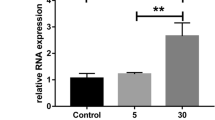
Abstract
Aims
Although diabetic retinopathy has long been considered a microvascular complication, retinal neurodegeneration and inflammation may precede its clinical manifestations. Despite all research efforts, the primary treatment options remain laser photocoagulation and anti-vascular endothelial growth factor (VEGF) intravitreal injections, both aggressive and targeting the late stages of the disease. Medical treatments addressing the early phases of diabetic retinopathy are therefore needed. We aimed at verifying if thiamine and fenofibrate protect the cells of the inner blood-retinal barrier from the metabolic stress induced by diabetic-like conditions.
Methods
Human microvascular endothelial cells (HMECs), retinal pericytes (HRPs) and Müller cells (MIO-M1) were cultured in intermittent high glucose (intHG) and/or hypoxia, with addition of fenofibrate or thiamine. Modulation of adhesion molecules and angiogenic factors was addressed.
Results
Integrins β1/αVβ3 and ICAM1 were upregulated in HMECs/HRPs cultured in diabetic-like conditions, as well as metalloproteases MMP2/9 in HRP, with a reduction in their inhibitor TIMP1; MMP2 increased also in HMEC, and TIMP1 decreased in MIO-M1. VEGF and HIF-1α were strongly increased in HMEC in intHG + hypoxia, and VEGF also in HRP. Ang-1/2 augmented in HMEC/MIO-M1, and MCP-1 in HRP/MIO-M1 in intHG + hypoxia. Thiamine was able to normalize all such abnormal modulations, while fenofibrate had effects in few cases only.
Conclusions
We suggest that endothelial cells and pericytes are more affected than Müller cells by diabetic-like conditions. Fenofibrate shows a controversial behavior, potentially positive on Müller cells and pericytes, but possibly detrimental to endothelium, while thiamine confirms once more to be an effective agent in reducing diabetes-induced retinal damage.






Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Arroba AI, Valverde ÁM (2017) Modulation of microglia in the retina: new insights into diabetic retinopathy. Acta Diabetol 54:527–533. https://doi.org/10.1007/s00592-017-0984-z
Armulik A, Abramsson A, Betsholtz C (2005) Endothelial/pericyte interactions. Circ Res 97:512–523. https://doi.org/10.1161/01.RES.0000182903.16652.d7
Coughlin BA, Feenstra DJ, Mohr S (2017) Müller cells and diabetic retinopathy. Vision Res 139:93–100. https://doi.org/10.1016/j.visres.2017.03.013
Thornalley PJ, Babaei-Jadidi R, Al Ali H et al (2007) High prevalence of low plasma thiamine concentration in diabetes linked to a marker of vascular disease. Diabetologia 50:2164–2170. https://doi.org/10.1007/s00125-007-0771-4
Hammes H, Du X, Edelstein D, Taguchi T et al (2003) Benfotiamine blocks three major pathways of hyperglycemic damage and prevents experimental diabetic retinopathy. Nat Med 9:294–299. https://doi.org/10.1038/nm834
Babaei-Jadidi R, Karachalias N, Ahmed N, Battah S, Thornalley PJ (2003) Prevention of incipient diabetic nephropathy by high-dose thiamine and benfotiamine. Diabetes 52:2110–2120. https://doi.org/10.2337/diabetes.52.8.2110
Beltramo E, Mazzeo A, Lopatina T, Trento M, Porta M (2020) Thiamine transporter 2 is involved in high glucose-induced damage and altered thiamine availability in cell models of diabetic retinopathy. Diab Vasc Dis Res 17:1479164119878427. https://doi.org/10.1177/1479164119878427
Keech AC, Mitchell P, Summanen PA et al (2007) Effect of fenofibrate on the need for laser treatment for diabetic retinopathy (FIELD study): a randomised controlled trial. Lancet 370:1687–1697. https://doi.org/10.1016/S0140-6736(07)61607-9
ACCORD Eye Study Group, Chew EY, Ambrosius WT et al (2010) Effects of medical therapies on retinopathy progression in type 2 diabetes. N Engl J Med 363:233–244. https://doi.org/10.1056/NEJMoa1001288
Roy S, Kim D, Hernández C, Simó R, Roy S (2015) Beneficial effects of fenofibric acid on overexpression of extracellular matrix components, COX-2, and impairment of endothelial permeability associated with diabetic retinopathy. Exp Eye Res 140:124–129. https://doi.org/10.1016/j.exer.2015.08.010
Chen Y, Hu Y, Lin M et al (2013) Therapeutic effects of PPARα agonists on diabetic retinopathy in type 1 diabetes models. Diabetes 62:261–272. https://doi.org/10.2337/db11-0413
Abcouwer SF (2013) Direct effects of PPARα agonists on retinal inflammation and angiogenesis may explain how fenofibrate lowers risk of severe proliferative diabetic retinopathy. Diabetes 62:36–38. https://doi.org/10.2337/db12-1223
