Abstract
Purpose
Positive spinal sagittal alignment is known to correlate with pain and disability. The association between lumbar spinal stenosis and spinal sagittal alignment is less known, as is the effect of lumbar decompressive surgery on the change in that alignment. The objective was to study the evidence on the effect of lumbar decompressive surgery on sagittal spinopelvic alignment.
Methods
The Cochrane Controlled Trials Register (CENTRAL), Medline, Embase, Scopus and Web of Science databases were searched in October 2019, unrestricted by date of publication. The study selection was performed by two independent reviewers. The risk of systematic bias was assessed according to the NIH Quality Assessment Tool. The data were extracted using a pre-defined standardized form.
Results
The search resulted in 807 records. Of these, 18 were considered relevant for the qualitative analysis and 15 for the meta-synthesis. The sample size varied from 21 to 89 and the average age was around 70 years. Decompression was mostly performed on one or two levels and the surgical techniques varied widely. The pooled effect sizes were most statistically significant but small. For lumbar lordosis, the effect size was 3.0 (95% CI 2.2 to 3.7) degrees. Respectively, for pelvic tilt and sagittal vertical axis, the effect sizes were − 1.6 (95% CI .2.6 to − 0.5) degrees and − 9.6 (95% CI − 16.0 to − 3.3) mm.
Conclusions
It appears that decompression may have a small, statistically significant but probably clinically insignificant effect on lumbar lordosis, sagittal vertical axis and pelvic tilt.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Lumbar spinal stenosis (LSS) is the most common cause of disability due to a spinal disorder [1]. It is also the most common reason for spinal surgery in the elderly [2]. For example, in the USA, the rate of lumbar decompression is around 136 per 100,000 Medicare beneficiaries. Simultaneously, the amount of fusion surgery for treating LSS has also increased [3]. Compared to conservative treatment, decompressive surgery with or without fusion has shown a positive effect on patients’ symptoms, especially leg pain, claudication and overall disability [4, 5].
Consideration of sagittal spinal alignment arose with the evolution of operative treatment in adolescent idiopathic scoliosis (AIS) in the late 1980s [6]. Since Legaye and Duval-Beaupère introduced pelvic incidence (PI) as a key parameter regulating sagittal spinal balance [7], sagittal balance and its correlation with the results of spine surgery have been widely studied. PI is considered a constant parameter with no significant change with age, while thoracic kyphosis (TK) increases and lumbar lordosis (LL) decreases with age [8, 9].
Sagittal spino-pelvic alignment (SSPA) describes spinal and pelvic orientation in the erect posture with radiographic parameters. A correlation has been found between the shape and orientation of the pelvis and the morphology of sagittal spinal curvatures in asymptomatic persons [10, 11]. Greater positive SSPA has been found in asymptomatic elderly people [8, 9, 12]. Decreased LL has been shown to have a strong correlation with low back pain [13]. When increased positive SSPA appears as part of degenerative scoliosis, the degenerative changes in spinal structures can be considered irrecoverable.
The Scoliosis Research Society-Schwab adult deformity classification describes spinal deformity two-dimensionally with coronal curve types and three sagittal modifiers [14]. The first of the sagittal modifiers is PI-LL mismatch (PI-LL), which is the difference between the current LL and the ideal based on the pelvic anatomy and PI. The second modifier is global alignment with the sagittal vertical axis (SVA), which is influenced by changes in LL and TK as well as compensatory mechanisms such as knee flexion and pelvic orientation, described with the third modifier, pelvic tilt (PT).
The correlation between SSPA and patient-reported outcome measures (PROM) has been reported with poorer PROM scores associated with increased SVA and PT in adults with spinal deformities [15,16,17]. Realignment surgery has been shown to have a superior effect on both back pain and quality of life with adult spinal deformity, compared to conservative treatment [18, 19], and a greater correction of SSPA is related to a higher health-related quality of life (HRQOL) [20].
A well-known phenomenon is relief from spinal claudication by bending forward. The movement reduces LL, providing additional space to the compressed nerve roots [21, 22]. There have only been a few studies on SSPA in LSS patients compared to the asymptomatic population, two of which suggest that LSS could affect SSPA [23, 24]. Comparing compensatory mechanisms between patients with LSS and those with adult spinal deformity (ASD), the former are more prone to recruit pelvic shift than PT, while the opposite is true of the those with ASD [25]. However, overall evidence on an association between LSS and SSPA is scarce.
While decompression surgery is still the most common operative treatment for LSS, its effect on SSPA is not well known. The objective of this systematic review was to examine the evidence on the effects of decompressive surgery on the parameters of SSPA among patients with LSS.
Methods
Inclusion and exclusion criteria (PICO)
Population
Adults undergoing lumbar laminectomy for degenerative conditions. Excluding traumas, malignancy, tuberculosis or other spinal infection, connective tissue disorders (rheumatoid arthritis, ankylosing spondylitis, sacroiliitis or respective), pregnancy, congenital or developmental abnormalities, cervical or thoracic spinal disorders and neuromuscular diseases.
Intervention
Laminectomy was understood as a surgical procedure, whereby a section of bone is removed from one or more vertebrae from L1 to L5 to relieve pressure on the affected nerve or spinal cord.
