Abstract
Purpose
This paper sets out to analyse mobility changes in segments adjacent to the operated segment. Additionally, it investigates the relationship between the degree of fusion in the operated disc space and mobility changes in the adjacent segments.
Methods
In total, 170 disc spaces were operated on in 104 consecutive patients qualified for one- or two-level surgery. The degree of mobility of segments directly above and below the implant insertion site was calculated. Measurements were performed the day before the surgery and 12 months post-surgery. Functional (flexion and extension) radiographs of the cervical spine and CT scans obtained 12 months post-surgery were used to evaluate the fusion status. The results were subjected to statistical analysis.
Results
Statistically significant increase in mobility was recorded for the segments situated immediately below the operative site, with a mean change in mobility of 1.7 mm. Complete fusion was demonstrated in 101 cases (71.1%), and partial fusion in 43 cases (29.9%). In the complete fusion subgroup, the ranges of both flexion and extension in the segments directly below the operative site were significantly greater than those in the partial fusion (pseudoarthrosis) subgroup.
Conclusion
The mobility of the adjacent segment below the implant insertion site was significantly increased at 12 months post-ACDF surgery. The range of this compensatory hypermobility was significantly greater in patients with complete fusion at the ACDF site than in cases of pseudoarthrosis. Implant subsidence was not associated with mobility changes in the segments directly above or directly below the site of ACDF surgery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Anterior cervical discectomy and fusion (ACDF), apart from a discectomy to decompress neural structures, aims to obtain bone union within the operated disc space. Biomechanically, if mobility is abolished within a disc space, the adjacent motion segments below and above the operated segment should partly take over the mobility of the non-mobile segment. The pathophysiology underlying the disease entity referred to as adjacent segment disease (ASD) has been explained in terms of increased load on the motion segments adjacent to the operated segment that has been blocked/fused [1, 2]. ASD depends on a number of factors, being the combined result of natural degenerative processes and biomechanical changes associated with fusion within the original motion segment operated on, such as alterations of the ranges of motion of the adjacent segments, changes in the sagittal profile of the spine, and increased intradiscal pressure in the adjacent discs [3]. While these phenomena are encountered in daily clinical practice, there is still a paucity of research papers representing detailed analyses of changes in the mobility of segments adjacent to the operated segment(s). This paper thus sets out to analyse mobility changes in segments adjacent to the operated segment. Additionally, it investigates the relationship between the degree of fusion in the operated disc space and mobility changes in the adjacent segments, and the relationship between implant subsidence and mobility changes in the adjacent segments.
Material and methods
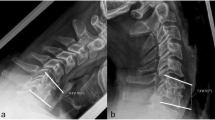
In total, 170 disc spaces were operated on in 104 patients, who were qualified for one- or two-level surgery (age: 51.2 ± 10.3; females 73.1%). The operations included a discectomy, removal of osteophytes, transection of the posterior longitudinal ligament, and decompression of neural structures. Those were followed by insertion of an interbody implant (cage), which was always filled with nanoparticle hydroxyapatite. The degree of mobility of segments directly above and below the implant insertion site was calculated based on changes in the distance between the bases of spinous processes in flexion and extension. Measurements were performed on functional radiographs of the cervical spine obtained on the day before the surgery and at 12 months post-surgery. The procedure for the measurements is explained in Fig. 1. Measurements were made on the basis of radiographs obtained in one X-ray centre, following exactly the same procedure (distance and patient’s position) and utilising the same equipment and software. Measurements were taken at the upper border of the base of spinous processes as this anatomical detail is appropriately visualised in most radiographs. We believe that this anatomic landmark ensures appropriate reproducibility of the measurements and leaves no uncertainty concerning the location of the point of measurement. The mobility of a motion segment was defined as the change in the distance between flexion and extension in millimetres. The measurements were noted down with an accuracy of 0.1 mm. We used Infinitt Healthcare software to record, view, and analyse the radiographs. Analysis of radiographs was performed by 3 individuals (2 neurosurgeons and 1 orthopaedist) who are among the authors of this paper. The radiographic studies were analysed jointly, and a final assessment was made (a numerical value was entered) taking into account the opinion of each participant.
