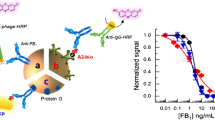
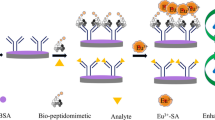
Abstract
Peptidomimetic and anti-immunocomplex peptides can be easily isolated from phage display libraries, and can be used as alternatives to chemical competing haptens to improve the sensitivity of small molecule immunoassay. In this work, 16 peptidomimetics and 7 anti-immunocomplex peptides of pendimethalin (PND) were obtained from cyclic 7-, 8-, 9-, and 10-residue peptide phage libraries. Peptidomimetic EJ-2 (CMFTGTDFPC) with the highest sensitivity in competitive phage enzyme-linked immunosorbent assay (ELISA) and immunocomplex peptide EF-30 (CNPGWPPIPC) with the highest sensitivity in noncompetitive phage ELISA were selected to prepare phage-free peptides with GGGSSK-biotin at the C-terminus. Competitive and noncompetitive lateral flow immunoassays (CLFIA and NLFIA) were developed by using the phage-free peptides. After optimization, the CLFIA and NLFIA showed visual limit of detections (vLODs) of 5 ng/mL and 2.5 ng/mL, respectively, which were improved two- and fourfold compared with a LFIA based on chemical hapten. The NLFIA showed better sensitivity than CLFIA in the detection of spiked samples, and can meet the detection requirements for agro-products regulated by EU and China. The detection results of CLFIA and NLFIA for blind samples were consistent with that of ultra performance liquid chromatography/tandem mass spectrometry.
Graphical abstract




Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Grant BD, Anderson CE, Williford JR, Alonzo LF, Glukhova VA, Boyle DS, Weigl BH, Nichols KP (2020) SARS-CoV-2 coronavirus nucleocapsid antigen-detecting half-strip lateral flow assay toward the development of point of care tests using commercially available reagents. Anal Chem 92(16):11305–11309. https://doi.org/10.1021/acs.analchem.0c01975
Cai XF, Ma F, Jiang J, Yang XL, Zhang ZW, Jian ZL, Liang MJ, Li PW, Yu L (2023) Fe-N-C single-atom nanozyme for ultrasensitive, on-site and multiplex detection of mycotoxins using lateral flow immunoassay. J Hazard Mater 441. https://doi.org/10.1016/j.jhazmat.2022.129853
Xu ZH, Wang JK, Ye QX, Jiang LF, Deng H, Liang JF, Deng H, Liang JF, Chen RX, Huang W, Lei HT, Xu ZL, Luo L (2023) Highly selective monoclonal antibody-based fluorescence immunochromatographic assay for the detection of fenpropathrin in vegetable and fruit samples. Anal Chim Acta 1246:340898–340898. https://doi.org/10.1016/j.aca.2023.340898
Zhang Y, Yang HJ, Xu Z, Liu X, Zhou J, Qu XF, Wang WL, Feng YW, Peng CF (2023) An ultra-sensitive photothermal lateral flow immunoassay for 17-estradiol in food samples. Food Chem 404. https://doi.org/10.1016/j.foodchem.2022.134482
Bai YC., Wang YH, Li Q, Dou LN, Liu MG, Shao SB, Zhu JY, Shen JZ, Wang ZH, Wen K, Yu WB (2021) Binding affinity-guided design of a highly sensitive noncompetitive immunoassay for small molecule detection. Food Chem 351. https://doi.org/10.1016/j.foodchem.2021.129270
Dang M, Li ZZ, Mao YX, Huang XQ, Song LJ, Li WF, Ma RX, Liu Y, Wang LY, Yu XZ, Yang HJ, Zhang XY (2023) A highly sensitive lateral flow immunoassay based on a group-specific monoclonal antibody and amorphous carbon nanoparticles for detection of sulfonamides in milk. Microchim Acta 190:186. https://doi.org/10.1007/s00604-023-05766-3
Liu YH, Xie R, Guo YR, Zhu GN, Tang FB (2012) Comparison of homologous and heterologous formats in nanocolloidal gold-based immunoassays for parathion residue determination. J Environ Sci Health B 47(5):475–483. https://doi.org/10.1080/03601234.2012.663613
Yan JX, Hu WJ, You KH, Ma ZE, Xu Y, Li YP, He QH (2020) Biosynthetic mycotoxin conjugate mimetics-mediated green strategy for multiplex mycotoxin immunochromatographic assay. J Agric Food Chem 68(7):2193–2200. https://doi.org/10.1021/acs.jafc.9b06383
