped those of subsp. africana at least partly (Table 2). Even SN, attaining an average value in the Alpine–Iberian populations of 3.71, the Iberian–Corsican 3.22 and in the African ones 1.33, ranged from 1 to 10 and 1 to 4, in Europe and Africa, respectively. Regardless, every African population (J. thurifera subsp. africana) differed from every other one in the mean values of SN as a result of the Tukey’s and Kruskal–Wallis’ tests among groups of populations. The next character, which differed European from most of the African populations, was SW. The European populations have smaller seeds, with SW ranging between 1.3 and 6.8 mm, 3.67 mm on average of Alpine–Iberian and 3.91 mm of Iberian–Corsican populations, while African between 2.1 and 7.6 mm, 4.75 mm on average. Most African populations also differed from European ones by having smaller cones (CL and CD). The northernmost Moroccan ones (MO 1 and MO 2) showed a smaller number of statistically significant differences than observed between other African and European populations (Table 3).
Geographic pattern of variation
The results of the Mantel test conducted on the Euclidean and the Mahalanobis distances among populations indicated statistically significant relationships to the geographic spatial distance. Correlations for Euclidean and Mahalanobis distance are r = 0.523, p < 0.001 and r = 0.411, p < 0.001, respectively. The geographic discontinuities among populations detected in the Barrier 2.2 analysis indicated the high importance of the Gibraltar Straight on species differentiation (Fig. 1a). The second and third barriers separate between the Alpine and NE Iberian populations (FR_1, SP_6 and SP_7), Corsican and E Iberian (FR_2, FR_3 and SP_3) and all the other Iberian ones (Fig. 1b, c). The fourth barrier separates the two northernmost populations sampled in the Middle Atlas from the rest of the analysed subsp. africana populations (Fig. 1d).
The result of the hierarchical analysis of variance showed that the African populations differed from the European at a statistically significant level as regards most characters, as likewise between three groups of populations detected during analyses of discrimination and agglomeration on the Euclidean distances (Table 4). The only exceptions are LN in both cases and ST between European and African populations.
Discussion
Evaluation of characters
Average values of particular characters of cone, seed and sprout with leaves of J. thurifera found during study is generally similar to the data reported earlier (do Amaral Franco 1986; Marcysiak et al. 2007; Romo and Boratyński 2007), but reveal broader ranges of distribution. The marginal values of particular characters found in our study, taking into account the numbers of tested individuals can, with high probability, be treated as real minima and maxima, while data published earlier resulted from only a few measurements on the herbarium materials (do Amaral Franco 1986) or only few characters (Barbero et al. 1994). Such kind of data was also collected and compared for J. thurifera subsp. thurifera and subsp. africana (Romo and Boratyński 2007).
The CL, CD and SN have been recognised as differentiating between J. thurifera subsp. thurifera and subsp. africana (Gauquelin et al. 1988; Lebreton 1990; Romo and Boratyński 2007). We confirmed this, stressing a very high probability of correct distinguishing of herbarium materials of subsp. thurifera from subsp. africana using only CD and SN.
The leaf-scale length, which attains about 2.0–2.5 mm and 1.0–1.5 mm in subsp. thurifera and subsp. africana, respectively, was also recognised as differentiating between them (Romo and Boratyński 2007). However, the latter difference did not correspond well with LN, as found in our study. The average values of LN for subsp. thurifera and subsp. africana were 20.21 and 18.86, respectively. The leaves of J. thurifera are decussate and form four rows, 5.01 and 4.71 leaves of a particular row 5 mm long on the sprout of subsp. thurifera and africana, respectively. Consequently, visible parts of the leaves are 1.01 and 0.94 mm for the apical 5 mm of lateral branchlets of subsp. thurifera and subsp. africana, respectively. These data confirm differences between subspecies but at a much smaller level than shown by Romo and Boratyński (2007) on the basis of data from Flora Iberica for subsp. thurifera (do Amaral Franco 1986). It can result from measurements of different parts of sprouts. We tested only apical parts of the lateral branchlets, while do Amaral Franco (1986) in all probability used other parts of the sprouts, which have larger leaves. The other analysed characters also differentiated significantly between subsp. thurifera and subsp. africana (Table 2).
