Abstract
Background
According to published meta-analyses, cervical total disc replacement (CTDR) seems to be superior to anterior cervical decompression and fusion (ACDF) in most clinical parameters. Despite short-term clinical success of CTDR, there are concerns regarding long-term durability of these prostheses.
Methods
This prospective study involved 382 patients who received standalone CTDR or a hybrid procedure (ACDF/CTDR). A retrospective comparison between different CTDR devices was conducted regarding patient-reported outcome measures (PROMs), failure scenarios, and revision surgeries. The M6-C™ Artificial Cervical Disc (Orthofix, Lewisville, Texas) cohort was compared to the other CTDR devices clinically. Etiological reasons for revision, and the surgical technique of the revision was investigated.
Results
Fifty-three patients received M6-C CTDR. Eighteen patients (34%) were revised at an average of 67 months postoperatively for wear-induced osteolysis. There were three additional cases of pending revision. The PROMs of the two groups were similar, indicating that the failure mode (wear-induced osteolysis) is often asymptomatic. The demographics of the two groups were also similar, with more women undergoing revision surgery than men. There were three one-level CTDR, four two-level hybrids, seven three-level hybrids, and three four-level hybrids revised anteriorly. Sixteen patients underwent removal of the prosthesis and were treated according to the extent of osteolysis. There were four vertebrectomies, six revisions to ACDF, and six revisions to another CTDR. One patient underwent supplemental fixation using a posterior approach. The other CTDR cohort had an incidence of 3.3% at the equivalent time, and none of these were due to osteolysis or wear-related events.
Conclusions
There is a concerning midterm failure rate related to ultra-high-molecular-weight-polyethylene wear-induced osteolysis in the M6-C. Patients implanted with the M6-C prosthesis should be contacted, informed, and clinically and radiologically assessed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Surgical management of symptomatic cervical degenerative disc disease (DDD) includes anterior cervical decompression and fusion (ACDF) or cervical total disc replacement (CTDR), and posterior foraminotomy [1]. These treatment modalities have been investigated extensively in clinical trials and are effective in reducing radicular pain, with CTDR having a lower revision and secondary surgery rate [2,3,4,5,6,7,8,9,10,11]. Data generated from multiple prospective randomised controlled studies comparing CTDR to ACDF have led to Food and Drug Administration (FDA) approval for eight devices [12]. These studies were industry sponsored, raising concerns of bias. A Cochrane systematic review comparing research sponsored by industry found that the treatment benefits were more likely to favour the sponsor’s products, and the authors’ conclusions were more favourable. Noting that the data collected were closely audited by the FDA, this potential issue should be mitigated. Meta-analyses have been carried out on these clinical trials to synthesise the results for comparison [13]. According to these meta-analyses, CTDR seems to be superior to ACDF with regard to most clinical parameters [14, 15]. Given the short-term clinical success of CTDR around the world, criticisms in the literature regarding long-term durability of these prostheses resulted in publications of midterm results [16,17,18].
The successful surgical treatment of DDD with CTDR requires collaboration between surgeons, engineers, and scientists to provide an evidence-based approach to optimise long-term outcomes. Each depends on the other for success, and the patient depends on all for success. Biomechanical simulations indicated high durability across the tested implants [19]. Multi-axis testing machines have combined in vivo kinematics and loading with in vitro testing in six degrees of freedom to offer more accurate predictions of the performance of new spinal instrumentation [20,21,22]. Initially there were no reported device failures among the identified studies. Implants were tested from 10 to 20 million cycles in axial loading, flexion and extension, and lateral bending. Optimal testing should be performed for up to 40 million cycles. During an average life, the spine may undergo 100 million cycles. No device failures were reported during these testing cycles and it has been suggested that this is equivalent to 50–100 years of in vivo wear [23, 24].
There are several biomechanical requirements for CTDR, perhaps the most important is maintenance of physiological motion. Studies have shown negative patient-reported outcome measures (PROMs) regarding hypomobility and hypermobility [25, 26]. Depending on the implant type, prostheses need to be able to tolerate load and minimise friction and wear. The device endplates must provide osteointegration qualities with bone endplates. These prostheses must be durable over a patient’s lifetime. Reeks and Liang state that total disc replacement (TDR) introduces unique problems that are separate from interbody fusion, including hypermobility, hypomobility, material wear, and particulate debris [27, 28].
The M6-C™ Artificial Cervical Disc (Orthofix, Lewisville, Texas) (M6-C) was designed as an innovative option for patients needing CTDR as an alternative to ACDF. The M6-C device incorporated a compressible polycarbonate urethane polymer (PCU) core (artificial nucleus). The annulus is mimicked by a woven fibre construct from ultra-high-molecular-weight-polyethylene (UHMWPE) fibres and is responsible for facilitating the semi-constrained six-degrees of freedom [29]. An additional design feature is the polymer sheath that surrounds the core and woven fibre construct to limit tissue ingrowth and contain possible wear debris. The M6-C artificial nucleus and annulus were designed to provide the same physiological motion characteristics of a natural disc, including compressive deformation to axial load [30]. The M6-C has two titanium outer plates coated with a titanium plasma spray to promote bony ingrowth. The initial stability was provided by two keels on each endplate. The device was implanted outside the USA before the completion of the FDA Investigational Device Exemption (IDE) study [31].
