Abstract
Purpose
To assess the impact of surgical approach on morbidity, mortality, and the oncological outcomes of synchronous (SC) and metachronous (MC) colorectal cancer (CRC).
Methods
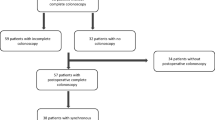
All patients undergoing resection for double location CRC (SC or MC) between 2006 and 2020 were included. The exclusion criteria were polyposis or SC located on the same side.
Results
Sixty-seven patients (age, 64.8 years; male, 78%) with SC (n = 41; 61%) or MC (n = 26; 39%) were included. SC was treated with segmental colectomy (right and left colectomy/proctectomy; n = 19) or extensive colectomy (subtotal/total colectomy or restorative proctocolectomy with pouch; n = 22). Segmental colectomy was associated with a higher incidence of anastomotic leakage (47.4 vs. 13.6%; p = 0.04) and a higher rate of medical morbidity (47.4 vs. 16.6%; p = 0.04). The mean number of lymph nodes harvested was similar. For MC, the second cancer was treated by iterative colectomy (n = 12) or extensive colectomy (n = 14) and there was no significant difference in postoperative outcomes between the two surgical approaches. The median follow-up period was 42.4 ± 29.1 months. The 5-year overall and disease-free survival of the SC and MC groups did not differ to a statistically significant extent.
Conclusions
Extensive colectomy should be preferred for SC to reduce morbidity and improve the prognosis. In contrast, iterative colectomy can be performed safely for patients with MC.


Similar content being viewed by others
Data availability
On request to the corresponding author.
Code availability
Not applicable.
References
van Leersum NJ, Aalbers AG, Snijders HS, Henneman D, Wouters MW, Tollenaar RA, et al. Synchronous colorectal carcinoma: a risk factor in colorectal cancer surgery. Dis Colon Rectum. 2014;57:460–6.
Latournerie M, Jooste V, Cottet V, Lepage C, Faivre J, Bouvier AM. Epidemiology and prognosis of synchronous colorectal cancers. Br J Surg. 2008;95:1528–33.
Velayos FS, Lee SH, Qiu H, Dykes S, Yiu R, Terdiman JP, et al. The mechanism of microsatellite instability is different in synchronous and metachronous colorectal cancer. J Gastrointest Surg. 2005;9:329–35.
Nosho K, Kure S, Irahara N, Shima K, Baba Y, Spiegelman D, et al. A prospective cohort study shows unique epigenetic, genetic, and prognostic features of synchronous colorectal cancers. Gastroenterology. 2009;137(1609–20):e1-3.
Rex DK, Kahi CJ, Levin B, Smith RA, Bond JH, Brooks D, et al. Guidelines for colonoscopy surveillance after cancer resection: a consensus update by the American cancer society and US multi-society task force on colorectal cancer. CA Cancer J Clin. 2006;56:160–7.
Park SH, Lee JH, Lee SS, Kim JC, Yu CS, Kim HC, et al. CT colonography for detection and characterisation of synchronous proximal colonic lesions in patients with stenosing colorectal cancer. Gut. 2012;61:1716–22.
Mulder SA, Kranse R, Damhuis RA, de Wilt JH, Ouwendijk RJ, Kuipers EJ, et al. Prevalence and prognosis of synchronous colorectal cancer: a Dutch population-based study. Cancer Epidemiol. 2011;35:442–7.
Passman MA, Pommier RF, Vetto JT. Synchronous colon primaries have the same prognosis as solitary colon cancers. Dis Colon Rectum. 1996;39:329–34.
Nikoloudis N, Saliangas K, Economou A, Andreadis E, Siminou S, Manna I, et al. Synchronous colorectal cancer. Tech Coloproctol. 2004;8(Suppl 1):s177–9.
Hu H, Chang DT, Nikiforova MN, Kuan SF, Pai RK. Clinicopathologic features of synchronous colorectal carcinoma: a distinct subset arising from multiple sessile serrated adenomas and associated with high levels of microsatellite instability and favorable prognosis. Am J Surg Pathol. 2013;37:1660–70.
Kaibara N, Koga S, Jinnai D. Synchronous and metachronous malignancies of the colon and rectum in Japan with special reference to a coexisting early cancer. Cancer. 1984;54:1870–4.
Wang HZ, Huang XF, Wang Y, Ji JF, Gu J. Clinical features, diagnosis, treatment and prognosis of multiple primary colorectal carcinoma. World J Gastroenterol. 2004;10:2136–9.
Easson AM, Cotterchio M, Crosby JA, Sutherland H, Dale D, Aronson M, et al. A population-based study of the extent of surgical resection of potentially curable colon cancer. Ann Surg Oncol. 2002;9:380–7.
Holubar SD, Wolff BG, Poola VP, Soop M. Multiple synchronous colonic anastomoses: are they safe? Colorectal Dis. 2010;12:135–40.
Steel EJ, Trainer AH, Heriot AG, Lynch C, Parry S, Win AK, et al. The experience of extended bowel resection in individuals with a high metachronous colorectal cancer risk: a qualitative study. Oncol Nurs Forum. 2016;43:444–52.
You YN, Chua HK, Nelson H, Hassan I, Barnes SA, Harrington J. Segmental vs. extended colectomy: measurable differences in morbidity, function, and quality of life. Dis Colon Rectum. 2008;51:1036–43.
Mulder SA, Kranse R, Damhuis RA, Ouwendijk RJ, Kuipers EJ, van Leerdam ME. The incidence and risk factors of metachronous colorectal cancer: an indication for follow-up. Dis Colon Rectum. 2012;55:522–31.