Measurement of mobility of motion segment immediately above and below the implant insertion site. Functional radiograph at 12 months post-surgery. A Flexion. B Extension. ACDF surgery had been performed on the C5/C6 disc space. Distances were measured between the bases of the C4-C5 and C6-C7 spinous processes. The mobility of a motion segment was defined as the change in the distance between flexion and extension in millimetres. The mobility of the segments directly above and below the implant insertion site was also measured in this way in radiographs obtained on the day before the surgery
The measurement data were used to document changes in mobility of the segments adjacent to the implant insertion site at 12 months after the surgery compared to pre-operative mobility. We also investigated the relationship between the degree of fusion of the operated disc space and change in the mobility of the adjacent segments to find out whether residual motion at the arthrodesis site would reduce compensatory hypermobility of the adjacent segments. Fusion was assessed according to an original method also employed in our previous research [4, 5]. Functional (flexion and extension) radiographs of the cervical spine and CT scans obtained 12 months after the surgery were used to evaluate the fusion status. It was classified as 1) complete fusion, 2) partial fusion, and 3) absence of fusion. The classification criteria are presented in Table 1. Figure 2 shows examples of cases classified as complete and partial fusion on CT scans.
Sample presentations of complete and partial fusion on CT scans at 12 months post-surgery: A complete fusion (A1—sagittal view, A2—transverse view at the level of the implant in C6/C7 disc space). B partial fusion (B1—sagittal view, B2—transverse view at the level of the implant in C4/C5 disc space. Reprinted by permission from Springer Nature: Acta Neurochir (Wien) 2022;164 (6):1501–1507. PEEK versus titanium-coated PEEK cervical cages: fusion rate. Godlewski B, Bebenek A, Dominiak M, Karpinski G, Cieslik P, Pawelczyk T
Furthermore, we investigated the relationship between implant subsidence and change in mobility of the adjacent motion segments. Implant subsidence was evaluated over 12 months post-surgery. Measurements of the height of interbody spaces in the centre of the vertebral bodies were taken with an accuracy of 0.1 mm, to determine the distance between the endplates of adjacent vertebral bodies. The radiographic indices were assessed in five different time instances: 1) before surgical procedure, 2) one day after surgery, 3) one month following the surgery, 4) six months post-surgery, and 5) one year post-surgery. Subsidence was found if the implant was displaced ≥ 3 mm into the adjacent endplates in reference to radiographs collected one day post-surgery. The relationship between mobility of the segments adjacent to the implant insertion site and patient age was also investigated. Consent for the study was obtained from the relevant ethical review board (Resolution 4/2019 of the Bioethics Committee at Andrzej Frycz Modrzewski Cracow University in Cracow of 24 January 2019).
Statistical analysis
Standard descriptive statistics were used to describe baseline characteristics of the patients. The distribution of the continuous variables was assessed using the D'Agostino–Pearson test for normal distribution. Parametric statistical methods were used for the analysis of differences and relationships between variables. The independent samples t test was used to assess differences between independent groups in the case of continuous or ordinal variables. For paired-group comparisons of continuous or ordinal variables, the paired samples t test was used to assess changes in mobility across time. Logarithmic transformation was used to correct for the deviation from the normal distribution to meet the assumption of parametric tests. The central tendency of the normally distributed variables is presented in tables as the arithmetic mean and the variation as standard deviation. In case of non-normally distributed variables, central tendency is presented as the geometric mean back-transformed after logarithmic transformation and variability is shown as 95% confidence intervals. For the variables that needed transformation, Pearson's correlation coefficient was used to assess relationships between non-normally distributed continuous variables. A two-sided P < 0.05 was considered statistically significant.
Results
The measurements were performed in conventional radiographs and CT scans obtained over 12 months of follow-up. At 12 months post-op, CT scans were available from 86 patients (a total of 144 disc spaces assessed), and conventional radiographs were available from 101 patients (166 disc spaces). As the method we employed for classifying fusion status relied on simultaneous evaluation of CT scans and radiographs, the final sample for evaluating fusion comprised 144 disc spaces, while implant subsidence and changes in adjacent segment mobility were evaluated in a total of 166 disc spaces. Measurements of change in adjacent segment mobility could not be performed in some radiographs because the anatomical landmark of upper edge of the base of the spinous process could not be visualised. Furthermore, the study group included some patients who had only plain A-P and lateral radiographs of the cervical spine performed before the surgery without functional radiographs in flexion and extension so that it was not possible to determine changes in mobility by comparing pre- and post-operative evidence. The changes in mobility noted for the segments situated immediately above the operative site/spondylodesis site at 12 months post-op compared to the pre-operative baseline were not significant. While mobility did increase (by 0.6 mm on average), the differences did not reach statistical significance. At the same time, a statistically significant increase in mobility was recorded for the segments situated immediately below the operative site/spondylodesis site, with a mean change in mobility for these segments of 1.7 mm. These results are presented in Table 2 and Fig. 3.
