Abstract
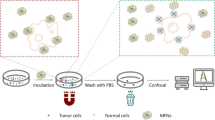
An integrated aptamer macroarray functionalized with reduced graphene oxide (rGO) to specifically capture and sensitively detect cancer cells is reported. The capture for cancer cells is based on effective recognition of the modified rGO surface through the aptamer against epithelial cell adhesion molecule (EpCAM). The rough structure of rGO enhances morphologic interactions between rGO film interface and the cancer cells, while super-hydrophilicity of modified rGO hinders nonspecific cell capture. The synergistic interactions offer the aptamer macroarray high efficiency of cancer cell capture. By means of a turn-on fluorescence strategy based on the conformation change of the aptamer induced by the target recognition, the enriched cancer cells can be directly read out at excitation/emission wavelengths of 550/680 nm without washing, separation, and dying steps. The working range is 1 × 102 to 2 × 104 cells per mL with a detection limit of 22 cells per mL. The results indicate that the aptamer macroarray has a considerable foreground for early diagnosis, therapy, and monitoring of cancer.

Graphical abstract





Similar content being viewed by others
References
Neil S (2011) Early detection: spotting the first signs. Nature 471(7339):14
Tan W, Donovan MJ, Jiang J (2013) Aptamers from cell-based selection for bioanalytical applications. Chem Rev 113(4):2842–2862
Svea P, José María A, Alexandre S, Portia D, Maurice G, Aranzazu C (2008) Phototriggering of cell adhesion by caged cyclic RGD peptides. Angew Chem 47(17):3192–3195
Christopher JB, Langer R, Jeffrey TB (2009) Engineering substrate topography at the micro- and nanoscale to control cell function. Angew Chem Int Ed 48(30):5406–5415
Sekine J, Luo SC, Wang S, Zhu B, Tseng HR, Yu HH (2011) Functionalized conducting polymer nanodots for enhanced cell capturing: the synergistic effect of capture agents and nanostructures. Adv Mater 23:4788–4792
Li Y, Lu Q, Liu H, Wang J, Zhang P, Liang H, Jiang L, Wang S (2015) Antibody-modified reduced graphene oxide films with extreme sensitivity to circulating tumor cells. Adv Mater 27(43):6848–6854
Zhang P, Chen L, Xu T, Liu H, Liu X, Meng J, Yang G, Jiang L, Wang S (2013) Programmable fractal nanostructured interfaces for specific recognition and electrochemical release of cancer cells. Adv Mater 25(26):3566–3570
Cai S, Chen M, Liu M, He W, Liu Z, Wu D (2016) A signal amplification electrochemical aptasensor for the detection of breast cancer cell via free-running DNA walker. Biosens Bioelectron 85:184–189
Shi HW, Zhao W, Liu Z, Liu XC, Xu JJ, Chen HY (2016) A temporal sensing platform based on bipolar electrode for the ultra-sensitive detection of cancer cell. Anal Chem 88:8795–8801
Medley CD, Bamrungsap S, Tan W, Smith JE (2011) Aptamer-conjugated nanoparticles for cancer cell detection. Anal Chem 83(3):727–734
Kim MG, Shon Y, Lee J, Byun Y, Choi BS, Kim YB, Oh YK (2014) Double stranded aptamer-anchored reduced graphene oxide as target-specific nano detector. Biomaterials 35(9):2999–3004
Yu T, Dai PP, Xu JJ, Chen HY (2016) Highly sensitive colorimetric cancer cell detection based on dual signal amplification. ACS Appl Mater Interfaces 8(7):4434–4441
Song Y, Chen Y, Feng L, Ren J, Qu X (2011) Selective and quantitative cancer cell detection using target-directed functionalized graphene and its synergetic peroxidase-like activity. Chem Commun 47(15):4436–4438
Le NDB, Yesilbag Tonga G, Mout R, Kim ST, Wille ME, Rana S, Dunphy KA, Jerry DJ, Yazdani M (2017) Cancer cell discrimination using host-guest “doubled” arrays. J Am Chem Soc 139:8008–8012
An L, Wang G, Han Y, Li T, Jin P, Liu S (2018) Electrochemical biosensor for cancer cell detection based on a surface 3D micro-array. Lab Chip 18:335–342
Subramanian N, Sreemanthula JB, Balaji B, Kanwar JR, Biswas J, Krishnakumar S (2014) A strain-promoted alkyne–azide cycloaddition (SPAAC) reaction of a novel EpCAM aptamer–fluorescent conjugate for imaging of cancer cells. Chem Commun 50(80):11810–11813
Meunier A, Hernández-Castro JA, Turner K, Li K, Veres T, Juncker D (2016) Combination of mechanical and molecular filtration for enhanced enrichment of circulating tumor cells. Anal Chem 88:8510–8517
Guo S, Xu J, Xie M, Huang W, Zhou X (2016) Degradable zinc phosphate-based hierarchical nanosubstrates for capture and release of circulating tumor cells. ACS Appl Mater Interfaces 8(25):15917–15925
