Abstract
Purpose
This study aimed to investigate the impact of the severity of cervical ossification of the posterior longitudinal ligament (OPLL) on the incidence of arteriosclerosis in the carotid artery.
Methods
Patients with OPLL-induced cervical myelopathy were prospectively enrolled. The study involved analyzing patient characteristics, blood samples, computed tomography scans of the spine, and intima-media thickness (IMT) measurements of the common carotid artery. Patients were divided into two groups based on the size of the cervical OPLL to compare demographic data, comorbidities, and the presence of thickening of the carotid intima-media (max IMT ≥ 1.1 mm).
Results
The study included 96 patients (mean age: 63.5 years; mean body mass index: 26.9 kg/m2; 71.8% male; 35.4% with diabetes mellitus). The mean maximum anteroposterior (AP) diameter of the OPLL was 4.9 mm, with a mean occupancy ratio of 43%. The mean maximum IMT was 1.23 mm. Arteriosclerosis of the carotid artery was diagnosed in 62.5% of the patients. On comparing the two groups based on OPLL size, the group with larger OPLL (≥ 5 mm) had a higher BMI and a greater prevalence of carotid intima-media thickening. This significant difference in the prevalence of carotid intima-media thickening persisted even after adjusting for patient backgrounds using propensity score matching.
Conclusions
Patients with a larger cervical OPLL showed a higher frequency of intima-media thickening in the carotid artery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ossification of the posterior longitudinal ligament (OPLL) is one of the causes of compressive myelopathy. OPLL is often reported in the Asian population, with a frequency of 1.9–4.3% diagnosed by X-ray or 6.7% diagnosed by computed tomography (CT) in Japan [1,2,3,4,5]. As the reported incidences of OPLL were 0.4–3.0% in Asian countries other than Japan and 0.1–1.7% in Europe and the USA [6,7,8], ethnicity is considered a factor related to the onset of OPLL [9]. Nevertheless, the precise mechanism of OPLL remains unclear.
Other factors related to OPLL include disorders of metabolism [10]. Specifically, the occurrence of diabetes mellitus is high in patients with OPLL [5, 11]. Besides, patients with severe OPLL often demonstrate obesity [12, 13]. Some experimental studies implied the involvement of several cytokines in the pathogenesis of OPLL, which could explicate the higher incidence of obesity or diabetes mellitus in patients with OPLL [14, 15].
Considering the correlation of obesity and metabolic diseases with arteriosclerosis, patients with OPLL tend to be at high risk of developing arteriosclerosis. However, to date, only one epidemiological study has reported a higher incidence of carotid plaques among asymptomatic patients with a relatively smaller size of OPLL [3], and the correlation between OPLL in myelopathic patients and arteriosclerosis remains unclear.
Hence, this study aims to investigate the impact of the OPLL severity on the incidence of arteriosclerosis in the carotid artery in myelopathic patients. To the best of our knowledge, this is the first study to identify an association between OPLL severity and a higher incidence of arteriosclerosis.
Materials and methods
Study design
This prospective, single-center study was approved by the Institutional Review Board of the authors’ institution (No. 2674–2) and was conducted per the approved guidelines and principles of the Declaration of Helsinki. All participants provided written informed consent before undergoing any study procedure.
Study participants
We prospectively enrolled 105 patients with OPLL-caused cervical myelopathy between April 2020 and July 2021. All patients underwent laboratory tests, CT scans of the whole spine, and ultrasound of the carotid artery. In this study, OPLL was defined when the ossification of the posterior longitudinal ligament was > 2 mm thick in the axial view in the cervical spine, which was measured as previously described [1, 16]. TD performed the CT measurements, and YO independently conducted measurements to determine the intraclass correlation coefficient (ICC). While CT examined the maximum anteroposterior (AP) diameter of the OPLL, OP index, and OPLL types (segmental/continuous/mixed), ultrasound examined the carotid artery and measured maximum intima-media thickness (IMT) in the common carotid artery (CCA). The presence of arteriosclerosis was defined as the maximum IMT of ≥ 1.1 mm [17]. All examinations were performed using Aplio 300 (Canon Medical Systems Corporation, Tochigi, Japan).
After excluding nine patients with incomplete data, we divided the remaining 96 patients into two groups based on their OPLL size. This division was based on a cutoff of 4.9 mm, which represents the average anteroposterior (AP) diameter of OPLL in our study group. We then compared the demographic characteristics between these two groups. In addition, we adjusted for factors such as age, gender, BMI, comorbidities, and the presence of thoracic spine lesions using propensity scores. This adjustment allowed for a more accurate comparison of the frequency of carotid intima-media thickening between the groups.