Zhao F, Shi R, Liu R, Tian Y, Yang Z (2021) Application of phage-displayed developed antibody and antigen substitutes in immunoassays for small molecule contaminants analysis: a mini-review. Food Chem 339. https://doi.org/10.1016/j.foodchem.2020.128084
Peltomaa R, Fikacek S, Benito-Pena E, Benito-Pena E, Head T, Deo S, Daunert S, Moreno-Bondi MC (2020) Bioluminescent detection of zearalenone using recombinant peptidomimetic Gaussia luciferase fusion protein. Microchim Acta 187(10):547. https://doi.org/10.1007/s00604-020-04538-7
Fu HJ, Chen ZJ, Wang H, Luo L, Wang Y, Huang RM, Xu ZL, Hammock BD (2021) Development of a sensitive non-competitive immunoassay via immunocomplex binding peptide for the determination of ethyl carbamate in wine samples. J Hazard Mater 406. https://doi.org/10.1016/j.jhazmat.2020.124288
Ishikawa E, Hashida S, Kohno T (1991) Immunoassay Reviewed with Emphasis on Factors Which Limit the Sensitivity. Mol Cell Probes 5(12):81–95. https://doi.org/10.1016/0890-8508(91)90001-Z
Rossotti MA, Carlomagno M, González-Techera A, Hammock BD, Last J, González-Sapienza G (2010) Phage anti-immunocomplex assay for clomazone: two-site recognition increasing assay specificity and facilitating adaptation into an on-site format. Anal Chem 82(21):8838–8843. https://doi.org/10.1021/ac101476f
Cardozo S, González-Techera A, Last JA, Hammock BD, Kramer K, González-Sapienza GG (2015) Analyte peptidomimetics selected from phage display peptide libraries: a systematic strategy for the development of environmental Immunoassays. Environ Sci Technol 39(11):4234–4241. https://doi.org/10.1021/es047931l
Vanrell L, Gonzalez-Techera A, Hammock BD, Gonzalez-Sapienza G (2013) Nanopeptamers for the development of small-analyte lateral flow tests with a positive readout. Anal Chem 85(2):1177–1182. https://doi.org/10.1021/ac3031114
Yeh CY, Hsiao JK, Wang YP, Lan CH, Wu HC (2016) Peptide-conjugated nanoparticles for targeted imaging and therapy of prostate cancer. Biomaterials 99:1–15. https://doi.org/10.1016/j.biomaterials.2016.05.015
Lassabe G, Rossotti M, Gonzalez-Techera A, Gonzalez-Sapienza G (2014) Recombinant streptavidin nanopeptamer anti-immunocomplex assay for noncompetitive detection of small analytes. Anal Chem 86:5541–5546. https://doi.org/10.1021/ac503130v
Jensen KJ (2013) Solid-phase peptide synthesis: an introduction. In: Jensen KJ, Shelton PT, Pedersen SL (eds) Peptide synthesis and applications, 2nd edn. Humana Press, Totewa, pp 1–21
Peltomaa R, Agudo-Maestro I, Mas V, Barderas R, Benito-Pena E, Moreno-Bondi MC (2019) Development and comparison of mimotope-based immunoassays for the analysis of fumonisin B-1. Anal Bioanal Chem 411(26):6801–6811. https://doi.org/10.1007/s00216-019-02068-7
Mohamed IA, Abdalla RM (2023) Weed control, growth, and yield of tomato after application of metribuzin and different pendimethalin products in upper Egypt. J Soil Sci Plant Nutr. https://doi.org/10.1007/s42729-022-01093-3
Hou LF, Lee WJ, Rusiecki J, Hoppin JA, Blair A, Bonner MR, Lubin JH, Samanic C, Sandler DP, Dosemeci M, Alavanja MCR (2006) Pendimethalin exposure and cancer incidence among pesticide applicators. Epidemiology 17(3):302–307. https://doi.org/10.1097/01.ede.0000201398.82658.50
Saquib Q, Siddiqui MA, Ansari SM, Alwathnani HA, Musarrat J, Al-Khedhairy AA (2021) Cytotoxicity and genotoxicity of methomyl, carbaryl, metalaxyl, and pendimethalin in human umbilical vein endothelial cells. J Appl Toxicol 41(5):832–846. https://doi.org/10.1002/jat.4139
US Environmental Protection Agency. R.E.D. Facts: pendimethalin. https://www3.epa.gov/pesticides/chem_search/reg_actions/reregistration/fs_PC-108501_1-Jun-97.pdf. Accessed 1 Nov 2023
Huang LR, Chen H, Cui PP, Ding Y, Wang MH, Hua XD (2022) Development of immunoassay based on rational hapten design for sensitive detection of pendimethalin in environment. Sci Total Environ 830. https://doi.org/10.1016/j.scitotenv.2022.154690
