Abstract
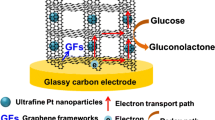
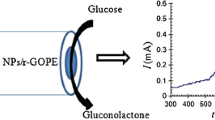
We have performed a study on the performance of two microbial glucose sensors based on immobilized Gluconobacter oxydans (G. oxydans). The first one was prepared by modifying a glassy carbon paste electrode (GCPE) containing the microbial cells with graphene oxide (GO), the other one by modifying it with graphene-platinum hybrid nanoparticles (graphene-Pt NPs). The electrode was characterized by following the voltammetric signals of the oxidation of hexacyanoferrate(II) to hexacyanoferrate(III) via the oxidative enzymes contained in G. oxydans which convert glucose to gluconic acid. Optimizations were conducted with a conventional GCPE containing G. oxydans. After material optimization, the biosensors were applied to the determination of glucose. The linear and analytical ranges for GO based biosensor range from 1 to 75 μM (linear) and 1 to 100 μM (analytical), respectively, with a limit of detection (LOD) of (3 s/m) 1.06 μM (at an S/m of 3). On the other hand, the graphene-Pt hybrid nanoparticle based biosensor showed two linear ranges (from 0.3 to 1 µM and from 1 to 10 μM), a full analytical range from 1 to 50 μM, and an LOD of 0.015 μM. The graphene-Pt hybrid NP based sensors performs better and was applied to the determination of glucose in synthetically prepared plasma samples where it gave recoveries as 101.8 and 104.37 % for two different concentrations. Selectivity studies concerning fructose, galactose, L-ascorbic acid and dopamine were also conducted.

The electrochemical performances of graphene oxide and graphene-platinum hybrid nanoparticle modified G. oxydans biosensors were compared.







Similar content being viewed by others
References
Anik Ü, Çevik S, Pumera M (2010) Effect of nitric acid “washing” procedure on electrochemical behavior of carbon nanotubes and glassy carbon μ-particles. Nanoscale Res Lett 5:846–852
Çevik S, Anik Ü (2010) Banana tissue-nanoparticle/nanotube based glassy carbon paste electrode biosensors for catechol detection. Sens Lett 8:667–671
Apetrei I, Apetrei C (2015) The biocomposite screen-printed biosensor based on immobilization of tyrosinase onto the carboxyl functionalised carbon nanotube for assaying tyramine in fish products. J Food Eng 149:1–8
Pumera M (2009) Electrochemistry of graphene: new horizons for sensing and energy storage. Chem Rec 9:211–223
Wang Y, Li Y, Tang L, Lu J, Li J (2009) Application of graphene-modified electrode for selective detection of dopamine. Electrochem Commun 11:889–892
Bo Y, Yang H, Hu Y, Yao T, Huang S (2011) A novel electrochemical DNA biosensor based on graphene and polyaniline nanowires. Electrochim Acta 56:2676–2681
Shan C, Yang H, Song J, Han D, Ivaska A, Niu L (2009) Direct electrochemistry of glucose oxidase and biosensing for glucose based on graphene. Anal Chem 81:2378–2382
Zhou M, Zhai Y, Dong S (2009) Electrochemical sensing and biosensing platform based on chemically reduced graphene oxide. Anal Chem 81:5603–5613
Khim Chng EL, Chua CK, Pumera M (2014) Graphene oxide nanoribbons exhibit significantly greater toxicity than graphene oxide nanoplatelets. Nanoscale 6(18):10792–10797
Georgakilas V, Gournisb D, Tzitziosa V, Pasquato L, Guldie DM, Prato M (2007) Decorating carbon nanotubes with metal or semiconductor nanoparticles. J Mater Chem 26:2679–2694
Xu C, Wang X, Zhu J (2008) Graphene-metal particle nanocomposites. J Phys Chem C 112:19841–19845
Li D, Kaner RB (2008) Graphene-based materials. Science 320:1170–1171
Xing YC (2004) Synthesis and electrochemical characterization of uniformly-dispersed high loading Pt nanoparticles on sonochemically-treated carbon nanotubes. J Phys Chem B 108:19255–19259
Novoselov KS, Geim AK, Morozov SV, Jiang D, Katsnelson MI, Grigorieva IV, Dubonos SV, Firsov AA (2005) Two-dimensional gas of massless Dirac fermions in graphene. Nature 438:197–200
Sultan SC, Anik Ü (2014) Gr–Pt hybrid NP modified GCPE as label and indicator free electrochemical genosensor platform. Talanta 129:523–528