Methods
This study was a retrospective cohort analysis from a prospective study of 382 patients who received a standalone CTDR or as part of a hybrid procedure (ACDF/CTDR). This study included 95 PCM® (Nuvasive, San Diego, California), 34 Discover® (Depuy Synthes, Raynham, Massachusetts), 189 Prodisc® C Vivo (Centinel Spine, West Chester, Pennsylvania), 9 CP-ESP® (FH Orthopaedics, Heimsbrunn, Alsace), 2 Mobi-C® (Zimmer Biomet, Austin, Texas), and 53 M6-C. This analysis was prompted by the clinical and radiologic failure of patients receiving the M6-C CTDR, after an Australian Therapeutic Goods Administration (TGA) Implant Hazard Alert was received. The TGA Implant Hazard Alert is available as an Online Resource (Online Resource 1). A search of the Database of adverse event notifications (DAEN) revealed 20 notifications to the TGA up to 4 August 2021, with the majority of these notifications referencing osteolysis and/ or implant failure.
All patients were contacted, and further radiologic and clinical reviews were performed. Plain films, bone scans, and magnetic resonance imaging (MRI) are unreliable in detecting osteolysis. A computed topography (CT) scan is the investigation of choice with 3-dimensional reformats (Fig. 1). The retrieval analysis revealed macroscopic disc failure and histologic evidence of wear induced osteolysis around the revised M6-C prosthesis raising significant concerns regarding durability of this device. Bacteriological contamination was not detected [32].
Mean differences in pain from baseline and 95% confidence interval (CI) at 3-, 6- and 12-months post-surgery for prosthesis groups M6-C™ (Orthofix, Lewisville, Texas) and other cervical total disc replacements (CTDR). a Visual Analogue Score neck (VAS-N) improvement was statistically and clinically significant within both groups. b Visual Analogue Score arm (VAS-A) improvement was statistically significant within both groups, and clinically significant for ‘Other CTDR’ group. Differences between groups were neither statistically nor clinically significant for the neck and arm pain outcomes
PROMs, including Visual Analogue Score neck (VAS-N), and arm (VAS-A), and the Neck Disability Index (NDI), were collected preoperatively, then postoperatively at 3, 6, and 12 months, and annually thereafter. Descriptive statistics were reported as mean (SD) for normally distributed continuous variables or median (IQR) for skewed variables. Categorical variables were summarised as counts and percentages. Differences in VAS-N, VAS-A, and NDI scores from baseline to follow-up timepoint within groups were compared using the paired t-test. Differences in continuous variables between the groups M6-C and other CTDR were compared using the independent t-test or the Mann–Whitney U test, depending on data distribution. Bonferroni correction was applied to multiple comparisons for each outcome so that statistical significance was assessed at the 0.006 level. Graphical representations of the mean changes from baseline and 95% confidence interval (CI) were plotted for each prosthesis group, along with the minimum clinically important difference (MCID) of 25 points for VAS-N and VAS-A, and 15 points for the NDI. All statistical analyses were carried out using R version 4.0.2.
Indications for the index surgery included cervical radiculopathy, myelopathy, and radiculomyelopathy secondary soft tissue herniations. Contraindications to CTDR included osteoporosis, advanced spondylosis, retro vertebral compression, kyphotic deformity, and facet arthropathy. A right sided Smith-Robinson approach was utilized with the neck secured in place in a neutral position, this being verified by image intensifier. Distraction pins were placed into the midline of vertebral body above and below the disc to be replaced. A complete discectomy and removal of the complete cartilaginous endplate to the posterior annulus and uncovertebral joints was performed. Following this uncovertebral decompression was performed followed by resection of the posterior annulus and longitudinal ligament. Removal of the sequestered fragment and osteophytes completed cord and neuroforaminal decompression. Sizing trials were then performed to achieve the best fit in the height, width, and depth. The same surgical technique was performed on all CTDRs.
Prospectively collected PROMs of the M6-C and the other CTDR cohort (Tables 1 and 2) are tabulated and displayed in Figs. 2 and 3. Characteristics of all CTDR surgery patients (Table 3) were tabulated. Revision rates were compared with other prostheses (Table 4), considering that the same indications and surgical techniques were utilized, and the follow-up was longer than 5 years for most of the prostheses. Subsequent surgical interventions were classified as revisions, removals, reoperations, or supplemental fixations (Table 5).
