Abstract
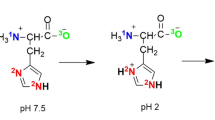
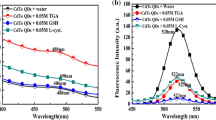
3-Mercaptopropionic acid (MPA)-capped CdTe nanocrystals (NCs) were synthesized in aqueous medium, and their interaction with cysteine (Cys) and homocysteine (Hcy) was studied by steady-state and time-resolved fluorescence spectra at different pH. At 6.4 < pH < 8.0, the fluorescence of CdTe NCs can be effectively enhanced by Cys and Hcy. While pH > 9.6, only Cys quenches the fluorescence of the CdTe NCs, no fluorescence changes are observed for Hcy. Mechanism study shows that these pH manipulating fluorescence responses can be attributed to the following two reasons: first, both the thiol–thiolate equilibrium of Cys (Hcy) and the number of undercoordinated NCs surface sites capped with dual coordinated ligands are strong pH-dependent; second, different thiol-containing amino acids, with different redox energy level, can lead to distinguishable fluorescence responses of NCs. Based on these unique fluorescence responses, the possibilities of developing a sensitive detecting technique for Cys/Hcy and Cys through pH modulation can be explored.






Similar content being viewed by others
References
Klostranec JM, Chan WCW (2006) Quantum dots in biological and biomedical research: recent progress and present challenges. Adv Mater 18:1953
Bruchez M Jr, Moronne M, Gin P, Weiss S, Alivisatos AP (1998) Semiconductor nanocrystals as fluorescent biological labels. Science 281:2013
Chan WCW, Nie S (1998) Quantum dot bioconjugates for ultrasensitive nonisotopic detection. Science 281:2016
Costa-Fernández JM, Pereiro R, Sanz-Medel A (2006) The use of luminescent quantum dots for optical sensing. TrAC Trends Anal Chem 25:207
Wang LY, Zhou YY, Wang L, Zhu CQ, Li YX, Gao F (2002) Synchronous fluorescence determination of protein with functionalized nanoparticles of CdS as a fluorescence probe. Anal Chim Acta 466:87
Wang LY, Wang L, Gao F, Yu ZY, Wu ZM (2002) Application of functionalized CdS nanoparticles as fluorescence probe in the determination of nucleic acids. Analyst 127:977
Zhu CQ, Zhao DH, Chen JL, Li YX, Wang LY, Wang L, Zhou YY, Zhuo SJ, Wu YQ (2004) Application of L-cysteine-capped nano-ZnS as a fluorescence probe for the determination of proteins. Anal Bioanal Chem 378:811
Priyam A, Chatterjee A, Das SK, Saha A (2005) Size dependent interaction of biofunctionalized CdS nanoparticles with tyrosine at different pH. Chem Commun 4122. DOI 10.1039/b505960g
Wang W, Rusin O, Xu X, Kim KK, Escobedo JO, Fakayode SO, Fletcher KA, Lowry M, Schowalter CM, Lawrence CM, Fronczek FR, Warner IM, Strongin RM (2005) Detection of homocysteine and cysteine. J Am Chem Soc 127:15949
Mamedova NN, Kotov NA, Rogach AL, Studer J (2001) Albumin-CdTe nanoparticle bioconjugates: preparation, structure, and interunit energy transfer with antenna effect. Nano Lett 1:281
Kho R, Torres-Martínez CL, Mehra RK (2000) A simple colloidal synthesis for gram-quantity production of water-soluble ZnS nanocrystal powders. J Colloids Interface Soc 227:561
Chen JL, Zhu CQ (2005) Functionalized cadmium sulfide quantum dots as fluorescence probe for silver ion determination. Anal Chim Acta 546:147
Xia Y, Zhang T, Diao X, Zhu C (2007) Measurable emission color change: size-dependent reversible fluorescence quenching of CdTe quantum dots by molecular oxygen. Chem Lett 36:242
Xia YS, Zhu CQ (2008) Aqueous synthesis of luminescent magic sized CdSe nanoclusters. Mater Lett DOI 10.1016/j.matlet.2007.11.027 (in press)
Aldana J, Lavelle N, Wang Y, Peng X (2005) Size-dependent dissociation pH of thiolate ligands from cadmium chalcogenide nanocrystals. J Am Chem Soc 127:2496
Friedman M, Cavins JF, Wall JS (1965) Relative nucleophilic reactivities of amino groups and mercaptide ions in addition reactions with α, β-unsaturated compounds1,2. J Am Chem Soc 87:3672
Jeong S, Achermann M, Nanda J, Ivanov S, Klimov VI, Hollingsworth JA (2005) Effect of the thiol–thiolate equilibrium on the photophysical properties of aqueous CdSe/ZnS nanocrystal quantum dots. J Am Chem Soc 127:10126
Klimov VI, McBranch DW, Leatherdale CA, Bawendi MG (1999) Electron and hole relaxation pathways in semiconductor quantum dots. Phys Rev B 60:13740
Wuister SF, de Mello Donegá C, Meijerink A (2004) Influence of thiol capping on the exciton luminescence and decay kinetics of CdTe and CdSe quantum dots. J Phys Chem B 108:17393
van Beek R, Zoombelt AP, Jenneskens LW, vanWalree CA, de Mello Donegá C, Veldman D, Janssen RAJ (2006) Side chain mediated electronic contact between a tetrahydro-4H-thiopyran-4-ylidene-appended polythiophene and CdTe quantum dots. Chem Eur J 12:8075
Zappacosta B, Mordente A, Persichilli S, Minucci A, Carlino P, Martorana GE, Giardina B, De Sole P (2001) Is homocysteine a pro-oxidant? Free Radical Res 35:499
Park JB (2001) Reduction of dehydroascorbic acid by homocysteine. Biochim Biophys Acta 1525:173
Chow C, Chiu BKW, Lam MHW, Wong W (2003) A trinuclear heterobimetallic Ru(II)/Pt(II) complex as a chemodosimeter selective for sulfhydryl-containing amino acids and peptides. J Am Chem Soc 125:7802
Fu Y, Li H, Hu W, Zhu D (2005) Fluorescence probes for thiol-containing amino acids and peptides in aqueous solution. Chem Commun 3189. DOI 10.1039/b503772g
Zhang M, Yu M, Li F, Zhu M, Li M, Gao Y, Li L, Liu Z, Zhang J, Zhang D, Yi T, Huang C (2007) A highly selective fluorescence turn-on sensor for cysteine/homocysteine and its application in bioimaging. J Am Chem Soc 129:10322
Nekrassoa O, Lawrence NS, Copton RG (2003) Analytical determination of homocysteine: a review. Talanta 60:1085
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 20575002), the Natural Science Foundation of Anhui Province (No. 070416239) and Program for Innovative Research Team in Anhui Normal University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xia, YS., Zhu, CQ. Interaction of CdTe nanocrystals with thiol-containing amino acids at different pH: a fluorimetric study. Microchim Acta 164, 29–34 (2009). https://doi.org/10.1007/s00604-008-0025-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-008-0025-2