Tepeli Y, Anik U (2015) Comparison of performances of bioanodes modified with graphene oxide and graphene-platinum hybrid nanoparticles. Electrochem Commun. doi:10.1016/j.elecom.2015.05.002
Alferov SV, Tomashevskaya LG, Ponamoreva ON, Bogdanovskaya VA, Reshetilov AN (2006) Biofuel cell anode based on the gluconobacter oxydans bacteria cells and 2,6-dichlorophenolindophenol as an electron transport mediator. Elektrokhimiya 42:456–457
Tkac J, Svitel J, Vostiar I, Navratil M, Gemeiner P (2009) Membrane-bound dehydrogenases from gluconobacter sp.: interfacial electrochemistry and direct bioelectrocatalysis. Bioelectrochemistry 76:53–62
Hummers WSJ, Offeman R (1958) Preparation of graphitic oxide. J Am Chem Soc 80:1339–1339
Li D, Müller MB, Gilje S, Kaner RB, Wallace GG (2008) Processable aqueous dispersions of graphene nanosheets. Nat Nanotechnol 3:101–105
Stankovich S, Dikin DA, Dommett GH, Kohlhaas KM, Zimney EJ, Stach EA, Piner RD, Nguyen ST, Ruoff RS (2006) Systematic post-assembly modification of graphene oxide paper with primary alkylamines. Nature 442:282–286
Vostiar I, Ferapontova EE, Gorton L (2004) Electrical wiring of viable Gluconobacter oxydans cells with flexible osmium-redox polyelectrolyte. Electrochem Commun 6:621–626
Gatgens C, Degner U, Bringer-Meyer S, Hermann U (2007) Biotransformation of glycerol to dihydroxyacetone by recombinant Gluconobacter oxydans DSM 2343. Appl Microbiol Biotechnol 76:553–559
Coldur F, Andac M, Isildak I (2010) Flow-injection potentiometric applications of solid state Li+ selective electrode in biological and pharmaceutical samples. J Solid State Electrochem 14:2241–2249
Wang J (2001) Glucose biosensors: 40 years of advances and challenges. Electroanalysis 13:983–988
Tkac J, Vostiar I, Gorton L, Gemeiner P, Sturdık E (2003) Improved selectivity of microbial biosensor using membrane coating. Biosens Bioelectron 18:1125–1134
Matsuo Y, Miyabe T, Fukutsuka T, Sugie Y (2007) Preparation and characterization of alkylamine-intercalated graphite oxides. Carbon 45:1005–1012
Kovtyukhova NI, Ollivier PJ, Martin BR, Mallouk TE, Chizhik SA, Buzaneva EV, Gorchinskiy AD (1999) Layer-by-layer assembly of ultrathin composite films from micron-sized graphite oxide sheets and polycations. Chem Mater 11:771–778
Herrera-Alonso M, Abdala AA, McAllister MJ, Aksay IA, Prud’homme RK (2007) Intercalation and stitching of graphite oxide with diaminoalkanes. Langmuir 23:10644–10649
Li D, Müller MB, Gilje S, Kaner RB, Wallace GG (2007) Processable aqueous dispersions of graphene nanosheets. Nat Nanotechnol 3:101–105
Zhang L, Li N, Jiu H, Qi G, Huang Y (2015) ZnO-reduced graphene oxide nanocomposites as efficient photocatalysts for photocatalytic reduction of CO2. Ceram Int 41:6256–6262
Yildirim N, Odaci Demirkol D, Timur S (2015) Modified gold surfaces with gold nanoparticles and 6-(ferrocenyl)hexanethiol: design of a mediated microbial sensor. Electroanalysis 27:52–57
Sharifirad M, Kiani F, Koohyar F (2014) Design of a microbial sensor using a conducting polymer of polyaniline/poly 4,4’-diaminodiphenyl sulphone-silver nanocomposite films on a carbon paste electrode. Mater Technol 48:209–214
Guler E, Soyleyici HC, Odaci Demirkol D, Ak M, Timur S (2014) A novel functional conducting polymer as an immobilization platform. Mater Sci Eng C 40:148–156
Yılmaz Ö, Odacı Demirkol D, Gülcemal S, Kılınc A, Timur S, Cetinkaya B (2012) Chitosan–ferrocene film as a platform for flow injection analysis applications of glucose oxidase and Gluconobacter oxydans biosensors. Colloids Surf B: Biointerfaces 100:62–68
Acknowledgments
The grant from Muğla Sıtkı Koçman University BAP Project number 13 / 17 is greatly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 198 kb)
Rights and permissions
About this article
Cite this article
Aslan, S., Anik, Ü. Microbial glucose biosensors based on glassy carbon paste electrodes modified with Gluconobacter Oxydans and graphene oxide or graphene-platinum hybrid nanoparticles. Microchim Acta 183, 73–81 (2016). https://doi.org/10.1007/s00604-015-1590-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-015-1590-9