Mean differences in Neck Disability Index (NDI) score from baseline and 95% confidence interval (CI) at 3-, 6- and 12-months post-surgery for the M6-C™ (Orthofix, Lewisville, Texas) and other cervical total disc replacement (CTDR) groups. NDI was statistically and clinically significant within both groups. Differences between groups were neither statistically nor clinically significant
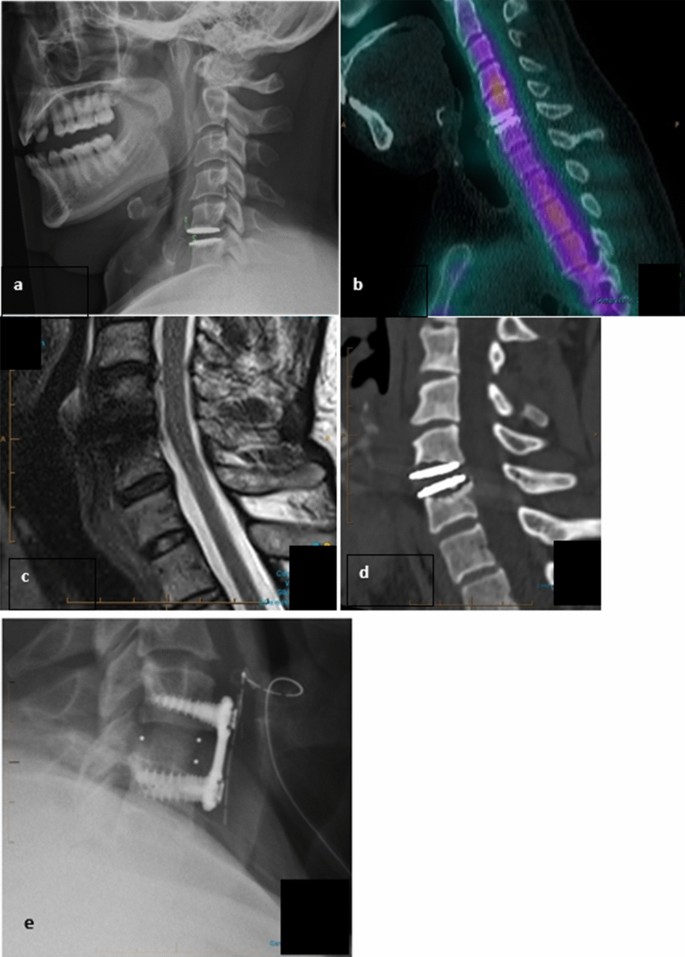
Patient 13 radiological images pre- and post-revision surgery of the M6-C™ (Orthofix, Lewisville, Texas), a. Pre-operative plain X-ray film with anterior migration secondary to osteolysis. b. Pre-operative SPECT scan with no osteoblastic activity. c. Pre-operative MRI scan with an interference signal making interpretation difficult. d. Pre-operative CT scan showing extensive osteolysis and potential mechanical and neurological instability. e. Post-operative plain X-ray film with conversion to anterior cervical fusion
Figures 2 and 3 show that all differences from baseline were clinically significant, except for VAS-A in the M6-C group. None of the differences between prosthesis groups were clinically significant.
Tables 2 and 3 show that the mean change-from-baseline scores were all statistically significant. However, there were no statistically significant differences in improvement between the M6-C and other CTDR groups. The largest difference was at 3 months with VAS-A improvement being lower in the M6-C group by a mean of 11.0 points (p = 0.049; not significant at the 0.006 level).
Results
Fifty-three patients received M6-C CTDR. Eighteen patients (34%) were revised at an average of 67 months postoperatively. The PROMs of the two groups were similar, indicating that the failure mode (wear-induced osteolysis) is often asymptomatic. The demographics of the two groups were also similar, with more women subjected to revision surgery than men. All surgeries (except for one case) were revised using a right anterior approach. Dissection superior to the omohyoid gained access to the cervical spine. The level was verified by an image intensifier, and the longus colli were mobilised to gain exposure to the device. Caspar distraction pins were inserted, and the anterior osteophytes and scar tissue were removed to visualise the entire implant. The disrupted polyethylene weaves and PCU core was removed. A small osteotome dislodged the titanium endplates. The osteolytic defects were then curetted, and a decision to replace, fuse, or perform vertebrectomy was made depending on the integrity of the bony endplates and facets, and bone stock. There were no intra-or post-operative complications.
The 18 cases of index-level revision of the M6-C prosthesis were all performed for polyethylene wear-induced osteolysis. There were three one-level CTDR, four two-level hybrids, seven three-level hybrids, and three four-level hybrids revised anteriorly. All patients underwent removal of the prosthesis and were treated according to the extent of osteolysis. There were four vertebrectomies, six revisions to ACDF, and seven revisions to another CTDR. One patient underwent supplemental fixation using a posterior approach. There was only one other mechanical failure in the other CTDR revision cohort. This was a CP-ESP that had mechanical failure of the internal coupling at 3 months postoperatively and presented as an asymptomatic subluxation. This has been revised to include another CTDR. The other 10 revisions occurred for combinations of subsidence, uncovertebral, and facet arthropathy causing recurrent neural compression and radiculopathy. These were all converted to the ACDF.
