Abstract
Purpose
To report our 11-year minimum clinical and radiological outcomes, as well as complications of the Charite III total disc replacement (TDR).
Methods
A total of 35 patients indicated for total disc replacement were implanted with the Charite III prosthesis. Clinical evaluation included visual analog scale (VAS) for back pain and the Oswestry disability index (ODI). Radiological parameters of intervertebral disc height (IDH), range of motion (ROM), lumbar lordosis, lumbar scoliosis and prosthesis position were evaluated. Complications and reoperation rates were also assessed.
Results
Thirty-two patients had a minimum 11-year follow-up, and 33 prostheses were implanted. The mean follow-up time was 11.8 years, ranging from 11.3 to 13.8 years. Twenty-eight patients (87.5 %) had a successful outcome, as defined by the FDA. Reoperation was performed in 2 patients for adjacent segment degeneration and pedicle fracture (1 case each). Both VAS and ODI scores showed significant improvement compared to baseline. At the final follow-up, the ROM of both the index- and adjacent-level showed an obvious decrease. The IDH of the index level showed a tendency to decrease, but the difference was not significant. The IDH of adjacent levels were not significantly affected by the surgery. Mean lumbar lordosis was increased at the final follow-up, and lumbar scoliosis over 3° was observed in 12 patients (37.5 %), with a mean angle of 5.6° (range 3°–12°). Of all 35 prostheses, 15 were left-shifted, 3 were right-shifted and 14 were just in the middle. In the coronal plane, 25 were rated as ideally placed, 5 were discretely shifted, 4 were slightly shifted and 1 was markedly shifted. In the sagittal plane, only 12 prostheses were rated as ideally placed, 14 were discretely shifted and 9 were suboptimally placed. Prosthesis subsidence was noted in 3 (9.4 %) patients (the subsidence distances were 3.1, 4.2 and 2.8 mm, respectively). Heterotopic ossification was detected in 25 segments (71.4 %), consisting of Class-I heterotopic ossification in 7 segments (20.0 %), Class-II in 9 segments (25.7 %), and Class-III in 9 segments (25.7 %). Class-IV heterotopic ossification was not observed.
Conclusion
The cumulative survival was 100 % at a mean follow-up of 11.8 years. Clinical and radiological results were satisfactory and long-term clinical results were maintained for a mean follow-up of 11.8 years. Reoperation and complication rates are acceptable, and our study does not substantiate the fear of reoperation or late complications. The results of our long-term follow-up indicate that, with strict indication, TDR is a safe and effective procedure as an alternative to lumbar fusion.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
As the standard surgical method for symptomatic degenerative disc disease (DDD), fusion has also brought some pertinent problems including pseudoarthrosis and persistent LBP (low back pain) [1–3]. In addition, there is evidence that fusion may accelerate adjacent segment degeneration [4–6], resulting in new pain and in some cases additional surgical intervention.
With the theoretical advantage of restoring kinematics of the intervertebral disc and posterior facet joints [7], lumbar TDR has increased in popularity as an alternative procedure for lumbar fusion since gaining approval by the FDA in 2000. Several randomized [7–12] controlled trials have been carried out to compare TDR with lumbar fusion techniques and found no clinically relevant differences in pain or physical function, thus demonstrating that TDR produces at least equivalent clinical results compared with lumbar fusion. Prospective cohort studies of TDR also showed a significant reduction of pain intensity and improvement of functional impairment over short- to mid-term follow-up [13–17].
However, late problems such as prosthesis subsidence, facet joint arthrosis, and spontaneous fusion have been reported [18–20]. Controversy still exists regarding whether TDR can prevent adjacent segment degeneration in the long term [23–27]. To assess the role of any new treatment method, long-term clinical results need to be evaluated in clinical studies with adequately sized patient cohorts and sufficient long-term results. To date, the number of published long-term follow-up studies on TDR is still very small. This prospective nonrandomized clinical trial aimed to assess the safety and effectiveness of lumbar TDR. The study had three objectives:
-
(1)
To observe the survival rate of the Charite III artificial intervertebral disc.
-
(2)
To assess the effectiveness of Charite III TDR for patients with symptomatic disc degeneration.