Geographic structure of morphological differentiation
Morphological differentiation of J. thurifera coincides with Terrab et al. (2008) suggestion, that the Strait of Gibraltar functioned as the principal barrier between European and African populations of the species. These authors pointed out a lack of gene flow between Moroccan and Iberian populations after glacial periods (Terrab et al. 2008). Our result is generally in accordance with the separate taxonomic position of the Moroccan populations (Gauquelin et al. 1988; Barbero et al. 1994; Adams et al. 2003; Jiménez et al. 2003; Romo and Boratyński 2005), treated as subspecies africana (Romo and Boratyński 2007) or ultimately proposed as maroccana (till now nomen nudum) (Vela and Gauquelin 2011). The more recently published results of the studies on the genetic differentiation of conifers across the Strait of Gibraltar confirmed a high level of differences (Terrab et al. 2007; Jaramillo-Correa et al. 2010; Dzialuk et al. 2011), however, some taxa did not follow this rule (Jaramillo-Correa et al. 2010). Species other than coniferous plants also revealed a high level of genetic differences between European and North African populations (Lumaret et al. 2002; Ortiz et al. 2007), but generally several patterns of species differentiation and gene flow across the Strait of Gibraltar have been described and both high and relatively low levels of differences were reported (Rodríguez-Sánchez et al. 2008). The significant differences between European and Moroccan populations of J. thurifera, found during our study in the majority of tested morphological characteristics, indicated that patterns of genetic and morphological variation of this species are convergent.
The biometrical differentiation of European populations of J. thurifera into two groups with an almost majority of well-distinguished individuals (Fig. 2b) only partly follows the pattern of genetic variation revealed by AFLP analysis (Terrab et al. 2008). It can be explained by the origin of part of our material from other populations and other individuals than those used by Terrab et al. (2008). In spite of this, we have to point out some similarities between genetic and morphological patterns of differentiation between populations representing the same regions. The first of them is the different multivariate character of populations from Corsica and the Iberian Peninsula found by Terrab et al. (l.c.) and observed in our study (compare our Figs. 1, 2a, 3 with Terrab et al. 2008: Figs. 1, 2, 3). Terrab et al. (2008) explained this type of connection between populations as resulting from the early Holocene migration from the Iberian Peninsula to Corsica, similarly to the case of several other plant species. The rejection of an origin of the Corsican population from much more remote times, when the microplate of contemporary Corsica and Sardinia separated from the Iberian microplate in the early Oligocene (Bellon et al. 1977; Rosenbaum et al. 2002) is supported by the low divergence between Corsican and Iberian populations. The divergence time of J. thurifera determined as before 10 Mya (Mao et al. 2010) also indicates the most recent migration between the Iberian Peninsula and Corsica.
The second example of similarity of our results and AFLP differentiation concerned Alpine and north Iberian populations. Unfortunately, we examined only one population from the Alps, but its biometric characteristics placed it into the clade of north- and central-Iberian populations (Fig. 3).
The reasons for detected morphological differentiation are, as also in the case of genetic variation, the history and isolation of particular populations in the fragmented habitats (Saunders et al. 1991; Lindenmayer and Fischer 2006). The success of gene flow between J. thurifera stands by pollen and seed was strongly reduced with increased distance (Rhanem 2010; Olano et al. 2012). The fragmentation of habitats suitable for J. thurifera (Gauquelin et al. 2003; Romo and Boratyński 2005; Gastón González 2006; DeSoto et al. 2010; Rhanem 2010; Olano et al. 2012) has altered not only genetic structure, but also phenotypic variation of the species (Santos et al. 1999; compare e.g. Douaihy et al. 2011, 2012). The pattern of morphological differentiation of compared populations can be interpreted as support for the hypothesis of migration of the species (or its ancestor) from the North and colonisation of the Iberian Peninsula and Africa during Miocene climate cooling (Barbero et al. 1994; Jiménez et al. 2003). However, the hypothesis that Iberian populations were divided into two morphologically different types, which then settled in the Alps and Corsica (Terrab et al. 2008) cannot be decisively excluded without more detailed investigation.