Discussion
There is limited data on the long-term effects of spinal loading on CTDR prostheses and the immune response that is initiated secondary to wear particles [33]. This presents significant challenges for developers of CTDR prostheses [34]. Understanding the properties of implantable biomaterials is important, considering the movement within the implanted biomaterials and between the implanted biomaterial and the natural tissues under normal in vivo physiological loads. Additionally, different surgeons, techniques, and pathologies [35] may result in an array of unique forces that create wear particles. These may be of different sizes and shapes and, consequently, a specific biological response may develop unique to that recipient, which can influence the immune response to a prosthesis debris and thereby the clinical course [36]. The mechanisms of osteolysis-induced aseptic loosening following joint arthroplasty and subsequent revision strategies are well understood [37, 38]. Revision surgery has been shown to be more technically demanding, associated with a higher complication rate, poorer outcomes, and considerable costs, both in direct and indirect terms.
Aseptic loosening, secondary to periprosthetic osteolysis, can have catastrophic secondary effects [39, 40]. Mechanical and neurological injuries have already been reported as case studies with the M6-C CTDR [41,42,43,44]. Blumenthal (2021) presented five cases of late osteolytic reaction after CTDR who required reoperation. Four of those cases involved the M6-C disc, while one involved the Prestige™ LP Cervical Disc System (Medtronic, Memphis, Tennessee) [45]. All M6-C disc retrievals showed evidence of wear and there was Propionibacterium acnes detected. A subsequent study by Mobbs suggest this may have been a contaminant [32].
There have been more than 55,000 M6-C prostheses inserted around the world as of 2019 [46]. The 2020 Demographics of Spinal Disc Arthroplasty Report is based on the analysis of 7,325 spinal disc procedures recorded by the Australian Orthopaedic Association National Joint Replacement Registry (AOANJR), with a procedure date up to and including 31 December 2019. In July 2020, the TGA issued an Implant Hazzard Alert about the M6-Cstating, “Routine long term clinical and radiographic monitoring of patients implanted with the M6 is suggested…. Changes in disc position, loss of height and periprosthetic bone loss may be indicative of onset of osteolysis”. The TGA alert initiated this study. Since 2016, 1,460 M6-C have been implanted in Australia. [6] At the current revision rate, approximately 496 patients may require revision for mechanical failure over the next 5–10 years [47].
The long-term success of a CTDR requires an optimised compromise among implant material, design, and biological performance [39]. Implants were initially designed as ball-and-socket CTDR devices and were mobile, fixed, or constrained. The mid- and long-term clinical success of most of these devices has been published [16,17,18]. A more recent design is the one-piece non-articulating disc implant (Monoblock), which replicates the physiologic movement of a normal disc and reduces the number of wears contributing articulations [48]. They were developed to address concerns about the ball-and-socket designs, which provided no axial cushioning and extra-physiological movement in rotation and lateral bending, leading to excessive stresses being placed on the facet joints [27]. The Monoblock’s nucleus/annulus couple in the new designs must be able to undergo repetitive elastic deformation without failure under physiological loads, raising new concerns regarding the choice of materials for the constitution of the viscoelastic cushion. An early example was the AcroFlex lumbar TDR (DePuy Acromed, Raynham, Massachusetts). This design used a polyolefin rubber to mimic the mechanical behaviour of a natural disc. Although the device was tested for biological and biomechanical compatibility prior to clinical trials, patients suffered core material tears and failure mechanisms associated with fatigue. Recently, PCU as a core material has been utilized which has a longer fatigue life [49,50,51]. The M6-C prosthesis is constructed of titanium plates with a PCU core with UHMWPE fibre encapsulation [30, 52]. This device has been classified as a (sandwiched endplate) mobile bumper design. The core is not bonded to the endplate; therefore, there are no peak stresses at the interface. The stresses are distributed between the PCU core and UHMWPE fibre [49, 50, 53].
The polymer sheath that envelops the PCU core and polyethylene- fibre construct was designed to limit soft tissue ingrowth and contain wear debris. This has clearly proven to be ineffective. It has been thought that the device, having a variable centre of rotation, was less susceptible to malpositioning [29, 64]. On the contrary, any malpositioning of the device in the coronal plane would result in lateral tilt if positioned eccentric from the midline and, in the sagittal plane, would result in kyphosis or excessive lordosis the implant. These both create altered mechanics and most probably a precursor to accelerate wear and subsequent osteolysis.
