Abstract
Purpose
Although anterior and posterior decompression surgery are both reported to treat patients with myelopathy caused by ossification of the posterior longitudinal ligament (OPLL). The surgical strategy of the disease is still controversial when the OPLL is multilevel and severe. This present study reports the preliminary clinical results of a novel technique named anterior controllable antidisplacement and fusion (ACAF) for the treatment of multilevel-severe OPLL with myelopathy.
Methods
A series of 15 patients with cervical myelopathy caused by compression of multilevel severe OPLL were enrolled. All the patients underwent ACAF after thorough surgical designing based on preoperative imaging. The patients were followed for a mean follow-up duration of 9 months in this study. The main surgical procedures include discectomy of the involved levels, thinning of the anterior part of the involved vertebrae, intervertebral cages, anterior plate and screws installation, bilateral osteotomies of the vertebrae, and antedisplacement of the vertebrae-OPLL complex (VOC). The Japanese Orthopaedic Association (JOA) scales, Visual Analog Scale (VAS) were studied. And the pre- and postoperative radiological parameters, and surgical complications were also investigated.
Results
Postoperative CT and MRI showed complete decompression of the cord by antidisplacement of the VOC. Restoration of neurological defects was confirmed at the last follow-up assessment. Bone fusion was confirmed by CT at 6 months follow-up. No specific complications were identified that were associated with this technique.
Conclusions
The present study demonstrates that excellent postoperative outcome can be achieved with the use of the ACAF. Though further study is required to confirm the conclusion, this novel technique has the potential to serve as an alternative surgical technique for the treatment of cervical OPLL.
Graphical abstract
These slides can be retrieved under Electronic Supplementary Material.

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ossification of the posterior longitudinal ligament (OPLL) is frequently related to cervical myelopathy [1]. The incidence of OPLL among Chinese people is reported to be about 3% [2]. The two main surgical strategies used for the treatment of OPLL are anterior decompression, via anterior cervical corpectomy and fusion (ACCF) or anterior decompression and fusion with floating method (ADF), and posterior decompression, via laminoplasty or laminectomy [3, 4]. Although anterior decompression and posterior decompression surgery are both reported to treat patients with myelopathy caused by OPLL. The shortcomings of the two techniques are both obvious [5, 6].
Anterior decompression surgery directly relieves the cervical spinal cord that has been invaded by OPLL, while at the same time restoring the spinal cord to its original morphology. Its satisfactory clinical outcome has been proven by a large clinical study [7, 8]. However, when dealing with severe OPLL, the anterior decompression becomes a technical challenge. The incidence rate of complications includes cerebrospinal fluid (CSF) leakage, hardware failure, neural injury, or insufficient decompression was reported higher than posterior decompression surgery. And the complications increase with the number of involved segments when anterior surgical approaches are used [9, 10].
Posterior decompression surgery is safer than the anterior decompression surgery for it needs no resection of the ventral compression component [7]. The effect of the indirect posterior decompression relies much on the cervical lordosis alignment to allow the spinal cord floating away from ventral compression. In cases with bad cervical lordosis or severe OPLL lesion, the neurological improvement is always diminished [11, 12].
To combine the advantages and avoid shortcomings of anterior and posterior decompression surgery, we have designed a novel technique named anterior controllable antidisplacement and fusion (ACAF) surgery that can achieve anterior direct decompression without cutting the OPLL. This present report is to introduce and demonstrate the primary clinical result of the novel technique.
Materials and methods
Patient population
Fifteen multilevel severe OPLL patients with myelopathy admitted to the Second Department of Spine Surgery at the Changzheng Hospital in Shanghai were prospectively identified and managed from July 2016 to January 2017. All patients underwent ACAF by the same surgical team. The inclusion criteria: (1) OPLL involved three or more than three vertebrae; (2) OPLL occupation ratio more than 60% [13]; (3) the patient with myelopathy caused by OPLL. The exclusion criteria: (1) patients with myelopathy caused by other diseases such as disc herniation or ossification of the ligamentum flavum; (2) with a history of injury; (3) with a history previous surgery. This study was approved by the institutional review board of our institution, and all patients signed informed consent.
