Abstract
Purpose
The preoperative platelet-to-lymphocyte ratio (PLR) has been reported as an important prognostic index for pancreatic ductal adenocarcinoma (PDAC); however, the significance of the postoperative (post-op) PLR for this disease has not been elucidated.
Methods
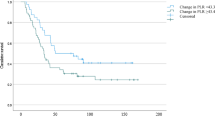
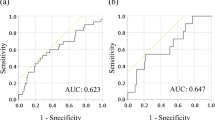
We analyzed data on 118 patients who underwent pancreaticoduodenectomy for pancreatic head PDAC, collected from a prospectively maintained database. The post-op PLR was obtained by dividing the platelet count after surgery by the lymphocyte count on post-op day (POD) 14. The patients were divided into two groups according to a post-op PLR of < 310 or ≥ 310. Survival data were analyzed.
Results
A high post-op PLR was identified as a significant prognostic index on univariate analysis for disease-free survival (DFS) and overall survival (OS). The post-op PLR remained significant, along with tumor differentiation and adjuvant chemotherapy, on multivariate analysis for OS (hazard ratio = 2.077, 95% confidence interval: 1.220–3.537; p = 0.007). The post-op PLR was a significant independent prognostic index for poor DFS, along with tumor differentiation and lymphatic invasion, on multivariate analysis (hazard ratio = 1.678, 95% confidence interval: 1.056–2.667; p = 0.028).
Conclusions
The post-op PLR in patients with pancreatic head PDAC was an independent predictor of DFS and OS after elective resection.



Similar content being viewed by others
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2000;71:209–49.
Oettle H, Neuhaus P, Hochhaus A, Hartmann JT, Gellert K, Ridwelski K, et al. Adjuvant chemotherapy with gemcitabine and long-term outcomes among patients with resected pancreatic cancer: the CONKO-001 randomized trial. JAMA. 2013;310:1473–81.
Conroy T, Hammel P, Hebbar M, Abdelghani MB, Wei AC, Raoul JL, et al. FOLFIRINOX or gemcitabine as adjuvant therapy for pancreatic cancer. N Engl J Med. 2018;379:2395–406.
Sakaguchi T, Satoi S, Yamamoto T, Yamaki S, Sekimoto M. The past, present, and future status of multimodality treatment for resectable/borderline resectable pancreatic ductal adenocarcinoma. Surg Today. 2020;50(4):335–43.
Gall TMH, Jacob J, Frampton AE, Krell J, Kyriakides C, Castellano L, et al. Reduced dissemination of circulating tumor cells with no-touch isolation surgical technique in patients with pancreatic cancer. JAMA Surg. 2014;149:482–5.
Nierodzik ML, Plotkin A, Kajumo F, Karpatkin S. Thrombin stimulates tumor-platelet adhesion in vitro and metastasis in vivo. J Clin Invest. 1991;87:229–36.
Shirai Y, Shiba H, Sakamoto T, Horiuchi K, Haruki K, Fujiwara Y, et al. Preoperative platelet to lymphocyte ratio predicts outcome of patients with pancreatic ductal adenocarcinoma after pancreatic resection. Surgery. 2015;158:360–5.
Shirai Y, Shiba H, Haruki K, Horiuchi T, Saito N, Fujiwara Y, et al. Preoperative platelet-to-albumin ratio predicts prognosis of patients with pancreatic ductal adenocarcinoma after pancreatic resection. Anticancer Res. 2017;37:787–93.
Watanabe J, Otani S, Sakamoto T, Arai Y, Hanaki T, Amisaki M, et al. Prognostic indicators based on inflammatory and nutritional factors after pancreaticoduodenectomy for pancreatic cancer. Surg Today. 2016;46:1258–67.
Asari S, Matsumoto I, Toyama H, Shinzeki M, Goto T, Ishida J, et al. Preoperative independent prognostic factors in patients with borderline resectable pancreatic ductal adenocarcinoma following curative resection: the neutrophil-lymphocyte and platelet-lymphocyte ratios. Surg Today. 2016;46:583–92.
Bong TSH, Tan GHC, Chia C, Soo KC, Teo MCC. Preoperative platelet-lymphocyte ratio is an independent prognostic marker and superior to carcinoembryonic antigen in colorectal peritoneal carcinomatosis patients undergoing cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. Int J Clin Oncol. 2017;22:511–8.
Kakita A, Takahashi T, Yoshida M, Furuta K. A simpler and more reliable technique of pancreatojejunal anastomosis. Surg Today. 1996;26:532–5.
Fujii T, Sugimoto H, Yamada S, Kanda M, Suenaga M, Takami H, et al. Modified Blumgart anastomosis for pancreaticojejunostomy: technical improvement in matched historical control study. J Gastrointest Surg. 2014;18:1108–15.
Brierley DJ GK, Wittekind C. TNM Classification of Malignant Tumor, 8th edition. Wiley-Blackwell. 2017.
Bassi C, Marchegiani G, Dervenis C, Sarr M, Hilal MA, Adham M, et al. The 2016 update of the International Study Group (ISGPS) definition and grading of postoperative pancreatic fistula: 11 years after. Surgery. 2017;161:584–91.
Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205–13.
