Abstract
Purpose
The purpose of this study was to evaluate the long-term outcomes of robotic rectal cancer surgery and to examine the risk factors for recurrence.
Methods
In a high-volume center in Japan, we retrospectively enrolled patients with pStage I–III rectal cancer within 15 cm of the anal verge who underwent robotic surgery from 2011 to 2017. Almost all patients underwent upfront surgery, and lateral lymph-node dissection (LLND) was performed for patients with locally advanced lower rectal cancer. We evaluated the 5-year overall survival (OS), relapse-free survival (RFS), and cumulative local recurrence (LR) rates and examined the risk factors for the RFS.
Results
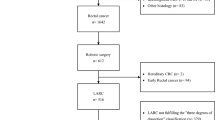
We evaluated 488 patients who underwent robotic rectal cancer surgery, including 5.1% who underwent preoperative chemoradiotherapy to obtain clear resection margins and 33.6% who underwent LLND. There were 203, 87, and 198 patients with pStage I, II, and III, respectively, and the positive resection margin rate was 1.0%. The 5-year OS, RFS, and LR rates were 95.5%, 81.7%, and 2.2%, respectively. The independent risk factors for the RFS were the presence of venous invasion, extramural tumor deposits without lymph-node structure, and pT ≥ 3.
Conclusion
This study demonstrated the favorable long-term outcomes of robotic rectal cancer surgery.




Similar content being viewed by others
Data availability
The datasets generated during and analyzed during the current study are available from the corresponding author on reasonable request.
References
Heald RJ, Husband EM, Ryall RD. The mesorectum in rectal cancer surgery–the clue to pelvic recurrence? Br J Surg. 1982;69:613–6.
Stevenson ARL, Solomon MJ, Brown CSB, Lumley JW, Hewett P, Clouston AD, et al. Disease-free survival and local recurrence after laparoscopic-assisted resection or open resection for rectal cancer: the Australasian laparoscopic cancer of the rectum randomized clinical trial. Ann Surg. 2019;269:596–602.
Fleshman J, Branda ME, Sargent DJ, Boller AM, George VV, Abbas MA, et al. Disease-free survival and local recurrence for laparoscopic resection compared with open resection of stage ii to iii rectal cancer: follow-up results of the ACOSOG Z6051 randomized controlled trial. Ann Surg. 2019;269:589–95.
Broholm M, Pommergaard H-C, Gögenür I. Possible benefits of robot-assisted rectal cancer surgery regarding urological and sexual dysfunction: a systematic review and meta-analysis. Colorectal Dis. 2015;17:375–81.
Prete FP, Pezzolla A, Prete F, Testini M, Marzaioli R, Patriti A, et al. Robotic versus laparoscopic minimally invasive surgery for rectal cancer: a systematic review and meta-analysis of randomized controlled trials. Ann Surg. 2018;267:1034–46.
Jayne D, Pigazzi A, Marshall H, Croft J, Corrigan N, Copeland J, et al. Effect of robotic-assisted vs conventional laparoscopic surgery on risk of conversion to open laparotomy among patients undergoing resection for rectal cancer: the ROLARR randomized clinical trial. JAMA. 2017;318:1569–80.
Kim MJ, Park SC, Park JW, Chang HJ, Kim DY, Nam B-H, et al. Robot-assisted versus laparoscopic surgery for rectal cancer: a phase ii open label prospective randomized controlled trial. Ann Surg. 2018;267:243–51.
Park EJ, Cho MS, Baek SJ, Hur H, Min BS, Baik SH, et al. Long-term oncologic outcomes of robotic low anterior resection for rectal cancer: a comparative study with laparoscopic surgery. Ann Surg. 2015;261:129–37.
Kim J, Baek S-J, Kang D-W, Roh Y-E, Lee JW, Kwak H-D, et al. Robotic resection is a good prognostic factor in rectal cancer compared with laparoscopic resection: long-term survival analysis using propensity score matching. Dis Colon Rectum. 2017;60:266–73.
Sammour T, Malakorn S, Bednarski BK, Kaur H, Shin US, Messick C, et al. Oncological outcomes after robotic proctectomy for rectal cancer: analysis of a prospective database. Ann Surg. 2018;267:521–6.
Chang W, Wei Y, Ren L, Jian M, Chen Y, Chen J, et al. Short-term and long-term outcomes of robotic rectal surgery-from the real word data of 1145 consecutive cases in China. Surg Endosc. 2020;34:4079–88.
Tilney HS, Huddy JR, Nizar AS, Smith R, Gudgeon AM. Minimal access rectal cancer surgery: an observational study of patient outcomes from a district general hospital with over a decade of experience with robotic rectal cancer surgery. Colorectal Dis. 2021;23:1961–70.