Clinical evaluation
Follow-up was conducted in all patients for at least 6 months. A Visual Analog Scale (VAS) was used to measure neck pain and arm pain. Japanese Orthopaedic Association (JOA) score were used to assess the degree of disability. An improvement rate (IR) of neurologic function was calculated as IR = (postoperative JOA score − preoperative JOA score/17 − preoperative JOA score)/100%. Surgical outcome was defined by the IR as follows: excellent (IR ≥ 75%), good (75% > IR ≥ 50%), fair (50% > IR ≥ 25%), and poor (IR < 25%).
Radiologic evaluation
All patients had preoperative and postoperative plain radiographs, computed tomography (CT) scans and three-dimensional reconstruction, and magnetic resonance images (MRI). The parameters described as follows were investigated: (1) cervical lordosis was measured as the angle between a line parallel to the posterior aspect of the C2 vertebral body and that of the C7 body; (2) the rate of narrowing in the spinal canal is calculated by occupation ratio (OR) and space available for the cord (SAC). OR was defined as the thickness of OPLL divided by the anteroposterior diameter of the spinal canal on the axial CT images; (3) extent and type of OPLL was also investigated in the CT sagittal reconstruction images; (4) fusion was determined by CT. CT criteria for fusion includes bridging bone inside or outside the graft and no lucencies extending > 50% of the graft-host interface. Flexion and extension views of lateral plain X-ray were performed to ensure that no pseudoarthrosis exists. (5) MRIs are used to evaluate the compression to the neural elements.
Preoperative design
The preoperative design should be performed according to patient’s symptoms, signs, and preoperative images. The thickness of the OPLL mass in each level is measured to determine how much of the corresponding anterior vertebral body will be resected. The width of the vertebrae–OPLL complex (VOC) to be created is determined by measuring the widest part of the OPLL mass (Figs. 1, 2). In most of the cases, the widest part of the OPLL mass is no wider than the distance between the uncovertebral joints. Therefore, we always chose uncovertebral joints as the bilateral border of VOC. The computed tomography angiography (CTA) was conducted preoperatively to evaluate the anatomic variation of vertebral artery. The anterioposterior diameter of the involved vertebrae is measured to choose the optimal length of vertebral screw during the hoisting procedure.
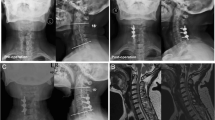
A 59-year-old male presented with clumsiness bilateral hands and spastic gait. Preoperative imaging studies demonstrated a segmental type multilevel severe OPLL at C3–C7 levels. The occupation ratio was 75%. After an ACAF from C3 to C6, he had a significant neurologic recovery, and JOA score increased from 7 to 15 points with an improvement rate of 80%. a Preoperative CT sagittal reconstruction demonstrated a kyphotic alignment in the cervical spine. The dash line square indicates the VOC levels. b Postoperative CT sagittal reconstruction demonstrated satisfactory antedisplacement of VOC from C3 to C6. Cervical lordosis and space available for the cord were restored. c Postoperative CT coronal reconstruction demonstrated the bilateral osteotomies conducted at the inner border of uncovertebral joints. d Preoperative CT demonstrated the OPLL at C4. e Postoperative CT demonstrated complete decompression occupation ratio decreased to 10%. f Preoperative T2-weighted MRI demonstrated severe compression of the spinal cord anteriorly. g Postoperative T2-weighted MRI showed the decompression. OPLL ossification of the posterior longitudinal ligament, ACAF anterior cervical corpectomy and fusion, JOA Japanese Orthopedic Association, CT computed tomography, VOC vertebrae-OPLL complex, MRI magnetic resonance images
Illustration of the procedures of the Bridge Crane technique. a Axial plane of level of huge OPLL mass (#). b Bilateral border of the OPLL mass (dashed lines). c Osteotomy in the contralateral side to the surgeon. d Installation of the “bridge”. e Osteotomy in the ipsilateral side to the surgeon. Dash lines indicates the VOC. f Antedispacement of the VOC. OPLL ossification of the posterior longitudinal ligament, VOC vertebrae-OPLL complex
Surgical technique
Anesthesia, positioning, and exposure
After general endotracheal anesthesia, patient was placed in a supine position appropriately padded under the shoulders and neck. Neurophysiologic monitoring involving somatosensory-evoked potentials (SSEPs) and motor-evoked potentials (MEPs) is utilized to predict the postoperative neurologic deficit. Exposure may be obtained through a right- or left-sided Smith–Robinson approach. Once the anterior surface of the cervical spine is exposed, subperiosteal dissection of the longus colli muscle of the involved segments is performed to the lateral to expose the uncovertebral joint. It is helpful to note or mark the midline before elevating the longus to maintain symmetric dissection.