Presently, there are two different testing protocols for studying implant wear, the ISO/FDIS 18192 [54] and ASTM 2423 [55]. Wear debris analysis were performed per ASTM1877 [56], and kinematic analysis per ATSM 2423 [55]. Spinal kinetics Inc. tested 6 prostheses through 20 million cycles combined motion modes (2 Hertz). All samples were functional and met the acceptance criteria. The average height loss was 1.6 ± − 0,3 mm. The average mass loss was 16.9 ± − 12,0.0 mg plus an additional 25.9 mg trapped inside the device [57].
The ASTM applied kinematic pattern of movement testing is unidirectional with a curvilinear shape, whereas the ISO is multidirectional. Nechtow et al. reported that cross-shear loading significantly increased the wear rate compared to curvilinear motion [58]. Grupp et al., when analysing wear rates in the activeL® (B Braun, Tuttlingen, Germany), found that the alternative wear simulations result in a difference in the gravimetric wear amount of approximately 20-fold between the ISO and ASTM methods. The main explanation for the divergence between ISO- and ASTM-driven wear simulations is the multidirectional pattern of movement described in the ISO document, resulting in cross-shear stress on the polyethylene material. It would be reasonable to assume that the testing according to ASTM F2423-05 with pure unidirectional motion does not reflect the kinematics of CTDR patients “daily activities”[59]. Unidirectional testing can lead to strain hardening of the polyethylene [60]. In vivo, large asymmetric loads and multidirectional forces are continuously applied to the polyethylene weave and the cross-shear loading significantly increases the wear rate by an order of magnitude [61]. The generation of wear particles then activate a biological cascade in the periprosthetic tissues with phagocytosis of the particles releasing inflammatory proteins resulting in osteolysis and probable mechanical and neurological instability [62].
The standard testing methods for wear defined by ASTM and ISO are only appropriate for determining wear because of friction at the interface between hard layers, as in metal-on-metal or metal-on-polymer implants [30]. There must be a discrepancy when using these standardised testing methods and application to viscoelastic TDR prostheses. These methods may be unsuitable for soft, deformable, viscoelastic implants because motion is not provided based on friction but rather on deformation. Assessments of the deformation characteristics (physical relaxation, creep, and hysteresis) and interfacial micro-motion with Rheometric Solids Analyser like techniques may be more appropriate [63].
These viscoelastic devices were developed to better mimic the kinematics of a natural intervertebral disc by incorporating a “soft” viscoelastic component [29, 64]. As a result, they appear to be more prone to having a lower physical endurance compared with ball-and-socket designs.
Retrieval of the data was from an ongoing prospective case series of PROMs in relation to patients receiving CTDR. This prospective study allows comparison between different CTDR devices regarding PROMs, failure scenarios, revision surgeries, and their outcomes. Therefore, this is a retrospective cohort study that compared CTDR revision aetiologies and how the revisions were managed. The cohort of patients with a similar diagnosis and treatment, selected based on exposure to a CTDR at the present time, and outcome data, which were measured in the past, were reconstructed for analysis. The primary disadvantage of this type of study design is usually the limited control the investigator has over data collection and follow-up. Only one of the 53 patients was lost to follow-up.
Conclusions
There is a concerning failure rate midterm that is related to UHMWPE wear-induced osteolysis with the M6-C. Patients implanted with the M6-C prosthesis were contacted, informed, and clinically and radiologically assessed. New standardised testing protocols which are more applicable for the new generation of viscoelastic CTDRs are required to ensure clinical safety based on these tests. These testing protocols, which assess the efficacy of medical devices, need to keep pace with the innovation in the medical device industry so that a particular revised characteristic of a device can be properly evaluated.