-
(3)
To analyze late complications of Charite III TDR.
Materials and methods
Study protocol and patient selection
Between March 1999 and March 2002, a total of 35 patients with one- or two-level symptomatic degenerative disc disease underwent Charite III lumbar TDR. All patients were nonresponders to an intensive inpatient and outpatient conservative treatment program conducted over a period of at least 6 months. The main inclusion criteria were patients with predominant (≥80 %) axial low back pain originating from lumbar DDD. A summary of inclusion and exclusion criteria for this study is listed in Table 1.
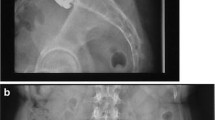
The preoperative diagnosis was made on the basis of clinical signs and symptoms, lumbar radiographs in standard anteroposterior and lateral views, functional flexion/extension images, and also magnetic resonance imaging of the lumbar spine. Discography was used to identify discogenic pain and to distinguish the symptomatic segment (Fig. 1).
Radiographic views of a subject implanted at L4/5 and L5/S1. a–c MRI showed severe degeneration of L4/5 and L5/S1, and obvious disc herniation in L4/5; d, e X-ray showed that ROM of L4/5 and L5/S1 were 5° and 8°, respectively, at 1-year follow-up; f, g ROM of L4/5 and L5/S1 were 4° and 5°, respectively, at 5.5-year follow-up; h, i ROM of L4/5 and L5/S1 were 3° and 5°, respectively, at 12.9-year follow-up, and ROM of the upper adjacent level was 2°
Outcome assessment
Both clinical and radiological outcomes were assessed. Clinical questionnaires examined lumbar function (Oswestry Disability Index, ODI), and lumbar visual analog scale (VAS) for back pain. Clinical success as defined by the FDA (non-validated clinical scale) was achieved when all the following four criteria were met: (1) disability (ODI) improvement of at least 15 points versus baseline; (2) no device failure; (3) no major complication associated with the prosthesis or surgery; (4) no neurological deterioration. Device failure included any reoperation required to modify or remove implants and any need for supplemental fixation. Major complications were defined as major vessel injury resulting in >1500 mL blood loss, neurological damage, or nerve root injury [9].
The radiographic parameters range of motion (ROM), intervertebral disc height (IDH), lumbar lordosis, lumbar scoliosis, and prosthesis position were measured. We used the Cobb technique to determine the range of motion (ROM). We measured the anterior and posterior disc height using digitized radiographs in the neutral position to obtain IDH. Lumbar lordosis is the angle formed between the upper endplate of L1 and the sacral endplate. All radiographs were analyzed by the same system, to avoid inter- and intraobserver variability. Patients with scoliosis were measured on standard anteroposterior radiographs using methods adopted in measuring spinal scoliosis. According to research by Boss et al. [26], coronal centering of the prosthesis was applied to the midline, measured from the pedicles of the two vertebrae. Placement was classified as ideal (<2 mm), discretely shifted (2–3 mm), slightly shifted (3–5 mm), and markedly shifted (>5 mm). Sagittal placement was determined at the index level from the dorsal border of the vertebra, with graduation defined as ideal (<2 mm), discretely shifted (2–3 mm), and suboptimally placed (>3 mm).
Complications of heterotopic ossification (HO) and prosthesis subsidence were also evaluated. Heterotopic ossification was evaluated using a 6-point scale adapted from McAfee et al. [9, 27]. The grades were defined as follows: 0, no evidence of HO; 1, HO present in islands of bone within soft tissue but not influencing segmental motion; 2, bone present within the disc space defined by the planes formed by the two adjacent end plates, but bony protrusions projecting more or less horizontally from the vertebral bodies; 3, bone present between the two planes without bridge; 4, apparent continuous connection of bridging bone between end plates; 5, indeterminate, for example, due to poor film quality.
Surgical technique
All patients underwent surgical treatment through an open anterior retroperitoneal approach. Patients were placed in the supine position on a folding operating table. Fluoroscopy was used to identify the location of the disc space and the approach angle with markings on the patient’s abdomen. A complete discectomy was performed using anterior lumbar surgical instruments. For patients with a herniated disc, complete decompression was performed to relieve the symptoms. Care was taken to preserve the peripheral annulus fibrosis when performing discectomy which can cause ligamentotaxis.