Statistical analysis
While continuous variables were compared using the Fisher’s exact test or Student t test, categorical variables were compared using the χ2 test. Interobserver reliability for the CT measurement was evaluated using the ICC. A propensity score-matched analysis was performed after adjusting for confounding factors related to arteriosclerosis such as age, sex, BMI, and comorbidities. All statistical analyses were performed using IBM SPSS Statistics software version 25.0 (IBM Corporation, Armonk, NY). In this study, all P values were two-sided, and P < 0.05 was considered statistically significant.
Results
Of 105 patients, we analyzed the data of 96 patients [mean age: 63.5 years; body mass index (BMI), 26.9 kg/m2; 69 men (71.8%); 27 women (28.2%); 34 patients (35.4%) with diabetes mellitus]. The mean value of the maximum AP diameter of the OPLL on CT was 4.9 mm, and the mean occupying ratio of the canal was 43%. The ICC for the CT measurement was found to be 0.97. The mean value of the maximum IMT of the CCA was 1.23 mm, and 60 patients (62.5%) were diagnosed with arteriosclerosis of the carotid artery (Table 1).
Patients were categorized into two groups based on the AP diameter of the ossification of the OPLL: one with an AP diameter ≥ 5.0 mm and the other with an AP diameter ≤ 4.9 mm. A comparative analysis of patient demographics and clinical characteristics was then conducted between these groups. The group with larger OPLL (≥ 5 mm) exhibited a higher body mass index (BMI), an increased prevalence of carotid intima-media thickening, and a more frequent involvement of thoracic lesions. However, the incidence of comorbid conditions remained similar across both groups. Subsequent adjustments for factors such as age, gender, BMI, existing comorbidities, and the presence of thoracic spine lesions were made using propensity scores. This propensity score matching revealed that the noted difference in the prevalence of carotid intima-media thickening remained significant even after these adjustments (Tables 2, 3).
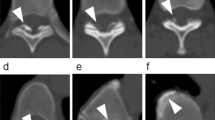
Illustrative case (Fig. 1).
A 56-year-old man with a body mass index (BMI) of 25.1 was diagnosed with a continuous type of cervical OPLL. a Sagittal (left) and axial images of cervical CT. The anteroposterior diameter of the OPLL at the C3/4 level was measured at 7.0 mm. b Ultrasound examination of the common carotid artery. A maximum intima-media thickness (IMT) was 2.7 mm (arrows)
A 56-year-old man with a body mass index (BMI) of 25.1 was diagnosed with a continuous type of cervical OPLL. The anteroposterior diameter of the OPLL at the C3/4 level was measured at 7.0 mm. Despite the absence of metabolic diseases such as diabetes mellitus, hyperlipidemia, or hypertension, ultrasound examination of the common carotid artery (CCA) revealed a maximum intima-media thickness (IMT) of 2.7 mm, and additionally, plaques were detected.
Discussion
In this study, the incidence of arteriosclerosis in the CCA as diagnosed by IMT ≥ 1.1 was 62.5% in myelopathic OPLL patients, considered high for the Japanese population. An epidemiological study on the Japanese population (mean age: 59.7 years) reported the incidence of IMT ≥ 1.1 mm at 24.4% [17]. In another study using the data of the Japanese population (average age: 58.5 years) from medical checkups, the proportion of patients with arteriosclerosis in the CCA was 17% in asymptomatic subjects with a relatively small OPLL and 13% without OPLL [3]. Although the average age was 63.5 years and IMT usually increased with age, the incidence of OPLL in this study was extremely high among symptomatic patients. Furthermore, patients with a larger cervical OPLL showed a higher frequency of intima-media thickening in the carotid artery even after adjusting for confounding factors such as age, sex, BMI, and comorbidities. Hence, our results could partly be elucidated by the crosstalk between OPLL and arteriosclerosis.