References
Gutman G, Rosenzweig D, Golan J (2017) The surgical treatment of cervical radiculopathy: meta-analysis of randomized controlled trials. Spine (Phila Pa 1976) 43:365–372. https://doi.org/10.1097/brs.0000000000002324
Coric D, Nunley PD, Guyer RD et al (2011) Prospective, randomized, multicenter study of cervical arthroplasty: 269 patients from the Kineflex|C artificial disc investigational device exemption study with a minimum 2-year follow-up. J Neurosurg Spine 15:348–358. https://doi.org/10.3171/2011.5.SPINE10769
Delamarter RB, Murrey D, Janssen ME et al (2010) Results at 24 months from the prospective, randomized, multicenter Investigational device exemption trial of ProDisc-C versus anterior cervical discectomy and fusion with 4-year follow-up and continued access patients. SAS J 4:122–128. https://doi.org/10.1016/j.esas.2010.09.001
Gornet MF, Burkus JK, Shaffrey ME et al (2015) Cervical disc arthroplasty with PRESTIGE LP disc versus anterior cervical discectomy and fusion: a prospective, multicenter investigational device exemption study. J Neurosurg Spine 23:558–573. https://doi.org/10.3171/2015.1.SPINE14589
Heller JG, Sasso RC, Papadopoulos SM et al (2009) Comparison of BRYAN cervical disc arthroplasty with anterior cervical decompression and fusion: clinical and radiographic results of a randomized, controlled, clinical trial. Spine (Phila Pa 1976) 34:101–107. https://doi.org/10.1097/BRS.0b013e31818ee263
Hisey MS, Bae HW, Davis R et al (2014) Multi-center, prospective, randomized, controlled investigational device exemption clinical trial comparing mobi-C cervical artificial disc to anterior discectomy and fusion in the treatment of symptomatic degenerative disc disease in the cervical spine. Int J Spine Surg. https://doi.org/10.14444/1007
Murrey D, Janssen M, Delamarter R et al (2009) Results of the prospective, randomized, controlled multicenter food and drug administration investigational device exemption study of the ProDisc-C total disc replacement versus anterior discectomy and fusion for the treatment of 1-level symptomatic cervi. Spine J 9:275–286. https://doi.org/10.1016/j.spinee.2008.05.006
Phillips FM, Lee JYB, Geisler FH et al (2013) A prospective, randomized, controlled clinical investigation comparing PCM cervical disc arthroplasty with anterior cervical discectomy and fusion: 2-year results from the US FDA IDE clinical trial. Spine (Phila Pa 1976) 38:907–918. https://doi.org/10.1097/BRS.0b013e318296232f
Vaccaro A, Beutler W, Peppelman W et al (2013) Clinical outcomes with selectively constrained SECURE-C cervical disc arthroplasty: two-year results from a prospective, randomized, controlled, multicenter investigational device exemption study. Spine (Phila Pa 1976) 38:2227–2239
Goffin J, Van Calenbergh F, Van Loon J et al (2003) Intermediate follow-up after treatment of degenerative disc disease with the bryan cervical disc prosthesis: single-level and bi-level. Spine (Phila Pa 1976) 28:2673–2678. https://doi.org/10.1097/01.BRS.0000099392.90849.AA
Davis RJ, Kim KD, Hisey MS et al (2013) Cervical total disc replacement with the mobi-C cervical artificial disc compared with anterior discectomy and fusion for treatment of 2-level symptomatic degenerative disc disease: a prospective, randomized, controlled multicenter clinical trial: clinica. J Neurosurg Spine 19:532–545. https://doi.org/10.3171/2013.6.SPINE12527
FDA Device Advice - Comprehensive regulatory assistance. https://www.fda.gov/medical-devices/device-advice-comprehensive-regulatory-assistance. Accessed 18 May 2021
Lundh A, Lexchin J, Mintzes B et al (2017) Industry sponsorship and research outcome. Cochrane Database Syst Rev Feb. https://doi.org/10.1002/14651858.MR000033.pub3
McAfee PC, Reah C, Gilder K et al (2012) A meta-analysis of comparative outcomes following cervical arthroplasty or anterior cervical fusion: results from 4 prospective multicenter randomized clinical trials and up to 1226 patients. Spine (Phila Pa 1976) 37:943–952. https://doi.org/10.1097/BRS.0b013e31823da169
Anderson PA, Nassr A, Currier BL et al (2017) Evaluation of adverse events in total disc replacement: a meta-analysis of FDA summary of safety and effectiveness data. Glob Spine J 7:76S-83S
Radcliff K, Coric D, Albert T (2016) Five-year clinical results of cervical total disc replacement compared with anterior discectomy and fusion for treatment of 2-level symptomatic degenerative disc disease: a prospective, randomized, controlled, multicenter investigational device exemption. J Neurosurg Spine 25:213–214. https://doi.org/10.3171/2015.12.SPINE15824
Lanman TH, Burkus JK, Dryer RG et al (2017) Long-term clinical and radiographic outcomes of the Prestige LP artificial cervical disc replacement at 2 levels: results from a prospective randomized controlled clinical trial. J Neurosurg Spine 27:7–19. https://doi.org/10.3171/2016.11.SPINE16746