Discectomy of the involved levels
Routine discectomies are carried out in the involved levels. The involved levels are defined as the discs with OPLL and one disc superior and one disc inferior to the OPLL. Kerrison rongeurs and high-speed burrs can be used to remove the overhanging osteophytes in the anterior and posterior endplates to gain a parallel roof and floor to avoid any obstacles in the hoisting procedure of the VOC. All bony resection and dissection should be extended laterally to the uncovertebral joints. In the discs, most caudal and cranial of the involved levels, the resection of posterior longitudinal ligament is required to facilitate the later hoisting of the VOC. In cases that the OPLL excess the caudal or cranial border of the VOC, transection of OPLL is required. After discectomy, the OPLL was meticulously separated from the PLL using the microdissector and then transected by 1–2 mm Kerrison rongeur in a piecemeal pattern. On the contrary, no resection of the posterior longitudinal ligament is needed in the levels of VOC (Figs. 2, 3).
Illustration of the idea of the Bridge Crane technique. a Demonstration of the preoperative sagittal plane CT of the OPLL. The C2–C7 are the involved levels in this case. And C3–C6 are the levels of VOC. b installation of the “bridge” c antedispacement of the VOC. d The VOC is hoisted by the anterior plate and screw as the way of Bridge Crane hoisting a container. CT computed tomography, VOC vertebrae-OPLL complex
Resection of the anterior vertebral bodies of the VOC
Resection of the anterior vertebral bodies of the VOC is performed by a Leksell rongeurs or a high-speed burr as preoperative planning. During the procedure, a prebent anterior cervical plate can be temporarily placed to evaluate if the space between the plate and remaining vertebral body is enough for the hoisting of the OPLL.
Installation of the intervertebral cages and anterior cervical plate (installation of the “bridge”)
Intervertebral carbon fiber cages with a lordosis of 7° filled with autogenic bone harvested from the previous procedure were then inserted at involved levels for further fusion. The height of each intervertebral cage should be based on the disc space heights to avoid excessive friction generated between the endplate of VOC and adjacent endplates during the hoisting procedure. The prebent anterior cervical titanium alloy plate is then placed. The screws are installed after proper drilling and taping on the remaining vertebral bodies. The screws used in this procedure should not be too short for enough purchase of the vertebral body. No attempt should be made on hoisting the VOC in this procedure. After the placement of the intervertebral cages, anterior cervical plate and screws (serve as the bridge for further hoisting of the VOC), the VOC is temporarily stabilized for the next procedure, bilateral osteotomies for the complete isolation of the VOC from the surrounding bone (Figs. 2, 3).
Bilateral osteotomies of the VOC
Bilateral troughs are created among the widest edge of the OPLL. We use a 2-mm high-speed cutting burr or piezosurgery to thin the corticocancellous bone first and 1–2 mm Kerrison rongeurs to remove the posterior vertebral wall on the bottom of the troughs. Osteotomy can be done in the contralateral side to the surgeon before the installation of the “bridge” to avoid the interruption of the surgeon’s operation from the “bridge” (Figs. 1, 2). After the removal of the posterior vertebral wall, we have reached the dorsal side of the lateral posterior longitudinal ligament, with OPLL to the medial, nerve roots to the ventral, pedicles and intervertebral foramens to the lateral. We suggest not resecting the lateral posterior longitudinal ligament on the bottom of the troughs for three reasons. First, the lateral posterior longitudinal ligament is quite thin and will not hinder the hoisting of VOC. Second, reserving the lateral posterior longitudinal ligament reduced the risk of injury to the nerve roots. Last but not least, by leaving the lateral posterior longitudinal ligament along, the main blood supply to the VOC is reserved to facilitate further fusion [14]. Intraoperative CT reconstruction can be done with O-arm to confirm the VOC is fully isolated from the spine.