Before inserting the prosthesis, the disc space was prepared in an identical manner to ensure proper positioning of the prosthesis. The anterior longitudinal ligament and anterior annulus fibrosis were resected. The posterior longitudinal ligament was stretched to facilitate restoration of normal disc space height. The bony vertebral endplates were left intact and shaped to be parallel. The subchondral bone on the vertebral endplates was preserved to provide stability and prevent implant subsidence. In some cases, the posterior osteophytes were carefully removed to allow satisfactory placement of the prosthesis. Creation of a flat surface to maximize the bone–implant contact area was of great importance. Final positioning was assessed by fluoroscopy. When the prosthesis was in position, the anterior longitudinal ligament was not fixed to avoid heterotopic ossification.
Statistical analysis
All data were evaluated by an external, independent statistician not involved in the process of pre- or postoperative decision-making. Continuous data were expressed as mean ± standard deviation (SD), and were tested for normality based on the Kolmogorov–Smirnov test. We use the paired t test to examine the differences in VAS, ODI, ROM, IDH, and lumbar lordosis preoperatively and at the final follow-up. All statistical analysis were conducted using SAS, version 9.2 (SAS Institute, Cary, NC), and statistical significance was accepted at the P < 0.05 level.
Results
Study population
A total of 35 patients were implanted with Charite III artificial disc, of whom 32 (35 prostheses) had a minimum of 11 years follow-up. One patient was lost to follow-up, one declined to participate in the study, and one died of an unrelated cause. The mean follow-up was 11.8 years, ranging from 11.3 to 13.8 years. Of the remaining 32 patients, there were 14 men and 18 women with a mean age at the time of surgery of 41.4 years (range 28.6–51.3 years). The mean time of symptomatology preoperatively was 5.4 years.
Clinical outcomes
Analysis of the clinical parameters showed a highly significant improvement in both VAS and ODI values at the final follow-up. Preoperative VAS was 8.50 ± 0.18, which decreased to 1.46 ± 0.32 at the final follow-up (P = 0.0015). Preoperative ODI was 41.36 ± 1.87, which decreased to 13.21 ± 2.38 at the final follow-up (P = 0.0047). Of all patients, 28 patients (87.5 %) had a successful outcome. One patient complained of residual back pain (VAS 4, ODI 36) especially on a cloudy day. The pain could be relieved by NSAIDs and the patient refused to undergo any examination or further treatment.
When asked whether they would undergo the same operation again, 25 (78.1 %) patients answered “certainly yes”, 5 answered “probably yes”, and 2 answered “definitely not”. Of the 32 patients working prior to surgery, 3 had retired (at the age of 60) at the final follow-up, leaving 29 patients to consider. Of those 29 patients, 22 (75.9 %) patients returned to work, 7 were asked not to work by their families.
Radiological outcomes
Of the 35 prostheses, 7 had a ROM of less than 2° and solid fusion was noted in 3 segments. The mean ROM of the other 28 prostheses was 5.4° (range 2°–12°). The ROM of the index level showed a significant decrease compared to a mean angle of 7.4° (range 6°–16°) preoperatively (P = 0.0031). The ROM of adjacent levels also showed statistically significant decreases. The upper adjacent level ROM decreased from 6.7° (range 4°–15°) preoperatively to 5.2° (range 1°–14°) at the final follow-up (P = 0.0143). The lower adjacent level ROM decreased from 4.5° (range 1°–13°) preoperatively to 2.4° (range 0°–6°) at the final follow-up (P = 0.0027).
At the final follow-up, IDH of the index level was decreased 2.1 mm compared to that before surgery (P = 0.0613), which showed a trend towards a significant difference. IDHs of the upper and lower adjacent levels were not significantly affected by the surgery (P1 = 0.2144 and P2 = 0.2658, respectively). Two patients showed an obvious decrease of IDH of the upper adjacent level and heterotopic ossification was observed in the two segments.