To date, the etiology of OPLL remains unclear. However, several previous studies have indicated an involvement of inflammatory cytokines in the pathogenesis of OPLL. For example, an experimental study reported higher expression levels of tumor necrosis factor-alpha (TNF-α) or interleukin-6 (IL-6) in ligament cells derived from ossified spinal ligaments [18]. Another study demonstrated a correlation of bone morphogenetic protein-2 and transforming growth factor-β with the pathogenesis of OPLL [15]. Moreover, the involvement of insulin-like growth factor I (IGF-I) has been reported in patients with OPLL [10, 14]. Besides, the involvement of the Wnt/β-catenin signaling pathway has been indicated in patients with OPLL, and a genome-wide association study of patients with OPLL identified RSPO2, which encodes R-spondin 2, as a susceptibility gene for OPLL [19]. Remarkably, it is well known that inflammatory cytokines also play vital roles in the pathogenesis of arteriosclerosis. For example, TNF-α and IL-6 significantly correlate with the incidence of cardiovascular events [20], whereas the increased levels of circulating inflammatory cytokines, including IL-6 and TNF-α, have also been reported [21]. Thus, we assume that these factors could play a crucial role as underlying mechanisms for both OPLL and arteriosclerosis.
This study demonstrated that patients with a larger cervical OPLL showed a higher frequency of intima-media thickening in the carotid artery even after adjusting for confounding factors such as age, sex, BMI, and comorbidities. A previous study reported that the maximum spinal canal stenosis rates were 38% and 27% in patients with and without myelopathy, respectively [6]. Another study from Japan revealed that the myelopathy-free rate in asymptomatic patients with cervical OPLL was 71% at 30 years [2]. Thus, OPLL is considered heterogeneous, and it would be reasonable to focus on patients, for example, in terms of the characteristic of OPLL or characteristics of patients to attain homogenous patients. For example, Endo et al. categorized patients with OPLL into four groups in terms of cervical or thoracic OPLL and based on conservative treatment or surgical treatment [22]; consequently, patients with thoracic OPLL who underwent surgical intervention exhibited a higher BMI both at the age of 20 years and at present compared with those in the other three groups. Doi et al. examined the bone mineral density (BMD) in patients with OPLL in comparison with cervical spondylosis [23], demonstrating that BMD was markedly higher in women but not in men. Hence, we believe in the necessity of classifying patients with OPLL in any form. Therefore, we established a cutoff value for the OPLL size to categorize patients, showing that those with larger OPLL were more likely to develop arteriosclerosis compared to those with smaller OPLL. However, it is important to note that the chosen cutoff value of 5 mm is not definitive.
Of note, several confounding factors should be addressed here. For example, about one-third of patients in this study were diabetic, which was relatively high considering the reported incidence of diabetes in Japan was 5.6% [24]. Thus, it would be reasonable to assume that IGF signaling or other cytokines might also play roles as common pathways for the pathogenesis of ossification of spinal ligaments, arteriosclerosis, and diabetes [10]. Likewise, the BMI of patients with OPLL in this study was 26.9 and considered higher for the Japanese population because an epidemiological study in Japan reported an average BMI of 22.7 kg/m2 [25]. Nevertheless, the higher BMI in our study sample aligned with previous studies [22]. While diabetes and obesity are known to be related to arteriosclerosis, our study found a higher incidence of arteriosclerosis in patients with larger OPLL, even after adjusting for BMI and comorbidities. This suggests a potential direct link in the pathogenesis between OPLL and arteriosclerosis. Nevertheless, further research is needed, including the inclusion of asymptomatic individuals without OPLL as a control group, to explore this connection more thoroughly.
This study has several limitations worth acknowledging. First, a key limitation of our study is the lack of a control group without OPLL. This is a significant oversight, as a control group would have provided essential baseline data for comparison, particularly in understanding the specific impact of OPLL on arteriosclerosis. Given the higher incidence of arteriosclerosis in our OPLL patient cohort, a comparison with asymptomatic individuals or those without OPLL would have helped discern whether this increased incidence was directly linked to OPLL or influenced by other factors like the notable prevalence of diabetes and obesity in our study group. Second, the sample size, though adequate considering the relatively rare condition of OPLL, was not large enough to definitively generalize our findings across broader populations. A larger sample would have allowed for a more robust statistical analysis and potentially more conclusive results. Third, while we adjusted for several known risk factors of arteriosclerosis, such as diabetes mellitus, hypertension, obesity, and age, there may be other unknown or unaddressed causes that could influence our results. Lastly, our reliance on imaging data for diagnosis and measurement, while standard, may carry inherent biases or limitations in accurately capturing the complexity of the conditions studied. Again, further studies are warranted to elucidate these problems. However, with all the limitations, we believe that the findings of this study will be informative for clinicians in anticipating and preventing comorbidities for patients with OPLL.
Conclusions
Patients with a larger cervical OPLL showed a higher frequency of atherosclerosis in the carotid artery.