You TY, Ding Y, Chen H, Song GY, Huang LR, Wang MH, Hua XD (2022) Development of competitive and noncompetitive immunoassays for clothianidin with high sensitivity and specificity using phage-displayed peptides. J Hazard Mater 425:128011. https://doi.org/10.1016/j.jhazmat.2021.128011
Wang J, Liu ZP, Li GQ, Li J, Kim HJ, Shelver WL, Li QX, Xu T (2013) Simultaneous development of both competitive and noncompetitive immunoassays for 2,2′,4,4′-tetrabromodiphenyl ether using phage-displayed peptides. Anal Bioanal Chem 405(29):9579–9583. https://doi.org/10.1007/s00216-013-7364-5
Zheng JS, Tang S, Qi YK, Wang ZP, Liu L (2013) Chemical synthesis of proteins using peptide hydrazides as thioester surrogates. Nat Protoc 8(12):2483–2495. https://doi.org/10.1038/nprot.2013.152
Ding Y, Huang Y, Sun WL, Li FX, Zhang SG, Fang S, Wang MH, Hua XD (2022) Carboxyl retained hapten of quinclorac produces highly sensitive antibody and immunochromatographic assay. Microchem J 183:107982. https://doi.org/10.1016/j.microc.2022.107982
Sompunga P, Pruksametanan N, Rangnoi K, Choowongkomon K, Yamabhai M (2019) Generation of human and rabbit recombinant antibodies for the detection of Zearalenone by phage display antibody technology. 201:397–4053. https://doi.org/10.1016/j.talanta.2019.04.034
Liu J, Wu AH, Song SS, Xu LG, Liu LQ, Xu CL, Kuang H (2022) Development of an immunochromatographic assay for the rapid screening of pendimethalin in potato and apple. Microchem J 182. https://doi.org/10.1016/j.microc.2022.107862
Huo JQ, Barnych B, Li ZF, Wan DB, Li DY, Vasylieva N, Knezevic SZ, Osipitan OA, Scott JE, Zhang JL, Hammock BD (2019) Hapten synthesis, antibody development, and a highly sensitive indirect competitive chemiluminescent enzyme immunoassay for detection of dicamba. J Agric Food Chem 67(20):5711–5719. https://doi.org/10.1021/acs.jafc.8b07134
Joint FAO/WHO Meeting on Pesticide Residues (2019) Pesticide residues in food 2019 – Evaluation 2019 Part I – Residues. 349–258. https://www.fao.org/fileadmin/user_upload/IPM_Pesticide/JMPR/Evaluations/2019_Extra/PENDIMETHALIN_292.pdf. Accessed 1 Nov 2023
Nardo FD, Anfossi L, Ozella L, Saccani A, Giovannoli C, Spano G, Baggiani C (2016) Validation of a qualitative immunochromatographic test for the noninvasive assessment of stress in dogs. J Chromatogr B 1028(15):192–198. https://doi.org/10.1016/j.jchromb.2016.06.019
Krotzky AJ, Zeeh B (1996) Pesticides report .33. Immunoassays for residue analysis of agrochemicals: proposed guidelines for precision, standardization and quality control. Pure Appl Chem 67(12):2065–2088. https://doi.org/10.1351/pac199567122065
Commission Regulation (EU) 2023/1049 of 30 May 2023 amending Annexes II and IV to Regulation (EC) No 396/2005 of the European Parliament and of the Council as regards maximum residue levels for fish oil, pendimethalin, sheep fat and spirotetramat in or on certain products (Text with EEA relevance). http://data.europa.eu/eli/reg/2023/1049/oj. Accessed 1 Nov 2023
Ministry of Agriculture and Rural Affairs of the People’s Republic of China, 2021. National food safety standard—maximum residue limits for pesticides in food. GB 2763–2021. http://2763.foodmate.net/pesticides.html. Accessed 1 Nov 2023
Funding
This work was supported by Jiangsu Agriculture Science and Technology Innovation Fund (Grant No. CX(22)3075) and Inner Mongolia Science & Technology Plan [Grant No. 2022YFSH0050].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, L., Wang, G., Wu, Y. et al. Development of competitive and noncompetitive lateral flow immunoassays for pendimethalin using synthetic peptides. Microchim Acta 191, 68 (2024). https://doi.org/10.1007/s00604-023-06151-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-023-06151-w