Comparison
Estimates of SSPA before and after surgery.
Outcome
Change in SSPA measured by any of the radiological parameters shown in Table 1.
Types of studies
Studies of any design published in peer-reviewed academic journals with abstract available. Conference proceedings, theses, case reports and case series were excluded.
Data sources
Medline via PubMed, Embase, Scopus, Web of Science, and the Cochrane Controlled Register of Trials (Central). The search clause for the Medline search was:
("Decompression, Surgical"[Mesh] OR laminectom*[TIAB] OR laminotom*[TIAB]) AND (*alignment*[TIAB] OR balance[TIAB] OR “sagittal plane”[TIAB] OR “sagittal vertical axis”[TIAB] OR “spinopelvic inclination”[TIAB] OR “pelvic angle”[TIAB] OR “pelvic incidence”[TIAB] OR “sacral slope”[TIAB] OR “pelvic tilt”[TIAB] OR “curvature”[TIAB] OR “cervical offset”[TIAB] OR “cervical angle”[TIAB] OR kyphosis[TIAB] OR lordosis[TIAB]) AND lumb*[TIAB] NOT Review[ptyp] AND (hasabstract[text] AND English[lang]).
In order to avoid missing potentially relevant studies, the use of other limiters and filters was restricted, and the authors relied instead on manual selection. Similar clauses were used when searching the other databases. The references of identified articles and reviews were also checked for relevance.
Selection strategy
The records identified from the data sources were stored using Endnote software (Endnote X7.8, Thomson Reuters). Using a built-in search engine of the Endnote software, duplicates, conference proceedings, theses, reviews and case reports were deleted. Two independent reviewers screened the titles and abstracts of the remaining articles and assessed the full texts of potentially relevant papers (Fig. 1). Disagreements between the reviewers were resolved by consensus or by a third reviewer.
Extraction strategy
The data needed for a quantitative assessment were extracted using a standardized form based on recommendations by the Cochrane Handbook for Systematic Reviews of Interventions [26]. The form included: a first author name, a year of publication, a country, a sample, a gender distribution, the average age of patients, the duration of follow-up, surgical techniques and the estimates of main outcomes.
Assessment of the methodological risks of systematic bias
Two independent reviewers rated the methodological quality of the included trials using the NIH Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies (Table 2). This 14-point tool assesses the risks of systematic bias based on the clarity of a research question, a participation rate, a power analysis, a follow-up and dropouts, among other criteria. The risk of bias is dichotomized as “yes” versus “no.” Disagreements between the reviewers were resolved by consensus or by a third reviewer.
Statistical analysis (meta-analysis)
A random-effects model was used to quantify the pooled effect size of the included studies, which was a more fitting choice than a fixed-effect model considering the context of medical decisions making and generalizing the results beyond the selected samples. The results were accompanied by 95% confidence intervals (95% CI). The heterogeneity was tested using the Q test and I2 statistic. Heterogeneity was deemed present if Q was greater than the degree of freedom (number of studies – 1). The I2 statistic described the percentage of the variability in effect estimates due to heterogeneity rather than to sampling error (chance). As the correlation between pre- and post-estimates within groups was not reported, the coefficient of pre/post correlation was set at 0.6, assuming that at least that strong a correlation should exist between two repeated measures. When the number of studies in the model was ≥ 10, a potential publication bias was assessed using Egger’s test (two-tailed p value considered significant if ≤ 0.05), and trim-and-fill correction was applied if needed. All calculations were performed using the Comprehensive Meta-Analysis CMA software, Version 3.0, available from www.meta-analysis.com.
Results
The search resulted in 807 records (Fig. 1). Of these, 211 were duplicates. Using the Endnote® software search engine, 197 records were excluded as conference proceedings, editorials, theses, etc. The remaining 399 records were screened based on titles and abstracts; the agreement between reviewers was good, kappa 0.77 (95% CI 0.66 to 0.87). After further exclusion, 47 records were screened based on their full texts; of these, 18 studies were included for further analysis. Out of 18 studies, 10 had been conducted in Japan and four in South Korea (Table 1). The sample size varied from 21 to 89 and the average age was around 70 years. The duration of follow-up ranged from 0.5 to 6 years. Fourteen studies were retrospective and four prospective. Decompression was performed mostly on one or two levels. Of 18 studies, three failed to produce the data needed for the meta-synthesis [27,28,29]. Thus, the quantitative meta-analysis was performed on 15 studies [30,31,32,33,34,35,36,37,38,39,40,41,42,43,44].
LSS had been used as an inclusion criterion in 15 studies. Others included subjects with decompressive surgery for spinal claudication [27], decompressive surgery for degenerative scoliosis [29] and interlaminar decompression for lumbar intervertebral disk herniation [41]. Six studies provided information on preoperative MRI [30, 35, 37, 42,43,44] and only two had assessed the severity of LSS on axial MRI [37, 44].