Papadopoulos V, Michalopoulos A, Basdanis G, Papapolychroniadis K, Paramythiotis D, Fotiadis P, et al. Synchronous and metachronous colorectal carcinoma. Tech Coloproctol. 2004;8(Suppl 1):s97–100.
Shitoh K, Konishi F, Miyakura Y, Togashi K, Okamoto T, Nagai H. Microsatellite instability as a marker in predicting metachronous multiple colorectal carcinomas after surgery: a cohort-like study. Dis Colon Rectum. 2002;45:329–33.
Ringland CL, Arkenau HT, O’Connell DL, Ward RL. Second primary colorectal cancers (SPCRCs): experiences from a large Australian cancer registry. Ann Oncol. 2010;21:92–7.
Amin MB, Greene FL, Edge SB, Compton CC, Gershenwald JE, Brookland RK, et al. The Eighth edition AJCC cancer staging manual: continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J Clin. 2017;67:93–9.
Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205–13.
Moertel CG, Bargen JA, Dockerty MB. Multiple carcinomas of the large intestine: a review of the literature and a study of 261 cases. Gastroenterology. 1958;34:85–98.
Lindberg LJ, Ladelund S, Bernstein I, Therkildsen C, Nilbert M. Risk of synchronous and metachronous colorectal cancer: population-based estimates in Denmark with focus on non-hereditary cases diagnosed after age 50. Scand J Surg. 2019;108:152–8.
Segelman J, Mattsson I, Jung B, Nilsson PJ, Palmer G, Buchli C. Risk factors for anastomotic leakage following ileosigmoid or ileorectal anastomosis. Colorectal Dis. 2018;20:304–11.
Duclos J, Lefevre JH, Lefrancois M, Lupinacci R, Shields C, Chafai N, et al. Immediate outcome, long-term function and quality of life after extended colectomy with ileorectal or ileosigmoid anastomosis. Colorectal Dis. 2014;16:O288–96.
Shen R, Zhang Y, Wang T. Indocyanine green fluorescence angiography and the incidence of anastomotic leak after colorectal resection for colorectal cancer: a meta-analysis. Dis Colon Rectum. 2018;61:1228–34.
Mohan HM, Ryan E, Balasubramanian I, Kennelly R, Geraghty R, Sclafani F, et al. Microsatellite instability is associated with reduced disease specific survival in stage III colon cancer. Eur J Surg Oncol. 2016;42:1680–6.
Colle R, Cohen R, Cochereau D, Duval A, Lascols O, Lopez-Trabada D, et al. Immunotherapy and patients treated for cancer with microsatellite instability. Bull Cancer. 2017;104:42–51.
Lynch HT, Lynch PM, Lanspa SJ, Snyder CL, Lynch JF, Boland CR. Review of the Lynch syndrome: history, molecular genetics, screening, differential diagnosis, and medicolegal ramifications. Clin Genet. 2009;76:1–18.
Parry S, Win AK, Parry B, Macrae FA, Gurrin LC, Church JM, et al. Metachronous colorectal cancer risk for mismatch repair gene mutation carriers: the advantage of more extensive colon surgery. Gut. 2011;60:950–7.
Jayasekara H, Reece JC, Buchanan DD, Rosty C, Dashti SG, Ait Ouakrim D, et al. Risk factors for metachronous colorectal cancer following a primary colorectal cancer: a prospective cohort study. Int J Cancer. 2016;139:1081–90.
Stjepanovic N, Moreira L, Carneiro F, Balaguer F, Cervantes A, Balmana J, et al. Hereditary gastrointestinal cancers: ESMO clinical practice guidelines for diagnosis, treatment and follow-updagger. Ann Oncol. 2019;30:1558–71.
Anyla M, Lefevre JH, Creavin B, Colas C, Svrcek M, Lascols O, et al. Metachronous colorectal cancer risk in Lynch syndrome patients-should the endoscopic surveillance be more intensive? Int J Colorectal Dis. 2018;33:703–8.
Haanstra JF, de Nederveen Cappel WH, Gopie JP, Vecht J, Vanhoutvin SA, Cats A, et al. Quality of life after surgery for colon cancer in patients with Lynch syndrome: partial versus subtotal colectomy. Dis Colon Rectum. 2012;55:653–9.
Pollett WG, Marion K, Moeslein G, Schneider C, Parry S, Veysey K, et al. Quality of life after surgery in individuals with familial colorectal cancer: does extended surgery have an adverse impact? ANZ J Surg. 2014;84:359–64.
Mirnezami A, Mirnezami R, Chandrakumaran K, Sasapu K, Sagar P, Finan P. Increased local recurrence and reduced survival from colorectal cancer following anastomotic leak: systematic review and meta-analysis. Ann Surg. 2011;253:890–9.
Hain E, Maggiori L, Manceau G, Mongin C, AlDJ P, Panis Y. Oncological impact of anastomotic leakage after laparoscopic mesorectal excision. Br J Surg. 2017;104:288–95.
Chen HS, Sheen-Chen SM. Synchronous and “early” metachronous colorectal adenocarcinoma: analysis of prognosis and current trends. Dis Colon Rectum. 2000;43:1093–9.
Funding
No funding to report.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare no conflict of interest in association with the present study.
Ethical approval
All the procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Consent to participate
Informed consent was obtained from all the individual participants included in the study.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nguyen, J., Lefèvre, J.H., Bouchet-Doumenq, C. et al. Surgery for synchronous and metachronous colorectal cancer: segmental or extensive colectomy?. Surg Today 53, 338–346 (2023). https://doi.org/10.1007/s00595-022-02624-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-022-02624-2