Lavelle WF, Riew KD, Levi AD, Florman JE (2019) Ten-year outcomes of cervical disc replacement with the BRYAN cervical disc. Spine (Phila Pa 1976) 44:601–608. https://doi.org/10.1097/brs.0000000000002907
Holsgrove TP, Amin DB, Pascual SR et al (2018) The equivalence of multi-axis spine systems: recommended stiffness limits using a standardized testing protocol. J Biomech 21:59–66. https://doi.org/10.1016/j.jbiomech.2017.09.010
ASTM International (2005) F2346–05 (Reapproved 2011) - Standard test methods for static and dynamic characterization of spinal artificial discs. ASTM International, West Conshohocken, PA, USA
ASTM International (2011) F2423–11 - Standard guide for functional, kinematic, and wear assessment of total disc prostheses. ASTM International, West Conshohocken, PA, USA
BS ISO 18192-1 (2011) Implants for surgery. Wear of total intervertebral spinal disc prostheses. Loading and displacement parameters for wear testing and corresponding environmental conditions for test. British Standards Institution, London
Anderson PA, Rouleau JP, Toth JM, Riew KD (2004) A comparison of simulator-tested and -retrieved cervical disc prostheses. J Neurosurg Spine 1:202–210. https://doi.org/10.3171/spi.2004.1.2.0202
Lehman R, Bevevino A, Brewer D et al (2012) A systematic review of cervical artificial disc replacement wear characteristics and durability. Evid Based Spine Care J 3:31–38. https://doi.org/10.1055/s-0031-1298606
Pettine K (2011) Clinical significance of hypermobility in cervical artificial disc replacements. Spine J 11:S106. https://doi.org/10.1016/j.spinee.2011.08.264
Huang RC, Tropiano P, Marnay T et al (2006) Range of motion and adjacent level degeneration after lumbar total disc replacement. Spine J 6:242–247. https://doi.org/10.1016/j.spinee.2005.04.013
Reeks J, Liang H (2015) Materials and their failure mechanisms in total disc replacement. Lubricants. https://doi.org/10.3390/lubricants3020346
Goel VK, Panjabi MM, Patwardhan AG et al (2006) Test protocols for evaluation of spinal implants. J Bone Jt Surg - Ser A 88:103–109. https://doi.org/10.2106/JBJS.E.01363
Patwardhan AG, Tzermiadianos MN, Tsitsopoulos PP et al (2012) Primary and coupled motions after cervical total disc replacement using a compressible six-degree-of-freedom prosthesis. Eur Spine J 21:S618–S629. https://doi.org/10.1007/s00586-010-1575-7
Jacobs CAM, Siepe CJ, Ito K (2020) Viscoelastic cervical total disc replacement devices: design concepts. Spine J 20:1911–1924. https://doi.org/10.1016/j.spinee.2020.08.007
Phillips FM, Coric D, Sasso R et al (2021) Prospective, multicenter clinical trial comparing M6-C compressible six degrees of freedom cervical disc with anterior cervical discectomy and fusion for the treatment of single-level degenerative cervical radiculopathy: 2-year results of an FDA investiga. Spine J 21:239–252. https://doi.org/10.1016/j.spinee.2020.10.014
Rao PJ, Maharaj M, Chau C et al (2020) Degenerate-disc infection study with contaminant control (DISC): a multicenter prospective case-control trial. Spine J 20:1544–1553. https://doi.org/10.1016/j.spinee.2020.03.013
Ruggiero A, Zhang H (2020) Editorial biotribology and biotribocorrosion properties of implantable biomaterials. Front Mech Eng April. https://doi.org/10.3389/fmech.2020.00017
Park CK, Ryu KS (2018) Are controversial issues in cervical total disc replacement resolved or unresolved?: A review of literature and recent updates. Asian Spine J 12:178–192. https://doi.org/10.4184/asj.2018.12.1.178
Nunley PD, Coric D, Frank KA, Stone MB (2018) Cervical disc arthroplasty: current evidence and real-world application. Clin Neurosurg 83:1087–1106. https://doi.org/10.1093/neuros/nyx579
Kaddick C, Catelas I, Pennekamp PH, Wimmer MA (2009) Implant wear and aseptic loosening: an overview. Implant Wear Aseptic Loos Overv 38:690–697. https://doi.org/10.1007/s00132-009-1431-9
Merola M, Affatato S (2019) Materials for hip prostheses: a review of wear and loading considerations. Materials (Basel) 12:495. https://doi.org/10.3390/ma12030495
Baxter RM, MacDonald DW, Kurtz SM, Steinbeck MJ (2013) Severe impingement of lumbar disc replacements increases the functional biological activity of polyethylene wear debris. J Bone Jt Surg - Ser A 95:e751-759. https://doi.org/10.2106/JBJS.K.00522
Veruva SY, Steinbeck MJ, Toth J et al (2014) Which design and biomaterial factors affect clinical wear performance of total disc replacements? A systematic review. Clin Orthop Relat Res 472:3759–3769. https://doi.org/10.1007/s11999-014-3751-2
Tumialán LM, Gluf WM (2011) Progressive vertebral body osteolysis after cervical disc arthroplasty. Spine (Phila Pa 1976) 36:E973-978. https://doi.org/10.1097/BRS.0b013e3181fd863b
Xia M-AM, Winder MJ (2019) M6-C cervical disc replacement failure associated with late onset infection. J Spine Surg 5:584–588. https://doi.org/10.21037/jss.2019.11.06