Conclusion
The morphological polymorphism of the compared populations of J. thurifera supports the subspecific taxonomic position of Moroccan populations of the species. The geographic differentiation of the morphological characteristics resembles those received with the AFLP and RAPDs (Jiménez et al. 2003; Terrab et al. 2008). The most eminent differences between European populations from the Western Alps and the Ebro Basin with its northern peripheries on the one side, and from Corsica and the central part of the Iberian Peninsula, on the other side, support another geographic origin, suggested by Terrab et al. (2008). The strong geographic differentiation between the two continents and among populations within Europe and Africa suggest rather local management of the J. thurifera woodlands, without seed exchange between the separated parts of the species’ geographic range.
References
Achhal A, Akabli O, Barbero M, Benabid A, M’hint A, Peyre C, Quézel P, Rivas-Martínez S (1980) À propos de la valeur bioclimatique et dynamique de quelques essences forestières au Maroc. Ecol Mediterr 5:211–249
Adams RP (2011) Junipers of the world: the genus Juniperus, 3rd edn. Trafford Publishing Co., Bloomington
Adams RP, Pandey RN, Gauquelin T, BadriW (2001) Systematics of Juniperus thurifera using DNA fingerprinting and leaf essential oils: comparison of the Moroccan J. thurifera with populations from Spain and France. 2eme Colloque International: Le genévrier thurifère et les forets d’altitude dans les montagnes du partout mediterraneen, Marrakech, Livre des résumés
Adams RP, Mumba LE, James SA, Pandey RN, Gauquelin T, Badri W (2003) Geographic variation in the leaf essential oils and DNA fingerprints (RAPDs) of Juniperus thurifera L. from Morocco and Europe. J Essential Oil Res 15:148–154
Akkad S, Akssira F, Mellouki F, Barrero AF, Quilez del Moral J, Arteaga P, Herrador MM, Belgarrab A (2001) Étude de la composition des huiles de Juniperus thurifera L. var. africana a l’aide du couplage CG-MS. 2eme Colloque International: Le genévrier thurifère et les forêts d’altitude dans les montagnes du partout méditerranéen, Marrakech, Livre des résumés
Barbero M, Hammoud A, Quézel P (1988) Sur la découverte dans les Alpes maritimes italiennes du Genévrier thurifère (Juniperus thurifera L.). Webbia 42:49–55
Barbero M, Lebreton Ph, Quézel P (1994) Sur les affinités biosystématiques et phytoécologiques de Juniperus thurifera L. et de Juniperus excelsa Bieb. Ecol Medit 20(3/4):21–37
Bellon H, Coulon C, Edel JB (1977) Le déplacement de la Sardaigne. Synthèse des données géochronologiques, magnétiques et paléomagnétiques. Bull Soc Géol Fr 19:825–831
DeSoto L, Olano JM, Rozas V, De la Cruz M (2010) Release of Juniperus thurifera woodlands from herbivore-mediated arrested succession in Spain. Appl Veg Sci 13:15–25
do Amaral Franco J (1986) Juniperus L. In: Castroviejo S, Laínz M, López González G, Montserrat P, Muñoz Garmendia F, Paiva J, Villar L (eds) Flora iberica, vol 1. Real Jardín Botánico, C.S.I.C, Madrid, pp 181–188
Douaihy B, Vendramin GG, Boratyński A, Machon N, Bou Dagher-Kharrat M. 2011. High genetic diversity with moderate differentiation in Juniperus excelsa from Lebanon and the eastern Mediterranean region. AoB PLANTS 2011 plr003 doi:10.1093/aobpla/plr003
Douaihy B, Sobierajska K, Jasińska AK, Boratyńska K, Ok T, Romo A, Machon N, Didukh Y, Bou Dagher-Kharrat M, Boratyński A. 2012. Morphological versus molecular markers to describe variability in Juniperus excelsa subsp. excelsa (Cupressaceae). AoB PLANTS 2012: pls013; doi:10.1093/aobpla/pls013
Dzialuk A, Mazur M, Boratyńska K, Montserrat JM, Romo A, Boratyński A (2011) Population genetic structure of Juniperus phoenicea (Cupressaceae) in the western Mediterranean Basin: gradient of diversity on a broad geographical scale. Ann For Sci 68:1341–1350