Hoisting (controllable antidisplacement) of the VOC
This procedure termed the “Bridge Crane technique,” is illustrated in Fig. 2. The VOC is hoisted via gradually tightening the screws in each vertebra at the same pace with screw drives (Fig. 2c). The hoisting, or we called it controllable antidisplacement, of the VOC can be directly observed as the remaining vertebral bodies are getting closer to the “bridge”. If the antidisplacement is not observed, surgeon should stop tightening the screws and check if the VOC is completely isolated from the surrounding bone by detecting with a nerve hook or taking an intraoperative CT image. The antidisplacement of the VOC should not be too much to prevent tear of the dura mater. Excessive blood loss is usually encountered after the antidisplacement of the VOC, which can be easily controlled by the usage of hemostasis materials. Before the closure of the wound, autogenous or allograft bone pieces were graft into the bilateral troughs to obtain further fusion of the VOC with the surrounding bone. The bilateral longus colli were reconstructed to the anterior plate by 3-0 silk sutures to cover the graft bones.
Postoperative immobilization
A halo vest is routinely used postoperatively for external bracing for 3 months. The halo vest maintains the alignment of the cervical spine to prevent failure of the internal fixation and enables early mobility of the patients.
Statistical analysis
Statistical analysis was performed using SPSS [15]. Preoperative and follow-up data such as JOA score, VAS score, OR, and SAC were compared using paired t test. The level of significance was set at p < 0.05.
Results
Clinicopathologic characteristics
The study group included nine male patients and six female patients with a mean age of 62 years (range 52–78 years). The mean duration of follow-up was 9 months (range 6–12 months). The spinal level of severe OPLL involvement was C3–C5 for three patients, C3–C6 for six, C4–C6 for four, and C4–C7 for two. The mean time of symptom duration was 36 months (range 2–214 months).
The mean operative time was 173 min (range 120–230 min), with the mean blood loss of 326 ml (range 250–800 ml). The mean JOA score increased from 9.1 ± 1.4 (range 5–14) at preoperation to 14.5 ± 1.2 (range 9–16) at the 6-month follow-up (p < 0.05). The average improvement rate (IR) was 65.2 ± 9.8%. Five (33.3%) patients were graded as excellent, seven (46.7%) as good, and three (20%) as fair. The average VAS was 5.2 ± 1.6 (range 2–7) preoperatively, 2.4 ± 1.6 (range 0–5) at the 6-month follow-up (p < 0.05). All the patients felt immediate release of numbness in upper limbs. The clinical data is summarized in Table 1.
Radiological results
The radiological outcomes are shown in Table 2. The OR decreased from 67.3 ± 5.1% preoperatively to 16.4 ± 6.5% postoperatively, which was statistically significant (p < 0.05). SAC increased from 4.5 ± 2.1 mm preoperatively to 11.6 ± 2.7 mm postoperatively, which was statistically significant (p < 0.05). Postoperative cervical lordosis was 18° ± 4.7° compared with preoperative 3.7° ± 4.2°. A fusion rate of 100% was achieved at 6-month follow-up. No pseudoarthrosis exists were ensured by flexion and extension views of lateral plain X-ray 6 months after the surgery. The imaging of a typical case is shown in Figs. 1 and 4.
A 57-year-old female presented with walking disturbance and dysfunction of sensation of lower limbs without other complains. After ACAF from C3 to C6, She had neurologic improvement and JOA score increased from 13 to 16 points with an improvement rate of 75%. a Preoperative sagittal CT image (left) shows a massive mixed type OPLL at C2–5 and C6, and postoperative sagittal CT image (right) shows the ossification was transected behind the C2, and the C3–7 VOC was hoisted and fused to an antedisplaced location. Preoperative axial CT image (up) and postoperative axial CT image (down) shows the massive OPLL mass and antedisplacement of the VOC at C3 (b), C5 (c), and C6 (d). e Preoperative sagittal MRI (left) shows severe compression of the spinal cord from C3 to C6, and postoperative sagittal MR image (right) shows sufficient decompression of the spinal cord. f Preoperative axial MRI (up) and postoperative axial MRI (down) shows decompression of the spinal cord. g Preoperative (left) and postoperative (right) lateral tomograph. ACAF anterior cervical corpectomy and fusion, JOA Japanese Orthopedic Association, CT computed tomography, OPLL ossification of the posterior longitudinal ligament, VOC vertebrae-OPLL complex, MRI magnetic resonance images
Surgical complication
Transient minor neurological deterioration, manifesting with the weakness of right arm occurred in one case (6.7%) after operation due to the irritation of the nerve root when dealing with the massive ossification mass on the very lateral of C4/5 segment. The weakness in right arm improved significantly 2 weeks following the surgery. The muscle strength of the right arm was improved to 4+ at the 6 months follow-up. The CSF leak occurred in one case (6.7%) during transecting the continuous OPLL in the cranial margin. In that case, lumbar drainage is performed at the second postoperative day and the CSF leak is then controlled and recovered 1 week after drainage. There was no occurrence of postoperative hematoma. No instrumented failure was observed during follow-up. No specific complications were identified that were associated with this technique.