Eight different surgical methods for lumbar decompression were employed. One study did not provide detailed information on the surgical method [29]. When laminectomy was involved, the spinous process and bony lamina of the index level were removed, providing a route to decompression of one or two intervertebral levels by removal of the ligamentum flavum. Laminotomy was done either on one or both sides and the bony laminar arch was partially removed, followed by removal of the ligamentum flavum. Some of the studies employed microsurgery and others endoscopic techniques. During spinous split osteotomy and laminoplasty, the spinous process was initially divided or shifted laterally and retracted back to its origin after decompression. Laminectomy was used in three studies. Laminotomy with or without microscopic or endoscopic assistance was used in 10 studies. Spinous split osteotomy or laminoplasty was used in four studies.
The exclusion criteria varied widely. Most of the studies excluded vertebral fracture or post-traumatic kyphosis, neurological disease (e.g., Parkinson’s) and previous spinal surgery. Even though several studies included patients with degenerative spondylolisthesis, none accepted severe spondylolisthesis of grade ≥ 2 according to the Meyerding classification. Degenerative scoliosis of varying definition was mainly excluded. Only five studies focused on changes in SSPA as a primary outcome [27, 34, 37, 39, 40].
Plain radiographs of the lumbar spine were used in eight studies assessing specifically SSPA parameters. The remaining studies employed radiographs of the entire spine or comparable imaging techniques (e.g., EOS™) providing wider information on SSPA and pelvic orientation. Of the SSPA parameters, LL measured between L1 to S1 were reported most frequently (13 studies), with group sizes varying from 11 to 89 resulting in a total of 827 patients (Table 3). The SVA and PT were estimated in 10 groups yielding a pooled sample of 547 patients for each parameter.
Risk of systematic bias
Risk of systematic bias was assessed with the NIH Quality Assessment Tool for Observational Cohort and Cross-Sectional studies [24] (Table 2). The most frequent sources of potential risk of systematic bias were risks related to absent study power analysis, unclear inclusion criteria and non-blinded design. The risk was mostly small regarding the clarity of study objectives, sample descriptions, sufficiently described pre- and post-measures and definitions of variables. Two subcategories, variation of exposures and amount of repeated measures, were considered “not applicable” for all 18 studies. In four studies, the outcome assessors were blinded. Two out of four prospective studies had a dropout of 20% or less. Only two studies reported a participation rate of at least 50%.
Meta-analysis
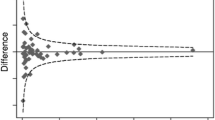
The occipital 7th cervical angle (C0-C7), T1 pelvic angle (TPA) and spinosacral angle (SSA) were used in the included studies only once (Table 3). As shown in Table 4, the pooled effects (when subgroups within the study were used as the unit of analysis) were most statistically significant, except for the PI and thoracolumbar kyphosis (TLK) (T10-L2). When taking into account the 95% confidence limits closest to zero, the difference estimates were small, varying from 1° to 3° (2 mm in the case of SVA). When pooling the results using the study as the unit of analysis, the pooled estimates did not substantially change (Fig. 2). PI, sacral slope (SS) and TLK (T10-L2) were statistically insignificant. Other parameters demonstrated only small fluctuations before and after surgery. Regarding the primary outcomes, LL (L1-S1) increased by 2.95° (95% CI 2.18° to 3.72°), PT decreased by − 1.57° (95% CI − 2.62° to − 0.53°) and SVA decreased by − 9.63 mm (95% CI − 15.99 mm to − 3.27 mm). In the case of LL, a risk of publication bias was not observed with Egger’s test, p value 0.87. The level of heterogeneity varied substantially.
Discussion
This systematic review of 18 observational studies evaluated the evidence on the effect of LSS decompression surgery on SSPA. The meta-analysis of 15 studies showed some small changes in SSPA after surgery. The observed pooled effect was toward more neutral alignment, while SVA and PT decreased, and LL increased after decompressive surgery. While these changes were mostly statistically significant, they showed only small fluctuations of a few degrees or millimeters and were probably not clinically significant. The overall risk of systematic bias of the included studies was considered high using the NIH Quality Assessment Tool for Observational Cohort and Cross-Sectional studies.
The speculated effect of decompression surgery on relieving the compression of cauda equina and the previously observed association between SVA and PT and severity of symptoms, as seen in ASD, could not be confirmed with the present results [15, 16]. A former study by Buckland et al. showed the importance of pelvic shift as compensatory mechanism in LSS [25], but none of studies in the present systematic review employed pelvic shift as a SSPA parameter.
The studies covered eight decompression techniques, laminotomy or its alternatives being the most common. Although these techniques varied substantially [27, 28, 35, 37, 38], they were well-described, allowing them to be compared. No superiority of a particular surgical technique was observed. Cochrane meta-analysis compared the effectiveness of different surgical techniques for LSS. Primary outcomes in the included studies were leg pain, satisfactory, disability indexes, postoperative instability and perioperative complications; no differences between techniques were found [45].
