Abstract
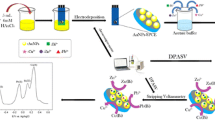
This paper presents an electrochemical sensor for Cr(VI) (chromate ion) in water. A disposable screen-printed electrode was modified with gold nanostars (AuNSs) that were synthesized by Good’s buffer method. Linear sweep voltammetry (LSV) was employed for the detection of Cr(VI) in 0.1 M sulfuric acid solution. The AuNSs are shown to provide higher current response to Cr(VI) than spherically shaped gold nanoparticles. The sensor gives the strongest response at a scan rate of 0.05 V (vs Ag/AgCl) and exhibits minimal interference from other electroactive species. The linear range extends from 10 to 75,000 ppb, and the limit of detection is 3.5 ppb. This is well below the provisional guideline value given by the World Health Organization. Excellent recoveries (ranging between 95 and 97%) were found when analyzing contaminated ground water samples obtained from a site situated in Wellesley, MA.

Schematic presentation of preparation of gold nanostars (AuNS) on carbon paste screen printed electrode (CPSPE) by drop casting and electrochemical detection of chromium (VI) using linear sweep voltammetry (LSV).






Similar content being viewed by others
References
Oze C, Bird DK, Fendorf S (2007) Genesis of hexavalent chromium from natural sources in soil and groundwater. Proc Natl Acad Sci 104(16):6544–6549
Moffat I, Martinova N, Seidel C, Thompson CM (2018) Hexavalent chromium in drinking water. Journal-American Water Works Association 110(5):E22–E35
Costa M (2003) Potential hazards of hexavalent chromate in our drinking water. Toxicol Appl Pharmacol 188(1):1–5
Smith AH, Steinmaus CM (2009) Health effects of arsenic and chromium in drinking water: recent human findings. Annu Rev Public Health 30:107–122
Paustenbach D, Rinehart W, Sheehan P (1991) The health hazards posed by chromium-contaminated soils in residential and industrial areas: conclusions of an expert panel. Regul Toxicol Pharmacol 13(2):195–222
Sharma P, Bihari V, Agarwal SK, Verma V, Kesavachandran CN, Pangtey BS, Mathur N, Singh KP, Srivastava M, Goel SK (2012) Groundwater contaminated with hexavalent chromium [Cr (VI)]: a health survey and clinical examination of community inhabitants (Kanpur, India). PLoS One 7(10):e47877
EPA U (2013) The third unregulated contaminant monitoring rule (UCMR 3): data summary. EPA, Washington DC
Jacobs JA, Testa SM (2005) Overview of chromium (VI) in the environment: background and history. Chromium (VI) handbook. CRC Press, New York, pp 1–21
Grabarczyk M (2008) Speciation analysis of chromium by adsorptive stripping voltammetry in tap and river water samples. Electroanalysis: An International Journal Devoted to Fundamental and Practical Aspects of Electroanalysis 20(20):2217–2222
Baś B, Bugajna A, Jakubowska M, Niewiara E (2012) Normal pulse voltammetric determination of subnanomolar concentrations of chromium (VI) with continuous wavelet transformation. Electroanalysis 24(11):2157–2164
Grabarczyk M, Baś B, Korolczuk M (2009) Application of a renewable silver based mercury film electrode to the determination of Cr (VI) in soil samples. Microchim Acta 164(3–4):465–470
Ouyang R, Zhang W, Zhou S, Xue Z-L, Xu L, Gu Y, Miao Y (2013) Improved bi film wrapped single walled carbon nanotubes for ultrasensitive electrochemical detection of trace Cr (VI). Electrochim Acta 113:686–693
Jorge E, Rocha M, Fonseca I, Neto M (2010) Studies on the stripping voltammetric determination and speciation of chromium at a rotating-disc bismuth film electrode. Talanta 81(1–2):556–564
Cox JA, Kulesza PJ (1983) Stripping voltammetry of chromium (VI) at a poly (4-vinvlpyridine)-coated platium electrode. Anal Chim Acta 154:71–78
Hallam PM, Kampouris DK, Kadara RO, Banks CE (2010) Graphite screen printed electrodes for the electrochemical sensing of chromium (VI). Analyst 135(8):1947–1952
Miscoria SA, Jacq C, Maeder T, Negri RM (2014) Screen-printed electrodes for electroanalytical sensing, of chromium VI in strong acid media. Sensors Actuators B Chem 195:294–302