Alexandrakis MG, Passam FH, Moschandrea IA, Christophoridou AV, Pappa CA, Coulocheri SA, et al. Levels of serum cytokines and acute phase proteins in patients with essential and cancer-related thrombocytosis. Am J Clin Oncol. 2003;26:135–40.
Ivert T, Dalén M, Ander C, Stålesen R, Lordkipanidzé M, Hjemdahl P, et al. Increased platelet reactivity and platelet-leukocyte aggregation after elective coronary bypass surgery. Platelets. 2019;30:975–81.
Pugalenthi A, Protic M, Gonen M, Kingham TP, D’Angelica MI, Dematteo RP, et al. Postoperative complications and overall survival after pancreaticoduodenectomy for pancreatic ductal adenocarcinoma. J Surg Oncol. 2016;113:188–93.
Aoyama T, Murakawa M, Katayama Y, Yamaoku K, Kanazawa A, Higuchi A, et al. Impact of postoperative complications on survival and recurrence in pancreatic cancer. Anticancer Res. 2015;35:2401–9.
Wojtukiewicz MZ, Sierko E, Hempel D, Tucker SC, Honn KV. Platelets and cancer angiogenesis nexus. Cancer Metastasis Rev. 2017;36:249–62.
Goubran HA, Stakiw J, Radosevic M, Burnouf T. Platelets effects on tumor growth. Semin Oncol. 2014;41:359–69.
Bastida E, Almirall L, Ordinas A. Tumor-cell-induced platelet aggregation is a glycoprotein-dependent and lipoxygenase-associated process. Int J Cancer. 1987;39:760–3.
Amo L, Tamayo-Orbegozo E, Maruri N, Eguizabal C, Zenarruzabeitia O, Riñón M, et al. Involvement of platelet-tumor cell interaction in immune evasion. Potential role of podocalyxin-like protein 1. Frontier Oncol. 2014;4:245.
Quail DF, Joyce JA. Microenvironmental regulation of tumor progression and metastasis. Nat Med. 2013;19:1423–37.
Miyashita T, Tajima H, Makino I, Nakagawa H, Kitagawa H, Fushida S, et al. Metastasis-promoting role of extravasated platelet activation in tumor. J Surg Res. 2015;193:289–94.
Okazaki M, Yamaguchi T, Tajima H, Fushida S, Ohta T. Platelet adherence to cancer cells promotes escape from innate immune surveillance in cancer metastasis. Int J Oncol. 2020;57:980–8.
Ishikawa M, Nishioka M, Hanaki N, Miyauchi T, Kashiwagi Y, Ioki H, et al. Perioperative immune responses in cancer patients undergoing digestive surgeries. World J Surg Oncol. 2009;7:7.
Yang MW, Tao LY, Jiang YS, Yang JY, Huo YM, Liu DJ, et al. Perineural invasion reprograms the immune microenvironment through cholinergic signaling in pancreatic ductal adenocarcinoma. Cancer Res. 2020;80:1991–2003.
Uesaka K, Boku N, Fukutomi A, Okamura Y, Konishi M, Matsumoto I, et al. Adjuvant chemotherapy of S-1 versus gemcitabine for resected pancreatic cancer: a phase 3, open-label, randomised, non-inferiority trial (JASPAC 01). Lancet. 2016;388:248–57.
Rothwell PM, Wilson M, Price JF, Belch JF, Meade TW, Mehta Z. Effect of daily aspirin on risk of cancer metastasis: a study of incident cancers during randomised controlled trials. Lancet. 2012;379:1591–601.
Khalaf N, Yuan C, Hamada T, Cao Y, Babic A, Morales-Oyarvide V, et al. Regular use of aspirin or non-aspirin nonsteroidal anti-inflammatory drugs is not associated with risk of incident pancreatic cancer in two large cohort studies. Gastroenterology. 2018;154:1380-90.e5.
Coyle C, Cafferty FH, Rowley S, MacKenzie M, Berkman L, Gupta S, et al. ADD-ASPIRIN: A phase III, double-blind, placebo controlled, randomised trial assessing the effects of aspirin on disease recurrence and survival after primary therapy in common non-metastatic solid tumours. Contemp Clin Trial. 2016;51:56–64.
Pretzsch E, D’Haese JG, Renz B, Ilmer M, Schiergens T, Miksch RC, et al. Effect of platelet inhibition with perioperative aspirin on survival in patients undergoing curative resection for pancreatic cancer: a propensity score matched analysis. BMC Surg. 2021;21:98.
Funding
This work was supported by Japan Society for the Promotion of Science (JSPS) KAKENHI Grant Number 20K17665 and 23K08202 (Dr. Yoshihiro Shirai).
Author information
Authors and Affiliations
Contributions
YS and RH designed the study. YS wrote the main manuscript. YT, TT, MY, KH, KF, SO and TS acquired the data. TG and TI made a critical revision. All the authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
We have no conflicts of interests to declare for this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shirai, Y., Hamura, R., Tanji, Y. et al. The postoperative platelet-to-lymphocyte ratio predicts the outcome of patients undergoing pancreaticoduodenectomy for pancreatic head cancer. Surg Today 54, 247–257 (2024). https://doi.org/10.1007/s00595-023-02727-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-023-02727-4