Bernhardt and Bridwell were one of the first to report normal values of sagittal spinal alignment in an asymptomatic population with Cobb measurements of the TK, thoracolumbar junction and lumbar spine [46]. Lenke introduced a new classification for AIS with a sagittal modifier evaluating the extent of TK [47]. The Scoliosis Research Society-Schwab adult deformity classification takes three parameters, PI-LL, SVA and PT, into account in sagittal plane evaluation [14]. Several previous studies have reported the important role of SSPA in spinal deformity [15, 16, 48]. Recently, new surgical techniques have been introduced to restore sagittal imbalance and to treat symptoms in adults with spinal deformity. While the results have been in favor of surgical treatment [18,19,20], the rates of complications and reoperations have been high [48, 49].
In the context of other spinal disorders, the role of SSPA in the reported results has been highly inconsistent. Barrey et al. found one- and two-level lumbar spondylolisthesis to correlate with greater positive SSPA and pelvic retroversion [50]. Rhee et al. did not observe a connection between clinical improvement and changes in LL or overall sagittal imbalance after treated lumbar spondylolisthesis [51]. Similar findings have been reported by Försth et al. when comparing fusion surgery with decompression alone in LSS [52]. Zárate-Kalfópulos et al. proposed pelvic morphology to have a predisposing role in the pathogenesis of lumbar degeneration, with a lower PI being associated with a risk of LSS and a higher PI with a risk of degenerative spondylolisthesis [53].
Evidence on the association between LSS and SSPA is scarce. While bending forward for relief is a well-known phenomenon, two studies have suggested that LSS might affect SSPA. Suzuki et al. found that LSS patients with claudication symptoms have greater positive sagittal balance and increased pelvic retroversion than LSS patients without claudication [24]. Farrokhi et al. reported similar results of greater positive sagittal balance and decreased LL among LSS patients compared with healthy controls [23]. Buckland et al. compared compensatory mechanisms between LSS and ASD patients and found that LSS patients were more prone to increased pelvic shift to allow a forward bending posture, especially in the well-aligned group [25]. Bayerl et al. classified patients undergoing decompression surgery for LSS according to the severity of sagittal imbalance; the results were comparable between groups in leg and back pain and quality of life [54]. While investigating the correlation between spinopelvic parameters and the effect of physiotherapy on the severity of back pain in mild LSS, Beyer et al. also reported that greater PI predicts greater relief in back pain [55]. Additionally, Liang et al. observed a normalization of increased positive sagittal balance after lumbar discectomy [56].
A single previous systematic review of the topic, which included 10 studies (eight of which were included in our review) [57], while lacking a quantitative meta-analysis, estimated that decompression surgery led to SVA correction in 25% to 73% of patients. It has also been suggested that greater PI-LL preoperatively correlates with residual sagittal malalignment postoperatively, which could be explained by structural degenerative changes rather than by reversible changes due to LSS itself [33, 34, 40].
Any generalization of these findings should be done carefully. A meta-analysis is always an approximation. The included studies differed widely regarding the used surgical techniques, inclusion criteria, and the radiological assessment of SSPA parameters. The pooled study sample was limited to a particular age group of around 70 years. The overall risk of systematic bias was high, and there has not been a single randomized controlled trial on the topic. Additionally, only four of the included studies were prospective. The substantial variety of follow-up might also weaken the conclusions of the review, considering that degenerative changes might affect SSPA parameters especially during long-term follow-up, as has previously been observed in the general population [8]. The interpretation of the results might also be uncertain, as there is no generally accepted radiological classification of LSS severity and diagnosis based on a combination of patient history, clinical findings, radiographs and neurophysiological assessment [58]. As only two of the 18 included studies provided some information about LSS grade, the pooled sample might be substantially mixed regarding LSS severity. The influence of coexisting spondylolisthesis on the magnitude of the studied effect is also unclear. Evaluating the clinical meaning of the changes found in our meta-analysis presents some challenges. The minimal clinically important difference is not known for SSPA parameters, and clinical thresholds for symptomatic SSPA are controversial. Although SVA > 47 mm, PT > 22° and PI-LL > 11° have been correlated with more severe disability, there are no generally accepted limits for normal SSPA parameters [17].
In conclusion, the quality of the evidence on the effect of decompressive surgery for LSS on SSPA was low, and there was substantial heterogeneity of the study design among the studies included. Although decompression surgery demonstrated a statistically significant effect on LL, SVA and PT toward more neutral alignment, the effect was small and probably clinically insignificant.