Lumbar lordosis showed a statistically significant increase at the final follow-up (P = 0.0328). Lumbar scoliosis over 3° was observed in 12 patients (37.5 %), with a mean angle of 5.6° (range 3°–12°). Of the 12 patients, 7 had left convex curvature. Of the total of 35 prostheses, 15 were left-shifted, 3 were right-shifted and 14 were just in the middle. In the coronal plane, 25 were rated as ideally placed, 5 were discretely shifted, 4 were slightly shifted and 1 was markedly shifted. In the sagittal plane, only 12 prostheses were rated as ideally placed, 14 were discretely shifted and 9 were suboptimally placed. We tried to correlate the degree of scoliosis with the prosthesis position in the coronal plane, but found only a low correlation.
Complications
By the completion of this follow-up, no device failure or major complications had occurred. However, one patient complained of severe leg pain 7 years after TDR and adjacent segment degeneration was confirmed. We performed spinal decompression and implanted an interspinous dynamic system at the upper adjacent level. One patient suffered a pedicle fracture when moving heavy objects 20 months after TDR, and underwent posterior fusion surgery without removal of the prosthesis. During surgery, two patients experienced a tear of the iliac vein, which were repaired immediately, leaving no hematoma after surgery. Two patients developed anhidrosis after surgery, and complained that their feet felt dry. Two patients suffered an abdominal hernia, but experienced no pain. Of all the male patients, no retrograde ejaculation was observed.
At the final follow-up, prosthesis subsidence was noted in 3 patients (9.4 %) (the subsidence distances were 3.1, 4.2 and 2.8 mm, respectively), but no symptoms appeared. Of these 3 patients, prosthesis subsidence was noted at the lower endplate in 2, and at the upper endplate in 1.
Heterotopic ossification was detected in 25 segments (71.4 %). According to McAfee’s classification, there was Class-I heterotopic ossification in 7 segments (20.0 %), Class-II in 9 segments (25.7 %), and Class-III in 9 segments (25.7 %). No instances of Class-IV heterotopic ossification were observed. The average segmental ROM, VAS, and ODI in each class of heterotopic ossification are shown in Table 2. There was no significant difference in the segmental ROM between the patients with Class-I or Class-II heterotopic ossification and those without heterotopic ossification. The segmental ROM in patients with Class-III heterotopic ossification, however, was significantly decreased as compared to patients without heterotopic ossification (P = 0.0142). In the VAS and ODI there was no significant difference between the patients with HO and those without (VAS: P = 0.359, 0.681, 0.217; ODI: P = 0.417, 0.714, 0.316, respectively, for Class I, II, and III).
Discussion
This detailed study represents an independent long-term follow-up of Charite III lumbar TDR carried out for the treatment of symptomatic degenerative disc disease by a single surgeon. This is the first long-term study of the Charite III artificial intervertebral disc used in a Chinese population.
In summary, this long-term follow-up study demonstrates satisfactory results after Charite III TDR in the majority of the evaluated cases with regard to clinical as well as radiological outcome. After a minimum 11-year follow-up, one revision surgery was performed at the upper adjacent level because of adjacent segment degeneration. However, compared with other long-term studies [28–33], we found a more favorable survival rate. There are several possible reasons for this which may be summarized as follows: one is our strict indication for TDR since we can never be too cautious; another important reason may be that most Chinese are much lighter in weight compared to the western population, which may affect the survival rate of the prosthesis since a much smaller continuous load is exerted on the prosthesis; the third reason could be that the surgeon is very familiar with the anterior retroperitoneal approach which helped to reduce and to deal with severe complications.
The clinical results of this study are encouraging, showing a significant improvement of VAS and ODI after a minimum 11-year follow-up. Of the total of 32 patients followed, 29 showed at least a 15-point improvement in ODI score versus baseline at the final follow-up and the clinical success rate was 90.6 % according to the criteria defined by the FDA (non-validated clinical scale). One patient complained of back pain at the final follow-up, but because NSAIDs were effective for her, she refused to undergo any examination. On her X-ray at the final follow-up, the upper adjacent segment showed a severe loss of IDH (3.6 mm) compared to that before surgery, and osteophytes were quite obvious in this segment with a ROM of 1°. We speculate that the degeneration of the upper adjacent level caused the back pain. In this study, 25 (78.1 %) patients answered “certainly yes” when asked whether they would undergo the same operation again, and 22 (75.9 %) patients returned to work after surgery, similar results to those reported in a study by Lemaireet et al. [28].