Discussion
Although anterior and posterior decompression surgery are both reported to treat patients with myelopathy caused by OPLL, controversy still exists over the surgical options. The posterior decompression surgery can safely achieve the decompression of the spinal cord that results from extensive OPLL. However, there are limitations in posterior decompression surgery, it does not always produce the expected space for cases with a highly narrowed spinal canal or a bad lordosis [16, 17]. Furthermore, the posterior decompression surgery is accused of higher incidence of C5 palsy, postoperative kyphotic change, and postoperative progression of the ossified OPLL lesion in many studies [18,19,20]. Anterior corpectomy and resection of OPLL can achieve direct decompression and gain satisfactory results, especially in cases with highly narrowed spinal canal and short segmental OPLL [21]. However, in this high technically demanding surgery, the risks of excessive hemorrhage, iatrogenic damage to neural tissue, and CSF leakage are reported as frequently occurring complications [22, 23]. In 1999, Yamaura et al. reported the anterior decompression surgery with floating method for cervical OPLL [24]. The method minimizes surgical invasion and the risk of hemorrhage, spinal cord damage, and leakage of CSF in traditional anterior decompression surgery. However, the anteriorly migration of the OPLL in floating method is not controlled by surgeon and much owing to the pressure of the CSF, which requires as long as 8 weeks for the ossification to complete the migration [25]. Thus, the impingement between OPLL and the surrounding bone cannot be foreseen during the procedure and often results in incomplete floating of the OPLL [26].
Intraoperative CT scan was used to ensure that the VOC was isolated prior to the hoist. Intraoperative CT scan was also used in the floating method to avoid insufficient decompression [26]. However, the intraoperative CT scan is not the only way to know if the VOC is fully isolated, surgeon could also use a nerve hook to detect the border of the VOC. The space for hoist can also be measured using a depth detector when bridge was set. So the intraoperative CT scan is not a must-have in any procedure in the ACAF technique. The use of the O-arm was useful in initial cases but was omitted from subsequent cases in a bid to reduce radiation for the patient.
The idea of ACAF is to isolate and “actively transport” the OPLL ventrally to restore the space of the spinal canal and thus achieve direct decompression of the neural elements with their location unchanged. The remaining vertebral body and OPLL mass are then served as an autogenous bone for the reconstruction of the anterior column. Different from the floating method, in ACAF the antidisplacement of the OPLL is achieved under the gradual hoisting force of the anterior plate and screws with an immediate feedback. The Bridge Crane device is vital in this technique, for it stabilizes the VOC in the procedure of osteotomy and gives the surgeon full control in the procedure of VOC antidisplacement.
As shown in the results, the patients who underwent ACAF have all gained satisfactory outcome. The anatomic basic for the clinical effect of ACAF lays in the director decompression for spinal cord and nerve roots. The procedure of bilateral osteotomies of the vertebrae gives enough decompression to the bilateral nerve roots. The antidisplacement of the VOC further lead to direct and thorough decompression of the spinal cord. The outcome of the ACAF relies on the complete isolation and antidisplacement of the VOC, which require thorough preoperative designing, proper selection and installation of internal fixation devises, and intraoperative evaluation and trimming of the VOC. Goel et al. reported that stand-alone fixation resulted in dramatic and sustained neurological recovery of OPLL. The authors proposed that instability plays a role in the pathogenesis and development of symptoms related to myelopathy of OPLL. As in ACAF, the satisfactory clinical recovery may also come from the stabilization and fusion of the spinal segments [27].
CSF leakage is one of the most frequently met complications of the anterior decompression surgery for OPLL [28]. In the ACCF surgery, the direct decompression is accomplished by resecting the OPLL in a piecemeal or enbloc pattern. During the resection of OPLL, the adhesion or ossified dura is vulnerable and dural tear is always encountered. Especially when there is a big area of ossified dura, the direct resection will lead to a big absence of dura and refractory postoperative CSF leak. The advantage of ACAF is that it does not need to deal with the adhesion of the dura and OPLL or ossified dura. Isolation of the VOC is conducted on the lateral side of the OPLL. In cases that the OPLL excess the lateral or caudocranial border of the VOC, separation of the dura and OPLL is required. But in this circumstance, the separation of dura and OPLL is safer because it is near the border of OPLL where the adhesion and compression are both mild. There were 6.7% patients with CSF leakage in this study, which was lower than that in anterior decompression and higher than the posterior decompression [29, 30].