Abbreviations
- SSPA:
-
Consideration of whole spine and pelvis orientation in the sagittal plane (Sagittal spino-pelvic alignment)
- LL:
-
Angle between the lines through measured endplates of lumbar vertebrae (e.g., upper endplate of L1 and upper endplate of S1) (Lumbar lordosis)
- SVA:
-
Horizontal distance from the C7 plumbline from the mid-C7 vertebral body to the posterior superior endplate of S1 (C7 sagittal vertical axis)
- TPA:
-
Angle between the line from the center of T1 to the axis of the femoral heads and the line from the axis of the femoral heads to the middle of the S1 endplate (T1 pelvic angle)
- PT:
-
Angle between the line connecting the midpoint of the sacral plate to the axis of the femoral heads and the vertical axis (Pelvic tilt)
- PI:
-
Angle between the line perpendicular to the sacral plate at its midpoint and the line connecting this point to the axis of the femoral heads (Pelvic incidence)
- SS:
-
Angle between the horizontal line and upper endplate of S1 (Sacral slope)
- PI-LL:
-
Difference between PI minus LL (PI-LL mismatch)
- TK:
-
Angle between the lines through measured endplates of the thoracic vertebrae (e.g., upper endplate of T5 and the lower endplate of T12) (Thoracic kyphosis)
- TLK:
-
Angle between upper endplate of T10 and lower endplate of L2 (Thoracolumbar kyphosis)
- SSA:
-
Angle between the sacral plate between the midpoint of C7 and the center of the sacral plate (Spinosacral angle)
- C0-C7:
-
Angle between the McGregor line and the lower endplate of C7 (Occipital 7th cervical angle)
- GT:
-
Angle subtended by a line from the center of the superior sacral endplate to the center of the C7 vertebral body and a line from the femoral heads to the center of the superior sacral endplate (Global tilt)
- LSS:
-
This article is about degenerative narrowing of the lumbar spinal canal, central or lateral, due to intervertebral disk height loss, hypertrophied ligamentum flavum, facet arthrosis and disk bulging. Foraminal stenosis is not within the scope of this study (Lumbar spinal stenosis)
References
Kalichman L, Cole R, Kim DH, Li L, Suri P, Guermazi A, Hunter DJ (2009) Spinal stenosis prevalence and association with symptoms: the Framingham Study. Spine J 9:545–550. https://doi.org/10.1016/j.spinee.2009.03.005
Deyo RA, Mirza SK, Martin BI, Kreuter W, Goodman DC, Jarvik JG (2010) Trends, major medical complications, and charges associated with surgery for lumbar spinal stenosis in older adults. JAMA 303:1259–1265. https://doi.org/10.1001/jama.2010.338
Bae HW, Rajaee SS, Kanim LE (2013) Nationwide trends in the surgical management of lumbar spinal stenosis. Spine (Phila Pa 1976) 38:916–926. https://doi.org/10.1097/BRS.0b013e3182833e7c
Malmivaara A, Slatis P, Heliovaara M, Sainio P, Kinnunen H, Kankare J, Dalin-Hirvonen N, Seitsalo S, Herno A, Kortekangas P, Niinimaki T, Ronty H, Tallroth K, Turunen V, Knekt P, Harkanen T, Hurri H (2007) Surgical or nonoperative treatment for lumbar spinal stenosis? a randomized controlled trial. Spine (Phila Pa 1976) 32:1–8. https://doi.org/10.1097/01.brs.0000251014.81875.6d
Rihn JA, Hilibrand AS, Zhao W, Lurie JD, Vaccaro AR, Albert TJ, Weinstein J (2015) Effectiveness of surgery for lumbar stenosis and degenerative spondylolisthesis in the octogenarian population: analysis of the Spine Patient Outcomes Research Trial (SPORT) data. J Bone Jt Surg Am 97:177–185. https://doi.org/10.2106/jbjs.N.00313
Thomson JD, Renshaw TS (1989) Analysis of lumbar lordosis in posterior spine fusions for idiopathic scoliosis. J Spinal Disord 2:93–98
Legaye J, Duval-Beaupere G, Hecquet J, Marty C (1998) Pelvic incidence: a fundamental pelvic parameter for three-dimensional regulation of spinal sagittal curves. Eur Spine J 7:99–103. https://doi.org/10.1007/s005860050038
Asai Y, Tsutsui S, Oka H, Yoshimura N, Hashizume H, Yamada H, Akune T, Muraki S, Matsudaira K, Kawaguchi H, Nakamura K, Tanaka S, Yoshida M (2017) Sagittal spino-pelvic alignment in adults: the Wakayama Spine Study. PLoS ONE 12:e0178697. https://doi.org/10.1371/journal.pone.0178697
Pratali RR, Nasreddine MA, Diebo B, Oliveira C, Lafage V (2018) Normal values for sagittal spinal alignment: a study of Brazilian subjects. Clinics (Sao Paulo) 73:e647. https://doi.org/10.6061/clinics/2018/e647
Berthonnaud E, Dimnet J, Roussouly P, Labelle H (2005) Analysis of the sagittal balance of the spine and pelvis using shape and orientation parameters. J Spinal Disord Tech 18:40–47. https://doi.org/10.1097/01.bsd.0000117542.88865.77