Our reported mean range of motion at the index level in flexion/extension was higher than that described in a study by Huanget et al. [34] (3.8°), but smaller than that reported in the series of Lemaireet et al. [28] (10.3°). Huang et al. reported that 20 patients (34.5 %) in their series achieved <2° of flexion/extension, whereas Lemaire et al. had 9 patients (9.0 %) with <2° and our series had 7 patients (21.9 %) with <2°. Johnsen et al. [17] explained the reduced movement of the index level by the hindrance of posterior tissues after surgery, location of the axis of rotation, loss of disc height and the natural history of degenerative disc disease. However, in this study, the prosthesis used was the Charite III and the disc height of the 7 patients showed no significant difference from that of the other patients. We noticed that patients with chronic low back pain often develop fear of pain on movement, which combined with permanent hindrance of soft-tissue changes, seems most likely to provide a reasonable explanation. Although the IDH of the index level showed a tendency to decrease, the difference was not significant (P = 0.0613) which suggests that the wear of the polyethylene core is minimal and that prosthesis subsidence is not common if strict inclusion criteria are applied.
It is easy to understand that in short- to mid-term follow-up, ROM of the adjacent segments will increase after TDR. However, few previous long-term studies evaluated ROM and IDH of the adjacent segments. In this study, both adjacent segments showed decreased ROM and unchanged IDH. Adjacent segment degeneration was confirmed in one patient at 7 years after TDR and reoperation was performed at the upper adjacent level. Two patients showed an obvious decrease of IDH of the upper adjacent segment and heterotopic ossification was observed in the two segments. Although adjacent segment degeneration was obvious in these two patients, it was difficult to tell whether this was related to the surgical intervention or simply the natural progression of degeneration. Further, the rate of adjacent segment degeneration after a minimum 11-year follow-up was lower than that of fusion patients in spite of the small number of patients and the lack of a comparative group [5, 25, 35]. This long-term follow-up clearly provides additional evidence for the protective effect of TDR on adjacent segments. We may also conclude that TDR with a Charite III prosthesis will not accelerate adjacent segment degeneration based on this study.
There are reports that fusion can significantly reduce lumbar lordosis [36], and that this reduction correlates with postoperative back pain [37, 38]. However, whether lumbar total disc replacement will influence lumbar lordosis and whether the lumbar lordosis correlates with clinical outcome are controversial [39–41]. In this study, lumbar lordosis showed a statistically significant increase at the final follow-up. We noticed that patients with less lumbar lordosis showed some increase, while those with greater lumbar lordosis showed some decrease, indicating improvement of lumbar lordosis in patients who underwent total disc replacement, an observation which was also mentioned by Lemaire et al. [28]. During the surgery, we did not place the prostheses in lordosis. One possible explanation of the improvement of lumbar lordosis may be partly because of the restoration of lumbar sequence. After the replacement of the degenerated disc, the intervertebral disc height is restored and so is the original biomechanics. The index segment may serve as a regulator and help lumbar to reach new sagittal equilibrium. This might be one important benefit of total disc replacement, in that it helps to improve lumbar sagittal balance.
The optimal prosthesis position is crucial to the long-term function of the intervertebral joint complex. In the coronal plane, the vertebral bodies are not symmetrically shaped and may be slightly rotated, which may even increase after discectomy and the release of the anterior or posterior ligament. Therefore, defining the midline in the coronal plane during surgery is highly complex. During surgery, we used the pedicles of the vertebral body as benchmarks instead of the spinous process of the vertebral ground plate. In the coronal plane, 25 prostheses were rated as ideally placed, 5 were discretely shifted, 4 were slightly shifted and 1 was markedly shifted. In the sagittal plane, 12 prostheses were rated as ideally placed, 14 were discretely shifted and 9 were suboptimally placed. Using the same criteria, our results are better than those reported from the study by Yue et al. [42]. The patient with markedly shifted prosthesis in the coronal plane and the 9 patients with suboptimally placed prostheses in the sagittal plane showed no inferior clinical results compared with the other patients. In Boss et al. [26] study, he also found that prosthesis positioning did not influence clinical outcome to any extent. The narrow system of classifying the three groups based on disc positioning (i.e., ideally placed, discretely shifted, and suboptimally placed) may be the main contributor to the poor position of the prostheses. Another possible explanation may be that the effect of suboptimally placed prostheses was minimized when the implanted segment gradually lost its range of motion with time. Although our stringent criteria to classify positioning of the protheses let to a large proportion of poorly positioned discs, the use of strict criteria is necessary to ensure the best possible placement of the prosthesis.