Meissner M, Stein M, Urbich C et al (2004) PPARalpha activators inhibit vascular endothelial growth factor receptor-2 expression by repressing Sp1-dependent DNA binding and transactivation. Circ Res 94:324–332. https://doi.org/10.1161/01.RES.0000113781.08139.81
Berrone E, Beltramo E, Buttiglieri S et al (2009) Establishment and characterization of a human retinal pericyte line: a novel tool for the study of diabetic retinopathy. Int J Mol Med 23:373–378. https://doi.org/10.3892/ijmm_00000141
Limb GA, Salt TE, Munro PMG, Moss SE, Khaw PT (2002) In vitro characterization of a spontaneously immortalized human Müller cell line (MIO-M1). Invest Ophthalmol Vis Sci 43:864–869
Beltramo E, Berrone E, Tarallo S, Porta M (2009) Different apoptotic responses of human and bovine pericytes to fluctuating glucose levels and protective role of thiamine. Diabetes Metab Res Rev 25:566–576. https://doi.org/10.1002/dmrr.996
Tarallo S, Beltramo E, Berrone E, Dentelli P, Porta M (2010) Effects of high glucose and thiamine on the balance between matrix metalloproteinases and their tissue inhibitors in vascular cells. Acta Diabetol 47:105–111. https://doi.org/10.1007/s00592-009-0124-5
Beltramo E, Lopatina T, Berrone E et al (2014) Extracellular vesicles derived from mesenchymal stem cells induce features of diabetic retinopathy in vitro. Acta Diabetol 51:1055–1064. https://doi.org/10.1007/s00592-014-0672-1
Reynolds AL, Alvarez Y, Sasore T et al (2016) Phenotype-based discovery of 2-[(E)-2-(quinolin-2-yl)vinyl]phenol as a novel regulator of ocular angiogenesis. J Biol Chem 291:7242–7245. https://doi.org/10.1074/jbc.M115.710665
Wu N, Shen H, Wang Y, He B et al (2017) Role of the PKCβII/JNK signaling pathway in acute glucose fluctuation-induced apoptosis of rat vascular endothelial cells. Acta Diabetol 54:727–736. https://doi.org/10.1007/s00592-017-0999-5
Picconi F, Parravano M, Sciarretta F et al (2019) Activation of retinal Müller cells in response to glucose variability. Endocrine 65:542–549. https://doi.org/10.1007/s12020-019-02017-5
Picconi F, Parravano M, Ylli D et al (2017) Retinal neurodegeneration in patients with type 1 diabetes mellitus: the role of glycemic variability. Acta Diabetol 54:489–497. https://doi.org/10.1007/s00592-017-0971-4
LaFoya B, Munroe JA, Miyamoto A et al (2018) Beyond the matrix: the many non-ECM ligands for integrins. Int J Mol Sci 19:449. https://doi.org/10.3390/ijms19020449
Yao Y, Du J, Li R et al (2019) Association between ICAM-1 level and diabetic retinopathy: a review and meta-analysis. Postgrad Med J 95(1121):162–168. https://doi.org/10.1136/postgradmedj-2018-136102
Klein T, Bischoff R (2011) Physiology and pathophysiology of matrix metalloproteases. Amino Acids 41:271–290. https://doi.org/10.1007/s00726-010-0689-x
Limb GA, Daniels JT, Pleass R, Charteris DG, Luthert PJ, Khaw PT (2002) Differential expression of matrix metalloproteinases 2 and 9 by glial Müller cells: response to soluble and extracellular matrix-bound tumor necrosis factor-alpha. Am J Pathol 160(5):1847–1855. https://doi.org/10.1016/s0002-9440(10)61131-5
Jin M, Kashiwagi K, Iizuka Y, Imai M, Tsukahara S (2001) Matrix metalloproteinases in human diabetic and nondiabetic vitreous. Retina 21:28–33. https://doi.org/10.1097/00006982-200102000-00005
D’Amico AG, Maugeri G, Rasà DM et al (2018) NAP counteracts hyperglycemia/hypoxia induced retinal pigment epithelial barrier breakdown through modulation of HIFs and VEGF expression. J Cell Physiol 233:1120–1128. https://doi.org/10.1002/jcp.25971
Mazzeo A, Lopatina T, Gai C, Trento M, Porta M, Beltramo E (2019) Functional analysis of miR-21-3p, miR-30b-5p and miR-150-5p shuttled by extracellular vesicles from diabetic subjects reveals their association with diabetic retinopathy. Exp Eye Res 184:56–63. https://doi.org/10.1016/j.exer.2019.04.015
Cvitkovic K, Sesar A, Sesar I et al (2020) Concentrations of selected cytokines and vascular endothelial growth factor in aqueous humor and serum of diabetic patients. Semin Ophthalmol 35:126–133. https://doi.org/10.1080/08820538.2020.1755320
Hammes HP, Lin J, Wagner P et al (2004) Angiopoietin-2 causes pericyte dropout in the normal retina. evidence for involvement in diabetic retinopathy. Diabetes 53:1104–1110. https://doi.org/10.2337/diabetes.53.4.1104