Duval-Beaupere G, Schmidt C, Cosson P (1992) A Barycentremetric study of the sagittal shape of spine and pelvis: the conditions required for an economic standing position. Ann Biomed Eng 20:451–462. https://doi.org/10.1007/bf02368136
Yukawa Y, Kato F, Suda K, Yamagata M, Ueta T, Yoshida M (2018) Normative data for parameters of sagittal spinal alignment in healthy subjects: an analysis of gender specific differences and changes with aging in 626 asymptomatic individuals. Eur Spine J 27:426–432. https://doi.org/10.1007/s00586-016-4807-7
Chun SW, Lim CY, Kim K, Hwang J, Chung SG (2017) The relationships between low back pain and lumbar lordosis: a systematic review and meta-analysis. Spine J 17:1180–1191. https://doi.org/10.1016/j.spinee.2017.04.034
Schwab F, Ungar B, Blondel B, Buchowski J, Coe J, Deinlein D, DeWald C, Mehdian H, Shaffrey C, Tribus C, Lafage V (2012) Scoliosis research society—schwab adult spinal deformity classification: a validation study. Spine 37:1077–1082. https://doi.org/10.1097/BRS.0b013e31823e15e2
Glassman SD, Bridwell K, Dimar JR, Horton W, Berven S, Schwab F (2005) The Impact of positive sagittal balance in adult spinal deformity. Spine 30:2024–2029. https://doi.org/10.1097/01.brs.0000179086.30449.96
Lafage V, Schwab F, Patel A, Hawkinson N, Farcy J-P (2009) Pelvic tilt and truncal inclination: two key radiographic parameters in the setting of adults with spinal deformity. Spine 34:E599–E606. https://doi.org/10.1097/BRS.0b013e3181aad219
Schwab FJ, Blondel B, Bess S, Hostin R, Shaffrey CI, Smith JS, Boachie-Adjei O, Burton DC, Akbarnia BA, Mundis GM, Ames CP, Kebaish K, Hart RA, Farcy J-P, Lafage V, International Spine Study G (2013) Radiographical spinopelvic parameters and disability in the setting of adult spinal deformity: a prospective multicenter analysis. Spine (Phila Pa 1976) 38:E803-E812 https://doi.org/10.1097/BRS.0b013e318292b7b9
Bridwell KH, Glassman S, Horton W, Shaffrey C, Schwab F, Zebala LP, Lenke LG, Hilton JF, Shainline M, Baldus C, Wootten D (2009) Does treatment (nonoperative and operative) improve the two-year quality of life in patients with adult symptomatic lumbar scoliosis: a prospective multicenter evidence-based medicine study. Spine (Phila Pa 1976) 34:2171–2178. https://doi.org/10.1097/BRS.0b013e3181a8fdc8
Smith JS, Shaffrey CI, Berven S, Glassman S, Hamill C, Horton W, Ondra S, Schwab F, Shainline M, Fu KM, Bridwell K (2009) Improvement of back pain with operative and nonoperative treatment in adults with scoliosis. Neurosurgery 65:86–93; discussion 93–84. https://doi.org/10.1227/01.Neu.0000347005.35282.6c
Blondel B, Schwab F, Ungar B, Smith J, Bridwell K, Glassman S, Shaffrey C, Farcy J-P, Lafage V (2012) Impact of magnitude and percentage of global sagittal plane correction on health-related quality of life at 2-years follow-up. Neurosurgery 71:341–348. https://doi.org/10.1227/NEU.0b013e31825d20c0
Katz JN, Dalgas M, Stucki G, Lipson SJ (1994) Diagnosis of lumbar spinal stenosis. Rheum Dis Clin North Am 20:471–483
Takahashi K, Miyazaki T, Takino T, Matsui T, Tomita K (1995) Epidural pressure measurements. relationship between epidural pressure and posture in patients with lumbar spinal stenosis. Spine (Phila Pa 1976) 20:650–653
Farrokhi MR, Haghnegahdar A, Rezaee H, Sharifi Rad MR (2016) Spinal sagittal balance and spinopelvic parameters in patients with degenerative lumbar spinal stenosis; a comparative study. Clin Neurol Neurosurg 151:136–141. https://doi.org/10.1016/j.clineuro.2016.10.020
Suzuki H, Endo K, Kobayashi H, Tanaka H, Yamamoto K (2010) Total sagittal spinal alignment in patients with lumbar canal stenosis accompanied by intermittent claudication. Spine (Phila Pa 1976) 35:E344-346. https://doi.org/10.1097/BRS.0b013e3181c91121
Buckland AJ, Vira S, Oren JH, Lafage R, Harris BY, Spiegel MA, Diebo BG, Liabaud B, Protopsaltis TS, Schwab FJ, Lafage V, Errico TJ, Bendo JA (2016) When is compensation for lumbar spinal stenosis a clinical sagittal plane deformity? Spine J 16:971–981. https://doi.org/10.1016/j.spinee.2016.03.047
Higgins JPT Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA (editors) (2019) Cochrane handbook for systematic reviews of interventions version 6.0 (updated July 2019). In. Cochrane
Dohzono S, Toyoda H, Hori Y, Takahashi S, Suzuki A, Terai H, Nakamura H (2018) Spinopelvic sagittal alignment after microendoscopic laminotomy in patients with lumbar degenerative spondylolisthesis. J Neurol Surg Part A Cent Eur Neurosurg 79:479–485. https://doi.org/10.1055/s-0038-1666847