Clark NJ, Francois EL, Freedman BA, Currier B (2020) Early implant failure of a 2-level M6-cervical total disc replacement: a case report. JBJS Case Connect 10:e1900644. https://doi.org/10.2106/JBJS.CC.19.00644
Harris L, Dyson E, Elliot M et al (2020) Delayed periprosthetic collection after cervical disc arthroplasty. J Neurosurg Spine 32:584–591. https://doi.org/10.3171/2019.9.SPINE19900
Xu JC, Goel C, Shriver MF et al (2018) Adverse events following cervical disc arthroplasty: a systematic review. Glob Spine J 8:178–189
Blumenthal SL, Kiridly D, Guyer RD et al (2021) Late failure of cervical disc arthroplasty due to osteolysis. In: [paper presentation] ISASS 2021; 21st annual conference. Miami, Florida
OrthoFix (2021) M6-C Cervical disc website. https://m6disc.com/. Accessed 9 Jul 2021
Virk S, Phillips F, Khan S, Qureshi S (2021) A cross-sectional analysis of 1347 complications for cervical disc replacements from medical device reports maintained by the United States Food and drug administration. Spine J 21:265–272. https://doi.org/10.1016/j.spinee.2020.09.005
Reyes-Sanchez A, Miramontes V, Olivarez LMR et al (2010) Initial clinical experience with a next-generation artificial disc for the treatment of symptomatic degenerative cervical radiculopathy. SAS J 4:9–15. https://doi.org/10.1016/j.esas.2010.01.002
Serhan H, Ross R, Lowery G, Fraser R (2002) Biomechanical characterization of a new lumbar disc prosthesis. J Bone Jt Surg [Br] 84:215
Moore RJ, Fraser RD, Vernon-Roberts B et al (2002) The biologic response to particles from a lumbar disc prosthesis. Spine (Phila Pa 1976) 27:2088–2094. https://doi.org/10.1097/00007632-200210010-00003
Fraser RD, Ross ER, Lowery GL, Freeman BJ, Dolan M (2004) AcroFlex design and results. Spine J. https://doi.org/10.1016/j.spinee.2004.07.020
Spinal Kinetics Inc (2009) Mechanical characterization of M6-C artificial cervical disc
Fraser RD, Ross ER, Lowery GL et al (2004) AcroFlex design and results. Spine J 4:S245–S251. https://doi.org/10.1016/j.spinee.2004.07.020
IMPLANTS for surgery — Wear of total intervertebral spinal disc prostheses — Part 1: Loading and displacement parameters for wear testing and corresponding environmental conditions for test (2011) International standard ISO 18192–1:2011
Standard guide for functional, kinematic, and wear assessment of total disc prostheses (2020) Annual Book of ASTM Standards ASTM F2423 - 11
Standard practice for characterization of particles (2016) Annual Book of ASTM Standards ASTM 1877–1
Sientra, Inc. Summary of Safety and Effectiveness Data (SSED) (2012) Santa Barbara, California, USA, Sientra
Nechtow W, Hintner M, Bushelow M, Kaddick C (2006) Intervertebral disc replacement mechanical performance depends strongly on input parameters. 52nd Orthopaedic Research Society, Chicago, Abstract 0118
Grupp TM, Yue JJ, Garcia R et al (2009) Biotribological evaluation of artificial disc arthroplasty devices: influence of loading and kinematic patterns during in vitro wear simulation. Eur Spine J 18:98–108. https://doi.org/10.1007/s00586-008-0840-5
Kang L, Galvin AL, Brown TD et al (2008) Quantification of the effect of cross-shear on the wear of conventional and highly cross-linked UHMWPE. J Biomech 41:340–346. https://doi.org/10.1016/j.jbiomech.2007.09.005
Wang A (2001) A unified theory of wear for ultra-high molecular weight polyethylene in multi-directional sliding. Wear 248:38–47. https://doi.org/10.1016/S0043-1648(00)00522-6
Korduba LA, Wang A (2011) The effect of cross-shear on the wear of virgin and highly-crosslinked polyethylene. Wear 271:1220–1223. https://doi.org/10.1016/j.wear.2011.01.039
DeLucca JF, Amin D, Peloquin JM et al (2019) Off-axis response due to mechanical coupling across all six degrees of freedom in the human disc. JOR SPINE. https://doi.org/10.1002/jsp2.1047
Patwardhan AG, Havey RM (2020) Prosthesis design influences segmental contribution to total cervical motion after cervical disc arthroplasty. Eur Spine J. https://doi.org/10.1007/s00586-019-06064-4
Acknowledgements
We thank Associate Professor John Costi for his contribution to this paper.
Funding
The authors did not receive support from any organization for the submitted work. M Scott-Young was a principal investigator in the Discover™ Artificial Disc IDE study between 2008 and 2015.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they do not have any conflict of interest to declare.
Ethical approval
Bond University’s Human Research Ethics Committee (BUHREC) approval number: 0000015881.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Scott-Young, M., Rathbone, E. & Grierson, L. Midterm osteolysis-induced aseptic failure of the M6-C™ cervical total disc replacement secondary to polyethylene wear debris. Eur Spine J 31, 1273–1282 (2022). https://doi.org/10.1007/s00586-021-07094-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-021-07094-7