Metheny-Barlow LJ, Li LY (2003) The enigmatic role of angiopoietin-1 in tumor angiogenesis. Cell Res 13:309–317. https://doi.org/10.1038/sj.cr.7290176
Akwii RG, Sajib MS, Zahra FT, Mikelis CM (2019) Role of angiopoietin-2 in vascular physiology and pathophysiology. Cells 8(5):471. https://doi.org/10.3390/cells8050471
Oh H, Takagi H, Suzuma K, Otani A, Matsumura M, Honda Y (1999) Hypoxia and vascular endothelial growth factor selectively up-regulate angiopoietin-2 in bovine microvascular endothelial cells. J Biol Chem 274:15732–15739. https://doi.org/10.1074/jbc.274.22.15732
Hakanpaa L, Sipila T, Leppanen VM et al (2015) Endothelial destabilization by angiopoietin-2 via integrin beta1 activation. Nat Commun 6:5962. https://doi.org/10.1038/ncomms6962
Beltramo E, Porta M (2013) Pericyte loss in diabetic retinopathy: mechanisms and consequences. Curr Med Chem 20:3218–3225. https://doi.org/10.2174/09298673113209990022
Olson LE, Soriano P (2011) PDGFRβ signaling regulates mural cell plasticity and inhibits fat development. Dev Cell 20:815–826. https://doi.org/10.1016/j.devcel.2011.04.019
La Selva M, Beltramo E, Pagnozzi F et al (1996) Thiamine corrects delayed replication and decreases production of lactate and advanced glycation end-products in bovine retinal and umbilical vein endothelial cells cultured under high glucose conditions. Diabetologia 39:1263–1268. https://doi.org/10.1007/s001250050568
Beltramo E, Berrone E, Tarallo S, Porta M (2008) Effects of thiamine and benfotiamine on intracellular glucose metabolism and relevance in the prevention of diabetic complications. Acta Diabetol 45:131–141. https://doi.org/10.1007/s00592-008-0042-y
Nishikawa T, Edelstein D, Brownlee M (2000) The missing link: a single unifying mechanism for diabetic complications. Kidney Int 58:S26–S30. https://doi.org/10.1046/j.1523-1755.2000.07705.x
Berrone E, Beltramo E, Solimine C, Ubertalli Ape A, Porta M (2006) Regulation of intracellular glucose and polyol pathway by thiamine and benfotiamine in vascular cells cultured in high glucose. J Biol Chem 281:9307–9313. https://doi.org/10.1074/jbc.M600418200
Sweet RL, Zastre JA (2013) HIF1-α-mediated gene expression induced by vitamin B1 deficiency. Int J Vitam Nutr Res 83(3):188–197. https://doi.org/10.1024/0300-9831/a000159
Beltramo E, Nizheradze K, Berrone E, Tarallo S, Porta M (2009) Thiamine and benfotiamine prevent apoptosis induced by high glucose-conditioned extracellular matrix in human retinal pericytes. Diabetes Metab Res Rev 25:647–656. https://doi.org/10.1002/dmrr.1008
Porta M, Toppila I, Sandholm N et al (2016) Variation in SLC19A3 and Protection from Microvascular Damage in Type 1 Diabetes. Diabetes 65:1022–1030. https://doi.org/10.2337/db15-1247
Garcia-Ramírez M, Hernández C, Palomer X, Vázquez-Carrera M, Simó R (2016) Fenofibrate prevents the disruption of the outer blood retinal barrier through downregulation of NF-κB activity. Acta Diabetol 53:109–118. https://doi.org/10.1007/s00592-015-0759-3
Stewart S, Lois N (2018) Fenofibrate for diabetic retinopathy. Asia Pac J Ophthalmol (Phila) 7:422–426. https://doi.org/10.22608/APO.2018288
Yuan J, Tan JTM, Rajamani K et al (2019) Fenofibrate rescues diabetes-related impairment of ischemia-mediated angiogenesis by PPARα-independent modulation of thioredoxin-interacting protein. Diabetes 68:1040–1053. https://doi.org/10.2337/db17-0926
Funding
This study was funded by MIUR-Italian Ministry for University and Research—PRIN 2015 (project number 2015JXE7E8) and the University of Turin (Ricerca locale ex-60% 2018).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Aurora Mazzeo, Chiara Gai and Elena Beltramo. The first draft of the manuscript was written by Elena Beltramo and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical approval
Ethical approval is not applicable to this work.
Informed consent
Informed consent is not applicable to this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article belongs to the topical collection Eye Complications of Diabetes, managed by Giuseppe Querques.
Rights and permissions
About this article
Cite this article
Mazzeo, A., Gai, C., Trento, M. et al. Effects of thiamine and fenofibrate on high glucose and hypoxia-induced damage in cell models of the inner blood-retinal barrier. Acta Diabetol 57, 1423–1433 (2020). https://doi.org/10.1007/s00592-020-01565-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-020-01565-x