Shin EK, Kim CH, Chung CK, Choi Y, Yim D, Jung W, Park SB, Moon JH, Heo W, Kim SM (2017) Sagittal imbalance in patients with lumbar spinal stenosis and outcomes after simple decompression surgery. Spine J Off J North Am Spine Soc 17:175–182. https://doi.org/10.1016/j.spinee.2016.08.023
Transfeldt EE, Topp R, Mehbod AA, Winter RB (2010) Surgical outcomes of decompression, decompression with limited fusion, and decompression with full curve fusion for degenerative scoliosis with radiculopathy. Spine 35:1872–1875. https://doi.org/10.1097/BRS.0b013e3181ce63a2
Arai Y, Hirai T, Yoshii T, Sakai K, Kato T, Enomoto M, Matsumoto R, Yamada T, Kawabata S, Shinomiya K, Okawa A (2014) A prospective comparative study of 2 minimally invasive decompression procedures for lumbar spinal canal stenosis: unilateral laminotomy for bilateral decompression (ULBD) versus muscle-preserving interlaminar decompression (MILD). Spine 39:332–340. https://doi.org/10.1097/brs.0000000000000136
Chang HS (2018) Effect of sagittal spinal balance on the outcome of decompression surgery for lumbar canal stenosis. World Neurosurg 119:e200–e208. https://doi.org/10.1016/j.wneu.2018.07.104
Chang HS (2018) Influence of lumbar lordosis on the outcome of decompression surgery for lumbar canal stenosis. World Neurosurg 109:e684–e690. https://doi.org/10.1016/j.wneu.2017.10.055
Dohzono S, Toyoda H, Takahashi S, Matsumoto T, Suzuki A, Terai H, Nakamura H (2016) Factors associated with improvement in sagittal spinal alignment after microendoscopic laminotomy in patients with lumbar spinal canal stenosis. J Neurosurg Spine 25:39–45. https://doi.org/10.3171/2015.12.spine15805
Fujii K, Kawamura N, Ikegami M, Niitsuma G, Kunogi J (2015) Radiological improvements in global sagittal alignment after lumbar decompression without fusion. Spine 40:703–709. https://doi.org/10.1097/brs.0000000000000708
Hikata T, Watanabe K, Fujita N, Iwanami A, Hosogane N, Ishii K, Nakamura M, Toyama Y, Matsumoto M (2015) Impact of sagittal spinopelvic alignment on clinical outcomes after decompression surgery for lumbar spinal canal stenosis without coronal imbalance. J Neurosurg Spine 23:451–458. https://doi.org/10.3171/2015.1.spine14642
Ikuta K, Masuda K, Tominaga F, Sakuragi T, Kai K, Kitamura T, Senba H, Shidahara S (2016) Clinical and radiological study focused on relief of low back pain after decompression surgery in selected patients with lumbar spinal stenosis associated with grade I degenerative spondylolisthesis. Spine 41:E1434–e1443. https://doi.org/10.1097/brs.0000000000001813
Jeon CH, Lee HD, Lee YS, Seo HS, Chung NS (2015) Change in sagittal profiles after decompressive laminectomy in patients with lumbar spinal canal stenosis. Spine 40:E279–E285. https://doi.org/10.1097/brs.0000000000000745
Kanbara S, Urasaki T, Tomita H, Ando K, Kobayashi K, Ito K, Tsushima M, Matsumoto A, Morozumi M, Tanaka S, Ota K, Machino M, Ito S, Nishida Y, Ishiguro N, Imagama S (2018) Editors’ choice surgical outcomes of decompressive laminoplasty with spinous process osteotomy to treat lumbar spinal stenosis. Nagoya J Med Sci 80:1–9. https://doi.org/10.18999/nagjms.80.1.1
Madkouri R, Brauge D, Vidon-Buthion A, Fahed E, Mourier KL, Beaurain J, Grelat M (2018) Improvement in sagittal balance after decompression surgery without fusion in patients with degenerative lumbar stenosis: clinical and radiographic results at 1 year. World Neurosurg 114:e417–e424. https://doi.org/10.1016/j.wneu.2018.03.002
Ogura Y, Shinozaki Y, Kobayashi Y, Kitagawa T, Yonezawa Y, Takahashi Y, Yoshida K, Yasuda A, Ogawa J (2019) Impact of decompression surgery without fusion for lumbar spinal stenosis on sagittal spinopelvic alignment: minimum 2-year follow-up. J Neurosurg Spine. https://doi.org/10.3171/2018.11.spine181092
Son S, Lee SG, Kim WK, Ahn Y (2018) Advantages of a microsurgical translaminar approach (keyhole laminotomy) for upper lumbar disc herniation. World Neurosurg 119:e16–e22. https://doi.org/10.1016/j.wneu.2018.06.004
Xia YP, Ishii K, Matsumoto M, Nakamura M, Toyama Y, Chiba K (2008) Radiographic predictors of residual low back pain after laminectomy for lumbar spinal canal stenosis - Minimum 5-year follow-up. J Spinal Disord Tech 21:153–158. https://doi.org/10.1097/BSD.0b013e318074dded
Xia YP, Xu TT, Shen QF, Zhang Xue-Li XL, Jiang H, Tian R (2008) Radiographic predictors of residual low back pain after laminectomy for lumbar canal stenosis: a minimum of 6-year follow-up. Chin J Traumatol Engl Ed 11:135–140. https://doi.org/10.1016/S1008-1275(08)60029-2