Hashiguchi Y, Muro K, Saito Y, Ito Y, Ajioka Y, Hamaguchi T, et al. Japanese society for cancer of the colon and rectum (JSCCR) guidelines 2019 for the treatment of colorectal cancer. Int J Clin Oncol. 2020;25:1–42.
Glynne-Jones R, Wyrwicz L, Tiret E, Brown G, Rödel C, Cervantes A, et al. Rectal cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2017;28:iv22–40.
Benson AB, Venook AP, Al-Hawary MH, Arain MA, Chen Y-J, Ciombor KK, et al. NCCN guidelines insights: rectal cancer, version 6.2020. J Natl Compr Canc Netw. 2020;18:806–15.
Cercek A, Roxburgh CSD, Strombom P, Smith JJ, Temple LKF, Nash GM, et al. Adoption of total neoadjuvant therapy for locally advanced rectal cancer. JAMA Oncol. 2018;4: e180071.
Yamaguchi T, Kinugasa Y, Shiomi A, Kagawa H, Yamakawa Y, Furuatni A, et al. Short- and long-term outcomes of robotic-assisted laparoscopic surgery for rectal cancer: results of a single high-volume center in Japan. Int J Colorectal Dis. 2018;33:1755–62.
Brierley JD, Gorley JD, Gospodarowicz MK, Wittekind C. TNM classification of malignant tumours. 8th ed. Hoboken: Wiley-Blackwell; 2017.
Akiyoshi T, Watanabe T, Miyata S, Kotake K, Muto T, Sugihara K, et al. Results of a Japanese nationwide multi-institutional study on lateral pelvic lymph node metastasis in low rectal cancer: is it regional or distant disease? Ann Surg. 2012;255:1129–34.
Ueno H, Mochizuki H, Shirouzu K, Kusumi T, Yamada K, Ikegami M, et al. Multicenter study for optimal categorization of extramural tumor deposits for colorectal cancer staging. Ann Surg. 2012;255:739–46.
Shiomi A, Kinugasa Y, Yamaguchi T, Tomioka H, Kagawa H. Robot-assisted rectal cancer surgery: short-term outcomes for 113 consecutive patients. Int J Colorectal Dis. 2014;29:1105–11.
Kasai S, Kagawa H, Shiomi A, Hino H, Manabe S, Yamaoka Y, et al. Advantages of robotic abdominoperineal resection compared with laparoscopic surgery: a single-center retrospective study. Surg Today. 2022;52(4):643–51.
Yamaoka Y, Shiomi A, Kagawa H, Hino H, Manabe S, Kato S, et al. Robotic surgery for clinical T4 rectal cancer: short- and long-term outcomes. Surg Endosc. 2022;36:91–9.
Kagawa H, Kinugasa Y, Shiomi A, Yamaguchi T, Tsukamoto S, Tomioka H, et al. Robotic-assisted lateral lymph node dissection for lower rectal cancer: short-term outcomes in 50 consecutive patients. Surg Endosc. 2015;29:995–1000.
Hino H, Shiomi A, Kagawa H, Manabe S, Yamaoka Y, Kato S, et al. Neoadjuvant chemoradiotherapy for borderline resectable low rectal cancer: short- and long-term outcomes at a single Japanese center. Surg Today. 2022. https://doi.org/10.1007/s00595-021-02419-x,January8.
Clavien PA, Barkun J, de Oliveira ML, Vauthey JN, Dindo D, Schulick RD, et al. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg. 2009;250:187–96.
Kanda Y. Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transpl. 2013;48:452–8.
Park SY, Lee SM, Park JS, Kim HJ, Choi G-S. Robot surgery shows similar long-term oncologic outcomes as laparoscopic surgery for mid/lower rectal cancer but is beneficial to ypT3/4 after preoperative chemoradiation. Dis Colon Rectum. 2021;64:812–21.
van Gijn W, Marijnen CAM, Nagtegaal ID, Kranenbarg EM-K, Putter H, Wiggers T, et al. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer: 12-year follow-up of the multicenter, randomised controlled TME trial. Lancet Oncol. 2011;12:575–82.
Sauer R, Liersch T, Merkel S, Fietkau R, Hohenberger W, Hess C, et al. Preoperative versus postoperative chemoradiotherapy for locally advanced rectal cancer: results of the German CAO/ARO/AIO-94 randomized phase III trial after a median follow-up of 11 years. J Clin Oncol. 2012;30:1926–33.