Availability of data and material
The manuscript has no associated data.
References
Fujimori T, Watabe T, Iwamoto Y, Hamada S, Iwasaki M, Oda T (2016) Prevalence, concomitance, and distribution of ossification of the spinal ligaments: results of whole spine CT scans in 1500 Japanese patients. Spine (Phila Pa 1976) 41:1668–1676. https://doi.org/10.1097/BRS.0000000000001643
Matsunaga S, Kukita M, Hayashi K, Shinkura R, Koriyama C, Sakou T, Komiya S (2002) Pathogenesis of myelopathy in patients with ossification of the posterior longitudinal ligament. J Neurosurg 96:168–172
Oshima Y, Doi T, Kato S, Taniguchi Y, Matsubayashi Y, Nakajima K, Oguchi F, Oka H, Hayashi N, Tanaka S (2020) Association between ossification of the longitudinal ligament of the cervical spine and arteriosclerosis in the carotid artery. Sci Rep 10:3369. https://doi.org/10.1038/s41598-020-60248-3
Terayama K (1989) Genetic studies on ossification of the posterior longitudinal ligament of the spine. Spine (Phila Pa 1976) 14:1184–1191
Yoshimura N, Nagata K, Muraki S, Oka H, Yoshida M, Enyo Y, Kagotani R, Hashizume H, Yamada H, Ishimoto Y, Teraguchi M, Tanaka S, Kawaguchi H, Toyama Y, Nakamura K, Akune T (2014) Prevalence and progression of radiographic ossification of the posterior longitudinal ligament and associated factors in the Japanese population: a 3-year follow-up of the ROAD study. Osteoporos Int 25:1089–1098. https://doi.org/10.1007/s00198-013-2489-0
Matsunaga S, Sakou T (2012) Ossification of the posterior longitudinal ligament of the cervical spine: etiology and natural history. Spine (Phila Pa 1976) 37:E309-314. https://doi.org/10.1097/BRS.0b013e318241ad33
Moon BJ, Choi SK, Shin DA, Yi S, Kim KN, Yoon DH, Ha Y (2018) Prevalence, incidence, comorbidity, and mortality rates of ossification of posterior longitudinal ligament in the cervical spine: a nested case-control cohort study. World Neurosurg 117:e323–e328. https://doi.org/10.1016/j.wneu.2018.06.023
Shin J, Choi JY, Kim YW, Chang JS, Yoon S (2019) Quantification of risk factors for cervical ossification of the posterior longitudinal ligament in Korean populations: a nationwide population-based case-control study. Spine (Phila Pa 1976). https://doi.org/10.1097/BRS.0000000000003027
Stapleton CJ, Pham MH, Attenello FJ, Hsieh PC (2011) Ossification of the posterior longitudinal ligament: genetics and pathophysiology. Neurosurg Focus 30:E6. https://doi.org/10.3171/2010.12.FOCUS10271
Akune T, Ogata N, Seichi A, Ohnishi I, Nakamura K, Kawaguchi H (2001) Insulin secretory response is positively associated with the extent of ossification of the posterior longitudinal ligament of the spine. J Bone Joint Surg Am 83:1537–1544. https://doi.org/10.2106/00004623-200110000-00013
Kobashi G, Washio M, Okamoto K, Sasaki S, Yokoyama T, Miyake Y, Sakamoto N, Ohta K, Inaba Y, Tanaka H, Japan Collaborative Epidemiological Study Group for Evaluation of Ossification of the Posterior Longitudinal Ligament of the Spine R (2004) High body mass index after age 20 and diabetes mellitus are independent risk factors for ossification of the posterior longitudinal ligament of the spine in Japanese subjects: a case-control study in multiple hospitals. Spine (Phila Pa 1976) 29:1006–1010. https://doi.org/10.1097/00007632-200405010-00011
Chaput CD, Siddiqui M, Rahm MD (2019) Obesity and calcification of the ligaments of the spine: a comprehensive CT analysis of the entire spine in a random trauma population. Spine J. https://doi.org/10.1016/j.spinee.2019.03.003
Endo T, Takahata M, Koike Y, Fujita R, Suzuki R, Hisada Y, Hasegawa Y, Suzuki H, Yamada K, Iwata A, Sudo H, Yoneoka D, Iwasaki N (2022) Association between obesity and ossification of spinal ligaments in 622 asymptomatic subjects: a cross-sectional study. J Bone Miner Metab 40:337–347. https://doi.org/10.1007/s00774-021-01292-5
Goto K, Yamazaki M, Tagawa M, Goto S, Kon T, Moriya H, Fujimura S (1998) Involvement of insulin-like growth factor I in development of ossification of the posterior longitudinal ligament of the spine. Calcif Tissue Int 62:158–165