Mobility differences between extension and flexion for vertebral segments directly above and below the implant insertion site at baseline and 12 months after the surgical procedure. Bars represent means; whiskers represent standard deviations. Symbols and * indicate significance level p < 0.05; ns —not significant, mm—millimetres
The relationship between complete fusion and mobility changes in the segments directly above and below the implant insertion site was also assessed. Complete fusion was demonstrated in 101 cases (71.1%), and partial fusion in 43 cases (29.9%). There were no cases of absence of fusion. In the complete fusion subgroup, the ranges of both flexion and extension in the segments directly below the operative site were greater as compared to the partial fusion (pseudoarthrosis) subgroup. These results are presented in Table 3.
An analysis of the relationship between patient age and the mobility of the segments directly above the implant insertion site found a statistically significant negative correlation, indicating that, at 12 months post-surgery, mobility of the segments directly above (Pearson’s correlation coefficient r = − 0.3149; p = 0.0143) the implant insertion site decreased with patient age. No significant correlation was found between the segments directly above the implant insertion and patient age (Pearson’s r = − 0.2124; p = 0.2). The decrement in mobility was progressively smaller with increasing age of the patients. Pre-operatively, there were no significant correlations between the mobility of the segments directly above (Pearson’s r = − 0.225; p = 0.084) or below (Pearson’s r = − 0.299; p = 0.065) the implant insertion site and the age of the patients. The relationship between implant subsidence and changes in the segments directly below and above the implant insertion site was also analysed, but no significant correlations were revealed. These results can be found in Table 4. Subsidence was found in 35 disc spaces, representing 21% of the 166 data sets.
Discussion
Adjacent segment disease is defined as new degenerative changes at a spinal level adjacent to the surgically treated level/levels in the spine, accompanied by symptoms such as radiculopathy, myelopathy, or instability. The theoretical underpinnings of adjacent segment disease remain controversial. One approach states that some individuals exhibit a predisposition to develop osteoarthritis of the spine, and other segments, including adjacent segments, will become involved regardless of surgery [6, 7]. On the other hand, there is evidence to suggest that ASD does develop following surgery. ACDF produces good outcomes, but it must be borne in mind that approximately 25% of ACDF patients will present with symptomatic ASD within 10 years of the index surgery [8,9,10,11,12]. The most accurate approach appears to be that considering ASD to be a multifactorial condition being the combined result of natural degenerative processes and biomechanical changes associated with fusion within the original motion segment operated on, such as alterations of the ranges of motion of the adjacent segments, changes in the sagittal profile of the spine, and increased intradiscal pressure in the adjacent discs [3, 13, 14]. ASD differs from adjacent segment degeneration, which may be asymptomatic in a large percentage of elderly patients [15, 16]. Our results indicate a significant increase in mobility in the segments situated directly below the implant insertion site. This constitutes a confirmation of the hypothesis of compensatory nature of adjacent segment hypermobility associated with the presence of fusion at the implant insertion site. Moreover, our study indicates greater compensatory hypermobility in the presence of complete fusion than in the presence of a pseudoarthrosis. Hypermobility, defined as significant differences in mobility before vs after the surgery, was detected in the segments situated directly below the implant insertion site. This may promote the development of adjacent segment disease at these locations in the future. As stated earlier, we believe, as stated earlier, that ASD is a multifactorial condition where increased mobility of the adjacent segments is just one factor. However, Nabhan A et al. found no significant difference of the segmental motion of the adjacent level, treated with either prostheses or fusion, 1 year after surgery, despite evidence of preserved mobility of the operated motion segment following artificial disc insertion [17]. Maldano et al. studied the prevalence of ASD with regard to a surgical history of ACDF vs disc arthroplasty. They demonstrated that preservation of the mobility of the operated segment with an artificial disc did not lead to lower rates of symptomatic ASD compared to ACDF surgery [18]. They believe that other factors are also at play and influence the development of ASD. Similar results were obtained by Yang