Farjon A (1993) The taxonomy of multiseed Junipers (Juniperus sect. Sabina) in Southwest Asia and East Africa (Taxonomic notes on Cupressaceae I). Edinburgh J Bot 49:251–283
Farjon A (2005) A monograph of Cupressaceae and Sciadopitys. Royal Botanic Gardens, Kew
Fromard F, Gauquelin T (1993) Les formations à genévrier thurifère des montagnes Marocaines: actions de recherche et de conservation sur un milieu et une espèce en régression. Unasylva 44:52–58
Gamisans J, Jeanmonod D, Regato P, Gruber M (1994) Le Genévrier thurifère (Juniperus thurifera L.) en Corse. Candollea 49:600–607
Gastón González AG (2006) Influencia del uso del suelo en la distribución de la sabina albar (Juniperus thurifera L.) en la Península Ibérica. III Coloquio Internacional sobre los Sabinares y Enebrales (Género Juniperus): Ecología y Gestión Forestal Sostenible, Soria
Gauquelin T, Hassani MI, Lebreton P (1988) Le genévrier thurifère, Juniperus thurifera L. (Cupressacees): analyse biométrique et biochimique; propositions systématiques. Ecol Medit 14:31–42
Gauquelin T, Bertaudiere V, Montes N, Badri W, Asmode JF (1999) Endangered stands of thuriferous juniper in the western Mediterranean basin: ecological status, conservation and management. Biodivers Conserv 8:1479–1498
Gauquelin T, Bertaudière T, Cambecèdes J, Largier G (2003) Le genévrier thurifère (Juniperus thurifera l.) dans les Pyrénées: état de conservation et perspectives. Acta Bot Barc 49:83–94
Jalas J, Suominen J (1973) Atlas Florae Europaeae, 1. The Committee for Mapping the Flora of Europe and Societatis Biologica Fennica Vanamo, Helsinki
Jaramillo-Correa JP, Grivet D, Terrab A, Kurt Y, De-Lucas AI, Wahid N, Vendramin GG, González-Martínez SC (2010) The Strait of Gibraltar as a major biogeographic barrier in Mediterranean conifers: a comparative phylogeographic survey. Mol Ecol 19:5452–5468
Jiménez JF, Werner O, Sánchez-Gómez P, Fernández S, Guerra J (2003) Genetic variation and migration pathway of Juniperus thurifera L. (Cupressaceae) in the western Mediterranean region. Israel J Plant Sci 51:11–22
Lathuillière L (1994) Le Genévrier thurifère: monographie et étude des différentes stations des Alps. Mémoire de la F.I.F., Nancy et Conservatoire Botanique, Gap-Charance
Lebreton P (1990) La chimiotaxonomie des Gymnospermes. Bull Soc Bot Fr 137(1):35–46
Lindenmayer DB, Fischer J (2006) Habitat fragmentation and landscape change: an ecological and conservation synthesis. Island Press, Washington
Lumaret R, Mir C, Michaud H, Raynal V (2002) Phylogeographical variation of chloroplast DNA in holm oak (Quercus ilex L.). Mol Ecol 11:2327–2336
Manni F, Guérard E, Heyer E (2004) Geographic patterns of (genetic, morphologic, linguistic) variation: how barriers can be detected by “Monmonier’s algorithm”. Human Biol 76:173–190
Mantel N (1967) The detection of disease clustering and a generalized regression approach. Cancer Res 27:209–220
Mao K, Hao G, Liu J, Adams RP, Milne RI (2010) Diversification and biogeography of Juniperus (Cupressaceae): variable diversification rates and multiple intercontinental dispersals. New Phytol 188:254–272
Marcysiak K, Mazur M, Romo A, Montserrat JM, Didukh Ya, Boratyńska K, Jasińska A, Kosiński P, Boratyński A (2007) Numerical taxonomy of Juniperus thurifera, J. excelsa and J. foetidissima (Cupressaceae) based on morphological characters. Bot J Linn Soc 155:483–495
Mazur M, Marcysiak K, Boratyński A, Boratyńska K, Romo A, Montserrat JM, Kosiński P (2005) Morphological variation of Juniperus thurifera L. (Cupressaceae) from four populations in South-Western Europe. In: Prus-Głowacki W, Pawlaczyk EM (eds) Variability and evolution—new perspectives, vol 72., Ser BiologiaAdam Mickiewicz University, Poznań, pp 399–409