Many studies have reported that the blood loss is larger in anterior decompression surgery than that in posterior decompression surgery. The massive compression in the spinal canal obstructs the vein drainage and lead to excessive blood loss, which is one of the reasons for unclear surgical view and inadequate decompression [31].
In our practice, the average blood loss is 328 ml in average and no postoperative hematoma occurred. In ACAF, the VOC provides as an ideal anchor site for hemostatic materials such as bone wax and gel foam. Another important fact is that the present of VOC occupies the potential space for the formation postoperative hematoma.
The current study has several limitations. Neurologic recovery, occurrence of complications, blood loss, and operative time of ACAF could not be directly compared with those associated with other anterior decompression surgery or posterior decompression surgery. Prospective, randomized, controlled studies may be required to adequately investigate these issues. And long-term follow-up is needed to evaluate the long-term outcome and progression of the ossification.
Our current study has shown that multilevel severe OPLL with myelopathy can obtain a safe and sufficient decompression using the novel ACAF technique. The antidisplacement procedure is controlled using a “bridge crane” device, which eliminates the need for direct resection of the OPLL, therefore, reducing the risk of injury to the dura and spinal cord. No specific complications were identified that were associated with this technique. As the procedure described in this technique is not technically demanding, it should be considered when dealing with cases of multilevel severe OPLL.
References
Epstein N (2002) Diagnosis and surgical management of cervical ossification of the posterior longitudinal ligament.[J]. Spine J 2(6):436
Liu S, Zhang D, Jia P et al (1995) Epidemiology research of ossification of posterior longitudinal ligament of cervical spine. J Jilin Univ 4:411–413
Nd EC, Heller JG, Murakami H (2002) Corpectomy versus laminoplasty for multilevel cervical myelopathy: an independent matched-cohort analysis. Spine 27(11):1168
Jain SK, Salunke PS, Vyas KH et al (2005) Multisegmental cervical ossification of the posterior longitudinal ligament: anterior vs posterior approach. Neurol India 53(3):283–285 (discussion 286)
Cardoso MJ, Koski TR, Aruna G et al (2011) Approach-related complications after decompression for cervical ossification of the posterior longitudinal ligament. Neurosurg Focus 30(3):285–293
Chen Y, Guo Y, Lu X et al (2011) Surgical strategy for multilevel severe ossification of posterior longitudinal ligament in the cervical spine[J]. J Spinal Disord Tech 24(1):24
Tomita K, Nomura S, Umeda S et al (1988) Cervical laminoplasty to enlarge the spinal canal in multilevel ossification of the posterior longitudinal ligament with myelopathy. Arch Orthop Trauma Surg 107(3):148–153
Chen Y, Yang L, Liu Y et al (2014) Surgical results and prognostic factors of anterior cervical corpectomy and fusion for ossification of the posterior longitudinal ligament. PLoS One 9(7):e102008
Shinomiya K, Matsuoka T, Kurosa Y et al (2006) Anterior cervical decompression for cervical myelopathy caused by ossification of the posterior longitudinal ligament (OPLL). Springer, Tokyo, pp 209–218
Pitzen T, Chrobok JJ, Ruffing S et al (2009) Implant complications, fusion, loss of lordosis, and outcome after anterior cervical plating with dynamic or rigid plates: 2-year results of a multi-centric, randomized, controlled study. Spine 34(7):641–646
Hirabayashi K, Watanabe K, Wakano K et al (2001) Expansive open-door laminoplasty for cervical spinal stenotic myelopathy. Spine 8(7):693
Iwasaki M, Kawaguchi Y, Kimura T et al (2002) Long-term results of expansive laminoplasty for ossification of the posterior longitudinal ligament of the cervical spine: more than 10 years follow up. J Neurosurg 96(2 Suppl):180
Fujimori T, Iwasaki M, Okuda S et al (2014) Long-term results of cervical myelopathy due to ossification of the posterior longitudinal ligament with an occupying ratio of 60% or more. Spine 39(1):58