X et al. who concluded that maintaining the range of motion after cervical discectomy with an artificial disc does not prevent adjacent segment degeneration [2]. On the other hand, a metaanalysis by Luo I et al. found a significantly lower rate of ASD in a cervical disc arthroplasty group than in an ACDF group and additionally observed that the rate of revision surgery was also lower in the cervical disc arthroplasty group compared with the ACDF group [19]. Biomechanical cadaver studies showed that the presence of fusion in a motion segment contributes to increased mobility (termed compensatory hypermobility) of the adjacent motion segments, itself influencing disc loading (increased intradiscal pressure) and the onset of ASD [1, 20,21,22,23]. Increased intradiscal pressure/disc loading induces biochemical changes within the disc. The intervertebral disc is dependent on nutrients diffusing through the extracellular matrix from peripheral blood vessels and vertebral endplates since it lacks a proper blood supply. Increased intradiscal pressure acts to alter the diffusion characteristics of nutrients from the periphery and leads to an accumulation of waste products in the disc. Failure to remove waste products adequately from the disc can lead to increased lactate levels and decreased pH, which can impair metabolism and lead to cell death [24]. Additionally, increased compressive forces over time have been reported to increase the presence of collagen Type 1 and to decrease proteoglycans, chondroitin sulphate, and collagen Type 2. All of these changes lead to disc degeneration and occur during the standard process of ageing [25]. Most clinical research investigating mobility in adjacent motion segments and the development of adjacent segment disease is based on manual measurements of ranges of motion on static sagittal images at maximum flexion and extension. Chen SR et al. used digital diagnostic imaging technology to increase measurement accuracy and improve its reliability: biplane radiographs were taken at a rate of thirty images per second for three seconds during flexion–extension and axial rotation movements. They studied the impact of surgery-related factors, such as the position for insertion of a cervical plate, type of implant, post-operative kyphosis of the operated segment, and the degree of distraction of the operated disc space, on changes in adjacent segment mobility both in the flexion–extension plane and in the axial plane. They found increased mobility in both analyses: mean change in flexion/extension in the segment immediately above the implant insertion site was 0.7° (range − 5.4° to 6.5°) compared to 1.2° (range − 11.3° to 11.2°) in the segment directly below. Mean change in the axial range of motion was 0.7° (− 2.8° to 5.5°) for the segment above and 0.7° (− 2.0° to 4.2°) for the segment below. Statistical analysis revealed that the surgery-related factors listed above did not influence the observed changes in adjacent segment mobility [12]. Alhashas M et al. analysed ACDF patients who developed symptomatic adjacent segment disease. The highest rate of ASD (7.9%) was associated with one-level procedures, compared to a rate of 6% following two-level procedures, 2.8% following three-level procedures, and no cases of symptomatic ASD requiring revision surgery following a baseline four-level procedure. With regard to disc levels, the highest rates of revision surgery for symptomatic ASD were noted for C5/C6 (28%) and C4/C5 (25%). The mean interval between the baseline ACDF procedure and surgery for ASD was 32 months (range 12–80 months) [3]. The limitations of our study comprise a relatively short follow-up period, analysis being limited to one- and two-level procedures, and that possible non-applicability of our results to longer fusions. The pre-operative range of motion may be limited because of pain that had been present before the surgery and might have affected the results. We cannot exclude the possibility that parallax effect could have affected the measurements we made from flexion and extension images. We initially considered several techniques for measuring changes in mobility and looked at a number of relevant papers. We considered using the change in Cobb’s angle as a parameter. We understand that any change in scale of the X-ray alters the linear measurement, while an angle measurement remains unchanged by scale. Finally, we decided to base our assessment on the distance between the bases of spinous processes. We believe that this anatomic landmark ensures appropriate reproducibility of the measurements and leaves no uncertainty concerning the location of the point of measurement. Also of importance, sagittal mobility, which we measured, accounts for approx. 75% of daily mobility, with the remaining 25% being associated with axial motion, which were not measured with regard to change after ACDF surgery [12, 26, 27].