Olano J, Zavala M, Rozas V (2012) Disruption of Juniperus thurifera woodland structure in its northwestern geographical range: potential drivers and limiting factors. Eur J For Res 131:563–570
Ortiz MÁ, Tremetsberger K, Talavera S, Stuessy T, García-Castaño JL (2007) Population structure of Hypochaeris salzmanniana DC. (Asteraceae), an endemic species to the Atlantic coast on both sides of the Strait of Gibraltar, in relation to quaternary sea level changes. Mol Ecol 16:541–552
Quézel P, Médail F (2003) Écologie et biogéographie des forêts du basin méditerranéen. Elsevier, Paris
Rhanem M (2010) Approche de la conservation et de la restauration du genévrier thurifère (Juniperus thurifera L.) au Maroc à travers l’exemple de la vallée des Aït-Bou-Guemmez (Haut Atlas). Bull Soc Bot Centre-Ouest, Nouv Série 41:99–138
Rodríguez-Sánchez F, Pérez-Barrales R, Ojeda F, Vargas P, Arroyo J (2008) The Strait of Gibraltar as a melting pot for plant biodiversity. Quat Sci Rev 27:2100–2117
Romo A, Boratyński A (2005) Chorology of Juniperus thurifera (Cupressaceae) in Morocco. Dendrobiol 54:41–50
Romo A, Boratyński A (2007) Nomenclatural note on Juniperus thurifera subsp. africana (Cupressaceae). Ann Bot Fenn 44:72–75
Rosenbaum G, Lister GS, Duboz C (2002) Reconstruction of the tectonic evolution of the Western Mediterranean since the Oligocene. J Virt Explor 8:107–130
Santos T, Tellería JL, Virgós E (1999) Dispersal of Spanish juniper Juniperus thurifera by birds and mammals in a fragmented landscape. Ecography 22:193–204
Saunders DA, Hobbs RJ, Margules CR (1991) Biological consequences of ecosystem fragmentation: a review. Conserv Biol 5:18–32
Sokal RR, Rohlf TJ (1997) Biometry: the principles and practice of statistics in biological research. Freeman W H & Comp, San Francisco
Tabachnik BG, Fidell LS (2007) Using multivariate statistics, 5th edn. California State University, Northridge
Terrab A, Talavera S, Arista M, Paun O, Stuessy TF, Tremetsberger K (2007) Genetic diversity and geographic structure at chloroplast microsatellites (cpSSRs) in endangered West Mediterranean firs (Abies spp., Pinaceae). Taxon 56:409–416
Terrab A, Schoenswetter P, Talavera S, Vela E, Stuessy TF (2008) Range-wide phylogeography of Juniperus thurifera L., a presumptive keystone species of western Mediterranean vegetation during cold stages of the Pleistocene. Mol Phylogenet Evol 48:94–102
Vela E, Gauquelin T (2011) Juniperus thurifera subsp. maroccana, subsp. nova, a new taxon to formally describe the maroccan Thuriferous juniper. IVème Colloque international sur le Genévrier thurifère et espèces affines, Mont-Dauphin and Saint Crépin, 2011, 88 pp
Zar JH (1999) Biostatistical analysis. Prenice-Hall, New Jersey
Acknowledgments
We would like to thank Amelia Lewandowska for technical help, Samuel Pyke and Proof-Reading Service for linguistic verification of the previous version of the manuscript. Funding the work was partly sponsored by the Institute of Dendrology of the Polish Academy of Sciences. The collection of material was made possible due to the bilateral cooperation of the Polish Academy of Sciences with the Spanish National Research Council “Consejo Superior de Investigaciones Científicas”.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Boratyński, A., Jasińska, A.K., Marcysiak, K. et al. Morphological differentiation supports the genetic pattern of the geographic structure of Juniperus thurifera (Cupressaceae). Plant Syst Evol 299, 773–784 (2013). https://doi.org/10.1007/s00606-013-0760-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00606-013-0760-7