Ogura A, Konishi T, Cunningham C, Garcia-Aguilar J, Iversen H, Toda S, et al. Neoadjuvant (chemo)radiotherapy with total mesorectal excision only is not sufficient to prevent lateral local recurrence in enlarged nodes: results of the multicenter Lateral Node Study of patients with low cT3/4 rectal cancer. J Clin Oncol. 2019;37:33–43.
Fujita S, Akasu T, Mizusawa J, Saito N, Kinugasa Y, Kanemitsu Y, et al. Postoperative morbidity and mortality after mesorectal excision with and without lateral lymph node dissection for clinical stage II or stage III lower rectal cancer (JCOG0212): results from a multicentre, randomised controlled, non-inferiority trial. Lancet Oncol. 2012;13:616–21.
Tsukamoto S, Fujita S, Ota M, Mizusawa J, Shida D, Kanemitsu Y, et al. Long-term follow-up of the randomized trial of mesorectal excision with or without lateral lymph node dissection in rectal cancer (JCOG0212). Br J Surg. 2020;107:586–94.
Yamaguchi T, Kinugasa Y, Shiomi A, Tomioka H, Kagawa H. Robotic-assisted laparoscopic versus open lateral lymph node dissection for advanced lower rectal cancer. Surg Endosc. 2016;30:721–8.
Yamaguchi T, Kinugasa Y, Shiomi A, Kagawa H, Yamakawa Y, Furutani A, et al. Oncological outcomes of robotic-assisted laparoscopic versus open lateral lymph node dissection for locally advanced low rectal cancer. Surg Endosc. 2018;32:4498–505.
Peacock O, Limvorapitak T, Bednarski BK, Kaur H, Taggart MW, Dasari A, et al. Robotic lateral pelvic lymph node dissection after chemoradiation for rectal cancer: a Western perspective. Colorectal Dis. 2020;22:2049–56.
Marijnen CAM, van de Velde CJH, Putter H, van den Brink M, Maas CP, Martijn H, et al. Impact of short-term preoperative radiotherapy on health-related quality of life and sexual functioning in primary rectal cancer: report of a multicenter randomized trial. J Clin Oncol. 2005;23:1847–58.
Peeters KCMJ, van de Velde CJH, Leer JWH, Martijn H, Junggeburt JMC, Kranenbarg EK, et al. Late side effects of short-course preoperative radiotherapy combined with total mesorectal excision for rectal cancer: increased bowel dysfunction in irradiated patients–a Dutch colorectal cancer group study. J Clin Oncol. 2005;23:6199–206.
Chand M, Siddiqui MRS, Swift I, Brown G. Systematic review of prognostic importance of extramural venous invasion in rectal cancer. World J Gastroenterol. 2016;22:1721–6.
Nagtegaal ID, Knijn N, Hugen N, Marshall HC, Sugihara K, Tot T, et al. Tumor deposits in colorectal cancer: improving the value of modern staging-a systematic review and meta-analysis. J Clin Oncol. 2017;35:1119–27.
Lord AC, DʼSouza N, Shaw A, Rokan Z, Moran B, Abulafi M, et al. MRI-diagnosed tumour deposits and EMVI status have superior prognostic accuracy to current clinical TNM staging in rectal cancer. Ann Surg. 2020. https://doi.org/10.1097/SLA.0000000000004499.
Lord AC, Knijn N, Brown G, Nagtegaal ID. Pathways of spread in rectal cancer: a reappraisal of the true routes to distant metastatic disease. Eur J Cancer. 2020;128:1–6.
Hashimoto DA, Rosman G, Rus D, Meireles OR. Artificial intelligence in surgery: promises and perils. Ann Surg. 2018;268:70–6.
Hino H, Shiomi A, Hatakeyama K, Kagawa H, Manabe S, Yamaoka Y, et al. Comprehensive genetic characterization of rectal cancer in a large cohort of Japanese patients: differences according to tumor location. J Gastroenterol. 2022. https://doi.org/10.1007/s00535-022-01875-7,April21.
Taylor FGM, Quirke P, Heald RJ, Moran BJ, Blomqvist L, Swift IR, et al. Preoperative magnetic resonance imaging assessment of circumferential resection margin predicts disease-free survival and local recurrence: 5-year follow-up results of the MERCURY study. J Clin Oncol. 2014;32:34–43.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Yusuke Kinugasa received lecture fees from Johnson and Johnson and Intuitive Surgical.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kasai, S., Kagawa, H., Shiomi, A. et al. Long-term outcomes of upfront robotic rectal cancer surgery: a single-center, retrospective cohort study in Japan. Surg Today 53, 1028–1037 (2023). https://doi.org/10.1007/s00595-023-02648-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-023-02648-2