Conclusion
The mobility of the adjacent segment below the implant insertion site was significantly increased at 12 months post-ACDF surgery. The range of this compensatory hypermobility was greater in patients with complete fusion at the ACDF site than in cases of pseudoarthrosis. Implant subsidence was not associated with mobility changes in the segments directly above or directly below the site of ACDF surgery.
Data availability
The datasets supporting results of this study are available from the corresponding author on reasonable request.
References
Schwab JS, Diangelo DJ, Foley KT (2006) Motion compensation associated with single-level cervical fusion: where does the lost motion go? Spine 31(21):2439–2448. https://doi.org/10.1097/01.brs.0000239125.54761.23
Yang X, Donk R, Arts MP, Arnts H, Walraevens J, Zhai Z, Depreitere B, Bartels RHMA, Vleggeest-Lankamp CLA (2019) Maintaining range of motion after cervical discectomy does not prevent adjacent segment degeneration. Spine J 19(11):1816–1823. https://doi.org/10.1016/j.spinee.2019.07.011
Alhashash M, Shousha M, Boehm H (2018) Adjacent segment disease after cervical spine fusion: evaluation of a 70 patient long-term follow-up. Spine 43(9):605–609. https://doi.org/10.1097/brs.0000000000002377
Godlewski B, Bebenek A, Dominiak M, Karpinski G, Cieslik P, Pawelczyk T (2022) PEEK versus titanium-coated PEEK cervical cages: fusion rate. Acta Neurochir (Wien) 164(6):1501–1507. https://doi.org/10.1007/s00701-022-05217-7
Godlewski B, Bebenek A, Dominiak M, Bochniak M, Cieslik P, Pawelczyk T (2022) Reliability and utility of various methods for evaluation of bone union after anterior cervical discectomy and fusion. J Clin Med 11(20):6066. https://doi.org/10.3390/jcm11206066
Battié MC, Videman T, Kaprio J, Gibbons LE, Gill K, Manninen H, Saarela J, Peltonen L (2009) The twin spine study: contributions to a changing view of disc degeneration. Spine J 9(1):47–59. https://doi.org/10.1016/j.spinee.2008.11.011
Seo M, Choi D (2008) Adjacent segment disease after fusion for cervical spondylosis; myth or reality? Br J Neurosurg 22(2):195–199. https://doi.org/10.1080/02688690701790605
Cho SK, Riew KD (2013) Adjacent segment disease following cervical spine surgery. J Am Acad Orthop Surg 21(1):3–11. https://doi.org/10.5435/jaaos-21-01-3
Hilibrand AS, Carlson GD, Palumbo MA, Jones PK, Bohlman HH (1999) Radiculopathy and myelopathy at segments adjacent to the site of a previous anterior cervical arthrodesis. J Bone Joint Surg Am 81(4):519–528. https://doi.org/10.2106/00004623-199904000-00009
Latka D, Kozlowska K, Miekisiak G, Latka K, Chowaniec J, Olbrycht T, Latka M (2019) Safety and efficacy of cervical disc arthroplasty in preventing the adjacent segment disease: a meta-analysis of mid- to long-term outcomes in prospective, randomized, controlled multicenter studies. Ther Clin Risk Manag 15:531–539. https://doi.org/10.2147/tcrm.s196349
Goffin J, Geusens E, Vantomme N, Quintens E, Waerzeggers Y, Depreitere B, van Calanbergh F, van Loon J (2004) Long-term follow-up after interbody fusion of the cervical spine. J Spinal Disord Tech 17(2):79–85. https://doi.org/10.1097/00024720-200404000-00001
Chen SR, LeVasseur CM, Pitcairn S, Kanter AS, Okonkwo DO, Shaw JD, Donaldson WF, Lee JY, Anderst WJ (2021) Surgery-related factors do not affect short-term adjacent segment kinematics after anterior cervical arthrodesis. Spine 46(23):1630–1636. https://doi.org/10.1097/brs.0000000000004080
Godlewski B, Stachura MK, Czepko RA, Banach M, Czepko R (2018) Analysis of changes in cervical spinal curvature and intervertebral disk space height following ACDF surgery in a group of 100 patients followed up for 12 months. J Clin Neurosci 52:92–99. https://doi.org/10.1016/j.jocn.2018.04.005