Kawa-Uchi T, Furuya K, Shinomiya K, Yama-Ura I, Kurosa Y, Wozney JM, Ueno N, Noda M (1996) Messenger RNA expression of the genes encoding receptors for bone morphogenetic protein (BMP) and transforming growth factor-beta (TGF-beta) in the cells from the posterior longitudinal ligament in cervical spine. Endocrine 5:307–314. https://doi.org/10.1007/BF02739064
Tsuyama N (1984) Ossification of the posterior longitudinal ligament of the spine. Clin Orthop Relat Res 184:71–84
Kokubo Y, Watanabe M, Higashiyama A, Nakao YM, Nakamura F, Miyamoto Y (2018) Impact of intima-media thickness progression in the common carotid arteries on the risk of incident cardiovascular disease in the suita study. J Am Heart Assoc. https://doi.org/10.1161/JAHA.117.007720
Ren L, Hu H, Sun X, Li F, Zhou JJ, Wang YM (2013) The roles of inflammatory cytokines in the pathogenesis of ossification of ligamentum flavum. Am J Transl Res 5:582–585
Nakajima M, Kou I, Ohashi H, Genetic Study Group of the Investigation Committee on the Ossification of Spinal L, Ikegawa S (2016) Identification and functional characterization of RSPO2 as a susceptibility gene for ossification of the posterior longitudinal ligament of the spine. Am J Hum Genet 99:202–207. https://doi.org/10.1016/j.ajhg.2016.05.018
Cesari M, Penninx BW, Newman AB, Kritchevsky SB, Nicklas BJ, Sutton-Tyrrell K, Tracy RP, Rubin SM, Harris TB, Pahor M (2003) Inflammatory markers and cardiovascular disease (The Health, Aging and Body Composition [Health ABC] Study). Am J Cardiol 92:522–528. https://doi.org/10.1016/s0002-9149(03)00718-5
Krabbe KS, Pedersen M, Bruunsgaard H (2004) Inflammatory mediators in the elderly. Exp Gerontol 39:687–699. https://doi.org/10.1016/j.exger.2004.01.009
Endo T, Takahata M, Koike Y, Iwasaki N (2020) Clinical characteristics of patients with thoracic myelopathy caused by ossification of the posterior longitudinal ligament. J Bone Miner Metab 38:63–69. https://doi.org/10.1007/s00774-019-01026-8
Doi T, Ohashi S, Ohtomo N, Tozawa K, Nakarai H, Yoshida Y, Ito Y, Sakamoto R, Nakajima K, Nagata K, Okamoto N, Nakamoto H, Kato S, Taniguchi Y, Matsubayashi Y, Tanaka S, Oshima Y (2022) Evaluation of bone strength using finite-element analysis in patients with ossification of the posterior longitudinal ligament. Spine J. https://doi.org/10.1016/j.spinee.2022.02.018
Kohsaka S, Morita N, Okami S, Kidani Y, Yajima T (2021) Current trends in diabetes mellitus database research in Japan. Diabetes Obes Metab 23(Suppl 2):3–18. https://doi.org/10.1111/dom.14325
Yoshimura N, Akune T, Fujiwara S, Shimizu Y, Yoshida H, Nishiwaki Y, Sudo A, Omori G, Yoshida M, Shimokata H, Suzuki T, Muraki S, Oka H, Nakamura K (2015) Incidence of disability and its associated factors in Japanese men and women: the longitudinal cohorts of motor system organ (LOCOMO) study. J Bone Miner Metab 33:186–191. https://doi.org/10.1007/s00774-014-0573-y
Funding
Open Access funding provided by The University of Tokyo. This study was not supported by any kind of funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No benefits in any form have been or will be received from a commercial party related directly or indirectly to the subject of this manuscript. The manuscript submitted does not contain information about medical device (s)/drug (s).
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee (Institutional Review Board of the Clinical Research Support Center of the University of Tokyo Hospital) and with the 1964 Helsinki Declaration.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Oshima, Y., Doi, T., Ohtomo, N. et al. Patients with a large ossification of the posterior longitudinal ligament have a higher incidence of arteriosclerosis in the carotid artery. Eur Spine J 33, 379–385 (2024). https://doi.org/10.1007/s00586-023-08107-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-023-08107-3