At the final follow-up, lumbar scoliosis over 3° was observed in 12 patients (37.5 %), with a mean angle of 5.6° (range 3°–12°). Of the 12 patients, 7 had left convex curvature. Theoretically, the shift of the prosthesis in the coronal plane will cause uneven loading and may contribute to lumbar scoliosis in the long run. Consequently, we tried to correlate the degree of scoliosis with the prosthesis position in the coronal plane but found only a low correlation, which may be partly because of the small number of patients.
Reoperations at the index level are required in 0–28.6 % of cases [31, 43–45]. Siepe et al. [45] categorized the reoperation rates into those resulting from general surgery-related complications, those for implant- or device-related complications, and those that were required for the treatment of adjacent-level pathologies. In this study, they found that the majority of revision surgeries were performed for persistent back pain, and that fewer were performed for implant failures. In our study, reoperation was performed in 2 patients, one because of adjacent segment degeneration and the other for pedicle fracture.
At the final follow-up, prosthesis subsidence was noted in 3 (9.4 %) patients (the subsidence distances were 3.1, 4.2 and 2.8 mm, respectively), but no symptoms appeared. Injury of the endplate during surgery and osteoporosis are the two main causes of prosthesis subsidence. However we cannot reach any conclusion on the effect of subsidence on segmental ROM based on these limited data.
Heterotopic ossification is not uncommon during the follow-up after lumbar TDR. The incidence of heterotopic ossification in short-term follow-up has been described as 1.4–83 %, mostly between 10 and 15 % [13, 18, 46]. Since heterotopic ossification can progress during follow-up, the occurrence rate in these studies might be an underestimate of the real incidence of late onset heterotopic ossification. And we believe that the reason for this is the meticulous search for any small ossification according to McAfee’s classification. The etiology of heterotopic ossification is still unknown. Some factors such as perioperative bleeding in the vicinity of implant, rough tissue dissection, underlying diffuse idiopathic skeletal hyperostosis and annular repair after implantation of the prosthesis may be associated with heterotopic ossification. However, further research is still needed to identify the potential risk factors and long-term clinical impact of heterotopic ossification.
Conclusions
This study presents the results of 32 patients implanted with the Charite III artificial disc with a mean follow-up of 11.8 years. Clinical success was achieved in 87.5 % of patients in this series. A return to work rate of 75.9 % was achieved. This study demonstrates that satisfactory clinical and radiological results can be achieved, and patient safety was proven with acceptable complication and reoperation rates. These results indicate that TDR with the Charite III artificial disc is a viable alternative to arthrodesis for the treatment of lumbar DDD at one or two levels. However, whether TDR can protect the adjacent segment over the long term still requires more evidence.