Yang X, Bartels RHMA, Donk R, Arts MP, Goedmakers CMW, Vleggeert-Lankamp CLA (2020) The association of cervical sagittal alignment with adjacent segment degeneration. Eur Spine J 29(11):2655–2664. https://doi.org/10.1007/s00586-019-06157-0
Boden SD, McCowin PR, Davis DO, Dina TS, Mark AS, Wiesel S (1990) Abnormal magnetic-resonance scans of the cervical spine in asymptomatic subjects. A prospective investigation. J Bone Joint Surg Am 72(8):1178–1184
Gore DR (2001) Roentgenographic findings in the cervical spine in asymptomatic persons: a ten-year follow-up. Spine 26(22):2463–2466. https://doi.org/10.1097/00007632-200111150-00013
Nabhan A, Ishak B, Steudel WI, Ramadhan S, Steimer O (2011) Assessment of adjacent-segment mobility after cervical disc replacement versus fusion: RCT with 1 year’s results. Eur Spine J 20(6):934–941. https://doi.org/10.1007/s00586-010-1588-2
Maldonado CV, Paz RD, Martin CB (2011) Adjacent-level degeneration after cervical disc arthroplasty versus fusion. Eur Spine J 20(Suppl 3):403–407. https://doi.org/10.1007/s00586-011-1916-1
Luo J, Wang H, Peng J, Deng Z, Zhang Z, Liu S, Wang D, Gong M, Tang S (2018) Rate of adjacent segment degeneration of cervical disc arthroplasty versus fusion meta-analysis of randomized controlled trials. World Neurosurg 113:225–231. https://doi.org/10.1016/j.wneu.2018.02.113
Eck JC, Humphreys SC, Lim TH, Jeong ST, Kim JG, Hodges SD, An HS (2002) Biomechanical study on the effect of cervical spine fusion on adjacent-level intradiscal pressure and segmental motion. Spine 27(22):2431–2434. https://doi.org/10.1097/00007632-200211150-00003
Matsunaga S, Kabayama S, Yamamoto T, Yone K, Sakou T, Nakanishi K (1999) Strain on intervertebral discs after anterior cervical decompression and fusion. Spine 24(7):670–675. https://doi.org/10.1097/00007632-199904010-00011
Prasarn ML, Baria D, Milne E, Latta L, Sukovich W (2012) Adjacent-level biomechanics after single versus multilevel cervical spine fusion. J Neurosurg Spine 16(2):172–177. https://doi.org/10.3171/2011.10.spine11116
Park J, Shin JJ, Lim J (2014) Biomechanical analysis of disc pressure and facet contact force after simulated two-level cervical surgeries (fusion and arthroplasty) and hybrid surgery. World Neurosurg 82(6):1388–1393. https://doi.org/10.1016/j.wneu.2014.06.013
Buckwalter JA (1995) Aging and degeneration of the human intervertebral disc. Spine 20(11):1307–1314. https://doi.org/10.1097/00007632-199506000-00022
Hutton WC, Toribatake Y, Elmer WA, Ganey TM, Tomita K, Whitesides TE (1998) The effect of compressive force applied to the intervertebral disc in vivo. A study of proteoglycans and collagen. Spine 23(23):2524–2537. https://doi.org/10.1097/00007632-199812010-00007
Boselie TF, van Mameren H, de Bie RA, van Santbrink H (2015) Cervical spine kinematics after anterior cervical discectomy with or without implantation of a mobile cervical disc prosthesis; an RCT. BMC Musculoskelet Disord 16:34. https://doi.org/10.1186/s12891-015-0479-4
Cobian DG, Sterling AC, Anderson PA, Heiderscheit BC (2009) Task-specific frequencies of neck motion measured in healthy young adults over a five-day period. Spine 34(6):E202–E207. https://doi.org/10.1097/brs.0b013e3181908c7b
Funding
This research received no external funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
This study obtained ethical approval from the institutional ethics committee of Andrzej Frycz Modrzewski Cracow University in Cracow (Resolution 4/2019) and was conducted in compliance with the Declaration of Helsinki. Written informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Godlewski, B., Bebenek, A., Dominiak, M. et al. Adjacent segment mobility after ACDF considering fusion status at the implant insertion site. Eur Spine J 32, 1616–1623 (2023). https://doi.org/10.1007/s00586-023-07634-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-023-07634-3