Petty P (1997) Surgical anatomy of the anterior cervical spine: the disc space, vertebral artery, and associated bony structures. Neurosurgery 41(1):769–776
SPSS (Computer program) (2009) Version 18.0. SPSS Inc, Chicago
Baba H, Furusawa N, Chen Q et al (1995) Cervical laminoplasty in patients with ossification of the posterior longitudinal ligaments. Paraplegia 33(1):25–29
Lee DH, Joo YS, Hwang CJ et al (2016) A novel technique to correct kyphosis in cervical myelopathy due to continuous-type ossification of the posterior longitudinal ligament. J Neurosurg Spine 26(3):1
Sakaura H, Hosono N, Mukai Y et al (2003) C5 palsy after decompression surgery for cervical myelopathy: review of the literature. Spine 28(21):2447–2451
Lee SE, Chung CK, Jahng TA et al (2013) Long-term outcome of laminectomy for cervical ossification of the posterior longitudinal ligament. J Neurosurg Spine 18(5):465–471
Kato Y, Iwasaki M, Fuji T et al (1998) Long-term follow-up results of laminectomy for cervical myelopathy caused by ossification of the posterior longitudinal ligament. J Neurosurg 89(2):217–223
Onari K, Akiyama N, Kondo S et al (2001) Long-term follow-up results of anterior interbody fusion applied for cervical myelopathy due to ossification of the posterior longitudinal ligament. Spine 26(5):488
Chen Y, Liu X, Chen D et al (2012) Surgical strategy for ossification of the posterior longitudinal ligament in the cervical spine. Orthopedics 35(8):e1231
Shin JH, Steinmetz MP, Benzel EC et al (2011) Dorsal versus ventral surgery for cervical ossification of the posterior longitudinal ligament: considerations for approach selection and review of surgical outcomes. Neurosurg Focus 30(3):E8
Yamaura I, Kurosa Y, Matuoka T et al (1999) Anterior floating method for cervical myelopathy caused by ossification of the posterior longitudinal ligament. Clin Orthop Relat Res 359(359):27
Matsuoka T, Yamaura I, Kurosa Y et al (2011) Long-term results of the anterior floating method for cervical myelopathy caused by ossification of the posterior longitudinal ligament. Spine 26(3):241–248
Yoshii T, Hirai T, Yamada T et al (2017) Intraoperative evaluation using mobile computed tomography in anterior cervical decompression with floating method for massive ossification of the posterior longitudinal ligament. J Orthop Surg Res 12(1):12
Goel A, Nadkarni T, Shah A et al (2015) Is only stabilization the ideal treatment for ossified posterior longitudinal ligament? Report of early results with a preliminary experience in 14 patients. World Neurosurg 84(3):813–819
Cho JY, Chan CK, Lee SH et al (2012) Management of cerebrospinal fluid leakage after anterior decompression for ossification of posterior longitudinal ligament in the thoracic spine: the utilization of a volume-controlled pseudomeningocele. J Spinal Disord Tech 25(4):E93
Belanger TA, Roh JS, Hanks SE et al (2005) Ossification of the posterior longitudinal ligament Results of anterior cervical decompression and arthrodesis in 61 North American patients. J Bone Jt Surg 87(3):610 (American Volume)
Hannallah D, Lee J, Khan M et al (2008) Cerebrospinal fluid leaks following cervical spine surgery. J Bone Jt Surg Am 90(5):1101–1105
Lin D, Ding Z, Lian K et al (2012) Cervical ossification of the posterior longitudinal ligament: anterior versus posterior approach. Indian J Orthop 46(1):92
Acknowledgements
The technique is designed and performed by the corresponding author Dr. Jiangang Shi. We express deep thanks to the illustrator Xiaomu Zhao. We thank Zhen Huang and Qi Zhang for their technical support of intraoperative CT.
Funding
This study was supported by Grants from the National Natural Science Foundation of China (No. 81650031) and Shanghai Municipal Commission of Health and Family Planning (No. 201640262).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors has any potential conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Sun, J., Shi, J., Xu, X. et al. Anterior controllable antidisplacement and fusion surgery for the treatment of multilevel severe ossification of the posterior longitudinal ligament with myelopathy: preliminary clinical results of a novel technique. Eur Spine J 27, 1469–1478 (2018). https://doi.org/10.1007/s00586-017-5437-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-017-5437-4