Gu Y, Ju C, Li Y, Shang Z, Wu Y, Jia Y, Niu Y (2015) Detection of circulating tumor cells in prostate cancer based on carboxylated graphene oxide modified light addressable potentiometric sensor. Biosens Bioelectron 66:24–31
Park MH, Reátegui E, Li W, Tessier SN, Wong KH, Jensen AE, Thapar V, Ting D, Toner M (2017) Enhanced isolation and release of circulating tumor cells using nanoparticle binding and ligand exchange in a microfluidic chip. J Am Chem Soc 139(7):2741–2749
Zhao L, Lu YT, Li F, Wu K, Hou S, Yu J, Shen Q, Wu D, Song M, OuYang WH, Luo Z (2013) High-purity prostate circulating tumor cell isolation by a polymer nanofiber-embedded microchip for whole exome sequencing. Adv Mater 25(21):2897–2902
Yin J, He X, Wang K, Xu F, Shangguan J, He D, Shi H (2013) Label-free and turn-on aptamer strategy for cancer cells detection based on a DNA–silver nanocluster fluorescence upon recognition-induced hybridization. Anal Chem 85(24):12011–12019
Wu LL, Wen CY, Hu J, Tang M, Qi CB, Li N, Liu C, Chen L, Pang DW, Zhang ZL (2017) Nanosphere-based one-step strategy for efficient and nondestructive detection of circulating tumor cells. Biosens Bioelectron 94:219–226
Ma L, Yang G, Wang N, Zhang PC, Guo FY, Meng JX, Zhang FL, Hu ZJ, Wang ST, Zhao Y (2015) Trap effect of three-dimensional fibers network for high efficient cancer-cell capture. Advanced Healthcare Materials 4(6):838–843
Zhu D, Yang RX, Tang YP, Li W, Miao ZY, Hu Y, Chen J, Yu S, Wang J, Xu CY (2016) Robust nanoplasmonic substrates for aptamer macroarrays with single-step detection of PDGF-BB. Biosens Bioelectron 85:429–436
Fang XH, Tan WH (2010) Aptamers generated from cell-SELEX for molecular medicine: a chemical biology approach. Acc Chem Res 43:48–57
Dong Y, Zhang T, Lin XY, Feng JT, Luo F, Gao H, Wu YP, Deng RJ, He Q (2020) Graphene/aptamer probes for small molecule detection: from in vitro test to in situ imaging. Microchim Acta 187:179
Pei SF, Cheng HM (2012) The reduction of graphene oxide. Carbon 50(9):3210–3228
Wu CC, Li P, Fan NN, Han JJ, Zhang W, Zhang W, Tang B (2019) A dual-targeting functionalized graphene film for rapid and highly sensitive fluorescence imaging detection of hepatocellular carcinoma circulating tumor cells. ACS Appl Mater Interfaces 11(48):44999–45006
Guo HL, Peng M, Zhu ZM, Sun LN (2013) Preparation of reduced graphene oxide by infrared irradiation induced photothermal reduction. Nanoscale 5(19):9040–9048
Mannoor MS, Tao H, Clayton JD, Sengupta A, Kaplan DL, Naik RR, Verma N, Omenetto FG, McAlpine MC (2012) Graphene-based wireless bacteria detection on tooth enamel. Nat Commun 3(3):763
Liu Z, Robinson JT, Sun XM, Dai HJ (2008) PEGylated nano-graphene oxide for delivery of water insoluble cancer drugs. J Am Chem Soc 130(33):10876–10877
Shen HW, Yang J, Chen ZP, Chen XP, Wang L, Hu J, Ji FH, Xie GM, Feng WL (2016) A novel label-free and reusable electrochemical cytosensor for highly sensitive detection and specific collection of CTCs. Biosens Bioelectron 81:495–502
Stolpe A, Pantel K, Sleijfer S, Terstappen LW, Toonder JMJ (2011) Circulating tumor cell isolation and diagnostics: toward routine clinical use. Cancer Res 71(18):5955–5960
Bu JY, Kim YJ, Kang YT, Lee TH, Kim JS, Cho YH, Han SW (2017) Polyester fabric sheet layers functionalized with graphene oxide for sensitive isolation of circulating tumor cells. Biomaterials. 125:1–11
Funding
We thank the financial support of the National Natural Science Foundation of China (Key Program: 81930117; General Program: 81573388, 81573910). This work was also supported by “Six talent peaks project of Jiangsu Province (YY-032).” This work was also supported by the Open Project Program of Jiangsu Key Laboratory for Pharmacology and Safety Evaluation of Chinese Materia Medica (No. JKLPSE201805).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The author(s) declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 2917 kb)
Rights and permissions
About this article
Cite this article
Qian, W., Miao, Z., Zhang, XJ. et al. Functionalized reduced graphene oxide with aptamer macroarray for cancer cell capture and fluorescence detection. Microchim Acta 187, 407 (2020). https://doi.org/10.1007/s00604-020-04402-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-020-04402-8