Youn MS, Shin JK, Goh TS, Son SM, Lee JS (2018) Endoscopic posterior decompression under local anesthesia for degenerative lumbar spinal stenosis. J Neurosurg Spine 29:661–666. https://doi.org/10.3171/2018.5.spine171337
Overdevest GM, Jacobs W, Vleggeert-Lankamp C, Thome C, Gunzburg R, Peul W (2015) Effectiveness of posterior decompression techniques compared with conventional laminectomy for lumbar stenosis. Cochrane Database Syst Rev: cd010036. https://doi.org/10.1002/14651858.CD010036.pub2
Bernhardt M, Bridwell KH (1989) Segmental analysis of the sagittal plane alignment of the normal thoracic and lumbar spines and thoracolumbar junction. Spine 14:717–721. https://doi.org/10.1097/00007632-198907000-00012
Lenke LG, Betz RR, Harms J, Bridwell KH, Clements DH, Lowe TG, Blanke K (2001) Adolescent idiopathic scoliosis: a new classification to determine extent of spinal arthrodesis. J Bone Jt Surg Am 83:1169–1181
Teles AR, Mattei TA, Righesso O, Falavigna A (2017) Effectiveness of operative and nonoperative care for adult spinal deformity: systematic review of the literature. Glob Spine J 7:170–178. https://doi.org/10.1177/2192568217699182
Smith JS, Shaffrey CI, Kelly MP, Yanik EL, Lurie JD, Baldus CR, Edwards C, Glassman SD, Lenke LG, Boachie-Adjei O, Buchowski JM, Carreon LY, Crawford CH 3rd, Errico TJ, Lewis SJ, Koski T, Parent S, Kim HJ, Ames CP, Bess S, Schwab FJ, Bridwell KH (2019) Effect of serious adverse events on health-related quality of life measures following surgery for adult symptomatic lumbar scoliosis. Spine (Phila Pa 1976) 44:1211–1219. https://doi.org/10.1097/BRS.0000000000003036
Barrey C, Jund J, Perrin G, Roussouly P (2007) Spinopelvic alignment of patients with degenerative spondylolisthesis. Neurosurgery 61:981–986; discussion 986. https://doi.org/10.1227/01.neu.0000303194.02921.30
Rhee C, Visintini S, Dunning CE, Oxner WM, Glennie RA (2017) Does restoration of focal lumbar lordosis for single level degenerative spondylolisthesis result in better patient-reported clinical outcomes? a systematic literature review. J Clin Neurosci 44:95–100. https://doi.org/10.1016/j.jocn.2017.06.039
Försth P, Ólafsson G, Carlsson T, Frost A, Borgström F, Fritzell P, Öhagen P, Michaëlsson K, Sandén B (2016) A randomized, controlled trial of fusion surgery for lumbar spinal stenosis. N Engl J Med 374:1413–1423. https://doi.org/10.1056/NEJMoa1513721
Zarate-Kalfopulos B, Reyes-Tarrago F, Navarro-Aceves LA, Garcia-Ramos CL, Reyes-Sanchez AA, Alpizar-Aguirre A, Rosales-Olivarez LM (2019) Characteristics of spinopelvic sagittal alignment in lumbar degenerative disease. World Neurosurg 126:e417–e421. https://doi.org/10.1016/j.wneu.2019.02.067
Bayerl SH, Pöhlmann F, Finger T, Onken J, Franke J, Czabanka M, Woitzik J, Vajkoczy P (2015) The sagittal balance does not influence the 1 year clinical outcome of patients with lumbar spinal stenosis without obvious instability after microsurgical decompression. Spine 40:1014–1021. https://doi.org/10.1097/brs.0000000000000928
Beyer F, Geier F, Bredow J, Oppermann J, Eysel P, Sobottke R (2015) Influence of spinopelvic parameters on non-operative treatment of lumbar spinal stenosis. Technol Health Care 23:871–879. https://doi.org/10.3233/thc-151032
Liang C, Sun J, Cui X, Jiang Z, Zhang W, Li T (2016) Spinal sagittal imbalance in patients with lumbar disc herniation: its spinopelvic characteristics, strength changes of the spinal musculature and natural history after lumbar discectomy. BMC Musculoskelet Disord 17:305. https://doi.org/10.1186/s12891-016-1164-y
Ogura Y, Kobayashi Y, Shinozaki Y, Ogawa J (2019) Spontaneous correction of sagittal spinopelvic malalignment after decompression surgery without corrective fusion procedure for lumbar spinal stenosis and its impact on clinical outcomes: a systematic review. J Orthop Sci. https://doi.org/10.1016/j.jos.2019.05.021
Andreisek G, Imhof M, Wertli M, Winklhofer S, Pfirrmann CW, Hodler J, Steurer J (2013) A systematic review of semiquantitative and qualitative radiologic criteria for the diagnosis of lumbar spinal stenosis. AJR Am J Roentgenol 201:W735-746. https://doi.org/10.2214/ajr.12.10163
Funding
Open access funding provided by University of Turku (UTU) including Turku University Central Hospital.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hatakka, J., Pernaa, K., Rantakokko, J. et al. Effect of lumbar laminectomy on spinal sagittal alignment: a systematic review. Eur Spine J 30, 2413–2426 (2021). https://doi.org/10.1007/s00586-021-06827-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-021-06827-y