Sánchez-Moreno RA, Gismera MJ, Sevilla MT, Procopio JR (2011) Potentiometric screen-printed Bisensor for simultaneous determination of chromium (III) and chromium (VI). Electroanalysis 23(1):287–294
Tsai M-C, Chen P-Y (2008) Voltammetric study and electrochemical detection of hexavalent chromium at gold nanoparticle-electrodeposited indium tinoxide (ITO) electrodes in acidic media. Talanta 76(3):533–539
Ouyang R, Bragg SA, Chambers JQ, Xue Z-L (2012) Flower-like self-assembly of gold nanoparticles for highly sensitive electrochemical detection of chromium (VI). Anal Chim Acta 722:1–7
Dutta S, Strack G, Kurup P (2019) Gold nanostar electrodes for heavy metal detection. Sensors Actuators B Chem 281:383–391
de Puig H, Tam JO, Yen C-W, Gehrke L, Hamad-Schifferli K (2015) Extinction coefficient of gold nanostars. J Phys Chem C 119(30):17408–17415
Haiss W, Thanh NT, Aveyard J, Fernig DG (2007) Determination of size and concentration of gold nanoparticles from UV− Vis spectra. Anal Chem 79(11):4215–4221
Jena BK, Raj CR (2008) Highly sensitive and selective electrochemical detection of sub-ppb level chromium (VI) using nano-sized gold particle. Talanta 76(1):161–165
Svancara I, Foret P, Vytras K (2004) A study on the determination of chromium as chromate at a carbon paste electrode modified with surfactants. Talanta 64(4):844–852
Bergamini MF, dos Santos DP, Zanoni MVB (2007) Development of a voltammetric sensor for chromium (VI) determination in wastewater sample. Sensors Actuators B Chem 123(2):902–908
Calvo-Pérez A, Domínguez-Renedo O, Alonso-Lomillo M, Arcos-Martínez M (2014) Speciation of chromium using chronoamperometric biosensors based on screen-printed electrodes. Anal Chim Acta 833:15–21
Domínguez-Renedo O, Ruiz-Espelt L, García-Astorgano N, Arcos-Martínez MJ (2008) Electrochemical determination of chromium (VI) using metallic nanoparticle-modified carbon screen-printed electrodes. Talanta 76(4):854–858
Metters JP, Kadara RO, Banks CE (2011) New directions in screen printed electroanalytical sensors: an overview of recent developments. Analyst 136(6):1067–1076
Kachoosangi RT, Compton RG (2013) Voltammetric determination of chromium (VI) using a gold film modified carbon composite electrode. Sensors Actuators B Chem 178:555–562
Jin W, Wu G, Chen A (2014) Sensitive and selective electrochemical detection of chromium (VI) based on gold nanoparticle-decorated titania nanotube arrays. Analyst 139(1):235–241
Wang Y, Laborda E, Crossley A, Compton RG (2013) Surface oxidation of gold nanoparticles supported on a glassy carbon electrode in sulphuric acid medium: contrasts with the behaviour of ‘macro’gold. Phys Chem Chem Phys 15(9):3133–3136
Purwidyantri A, Chen C-H, Chen L-Y, Chen C-C, Luo J-D, Chiou C-C, Tian Y-C, Lin C-Y, Yang C-M, Lai H-C (2017) Speckled zno nanograss electrochemical sensor for staphylococcus epidermidis detection. J Electrochem Soc 164(6):B205–B211
Khairy M, Choudry NA, Ouasti M, Kampouris DK, Kadara RO, Banks CE (2010) Gold nanoparticle ensembles allow mechanistic insights into electrochemical processes. ChemPhysChem 11(4):875–879
Seo M, Chung TD (2019) Nanoconfinement effects in electrochemical reactions. Current Opinion in Electrochemistry 13:47–54
Acknowledgments
This work was supported by the National Science Foundation award # 1543042. Any opinions, findings, and conclusions or recommendations expressed in this paper are those of the researchers and do not necessarily reflect the views of the funding agency. The authors would like to thank Dr. Earl Ada for performing the TEM imaging. The authors acknowledge Mr. John Fitzgerald (MassDEP), and two graduate students Connor Sullivan and Michaela Fitzgerald for sampling water from groundwater monitoring wells located in Wellesley, MA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author(s) declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 693 kb)
Rights and permissions
About this article
Cite this article
Dutta, S., Strack, G. & Kurup, P. Gold nanostar-based voltammetric sensor for chromium(VI). Microchim Acta 186, 734 (2019). https://doi.org/10.1007/s00604-019-3847-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-019-3847-1