References
Frelinghuysen P, Huang RC, Girardi FP et al (2005) Lumbar total disc replacement part I: rationale, biomechanics, and implant types. Orthop Clin North Am 36:293–299
Geisler FH (2005) Surgical technique of lumbar artificial disc replacement with the Charité artificial disc. Neurosurgery 56:46–57
Zhou J, Wang X, Hou T et al (2002) One versus two BAK fusion cages in posterior lumbar interbody fusion to L4–L5 degenerative spondylolisthesis: a randomized, controlled prospective study in 25 patients with minimum two-year follow-up. Spine 27:2753–2757
Radcliff KE, Kepler CK, Jakoi A et al (2013) Adjacent segment disease in the lumbar spine following different treatment interventions. Spine J 13:1339–1349
Helgeson MD, Bevevino AJ, Hilibrand AS (2013) Update on the evidence for adjacent segment degeneration and disease. Spine J 13:342–351
Ghiselli G, Wang JC, Bhatia NN et al (2004) Adjacent segment degeneration in the lumbar spine. J Bone Joint Surg Am 86:1497–1503
Bertagnoli R, Yue JJ, Shah RV et al (2005) The treatment of disabling multilevel lumbar discogenic low back pain with total disc arthroplasty utilizing the ProDisc prosthesis: a prospective study with 2-year minimum follow-up. Spine (Phila Pa 1976) 30:2192–2199
Blumenthal S, McAfee PC, Guyer RD et al (2005) A prospective, randomized, multicenter Food and Drug Administration investigational device exemptions study of lumbar total disc replacement with the CHARITE artificial disc versus lumbar fusion: part I: evaluation of clinical outcomes. Spine 30:1565–1575
Guyer RD, McAfee PC, Banco RJ et al (2009) Prospective, randomized, multicenter Food and Drug Administration investigational device exemption study of lumbar total disc replacement with the CHARITE artificial disc versus lumbar fusion: five-year follow-up. Spine J 9:374–386
Sasso RC, Foulk DM, Hahn M (2008) Prospective, randomized trial of metal-on-metal artificial lumbar disc replacement: initial results for treatment of discogenic pain. Spine 33:123–131
Berg S, Tullberg T, Branth B et al (2009) Total disc replacement compared to lumbar fusion: a randomised controlled trial with 2-year follow-up. Eur Spine J 18:1512–1519
Gornet MF, Burkus JK, Dryer RF et al (2011) Lumbar disc arthroplasty with Maverick disc versus stand-alone interbody fusion: a prospective, randomized, controlled, multicenter investigational device exemption trial. Spine (Phila Pa 1976) 36:E1600–E1611
Regan JJ (2005) Clinical results of Charité lumbar total disc replacement. Orthop Clin North Am 36:323–340
Ross R, Mirza AH, Norris HE et al (2007) Survival and clinical outcome of SB Charite III disc replacement for back pain. J Bone Joint Surg Br 89:785–789
Gioia G, Mandelli D, Randelli F (2007) The Charite III artificial disc lumbar disc prosthesis: assessment of medium-term results. J Orthop Traumatol 8:134–139
Van de Kelft E, Verguts L (2012) Clinical outcome of monosegmental total disc replacement for lumbar disc disease with ball-and-socket prosthesis (Maverick): prospective study with four-year follow-up. World Neurosurg 78:355–363
Johnsen LG, Brinckmann P, Hellum C et al (2013) Segmental mobility, disc height and patient-reported outcomes after surgery for degenerative disc disease: a prospective randomised trial comparing disc replacement and multidisciplinary rehabilitation. Bone Joint J 95-B:81–89
vanOoij A, Oner FC, Verbout AJ (2003) Complications of artificial disc replacement: a report of 27 patients with the SB Charité disc. J Spinal Discord Tech 16:369–383
Shim CS, Lee SH, Shin HD et al (2007) CHARITE versus ProDisc: a comparative study of a minimum 3-year follow-up. Spine (Phila Pa 1976) 32:1012–1018
Hellum C, Berg L, Gjertsen Ø et al (2012) Adjacent level degeneration and facet arthropathy after disc prosthesis surgery or rehabilitation in patients with chronic low back pain and degenerative disc: second report of a randomized study. Spine (Phila Pa 1976) 37:2063–2073
Huang RC, Tropiano P, Marnay T et al (2006) Range of motion and adjacent level degeneration after lumbar total disc replacement. Spine J 6:242–247
Chung SK, Kim YE, Wang KC (2009) Biomechanical effect of constraint in lumbar total disc replacement: a study with finite element analysis. Spine (Phila Pa 1976) 34:1281–1286
Park CK, Ryu KS, Jee WH (2008) Degenerative changes of discs and facet joints in lumbar total disc replacement using ProDisc II: minimum two-year follow-up. Spine (Phila Pa 1976) 33:1755–1761
Harrop JS, Youssef JA, Maltenfort M et al (2008) Lumbar adjacent segment degeneration and disease after arthrodesis and total disc arthroplasty. Spine (Phila Pa 1976) 33:1701–1707
Rousseau MA, Bradford DS, Bertagnoli R et al (2006) Disc arthroplasty design influences intervertebral kinematics and facet forces. Spine J 6:258–266
Boss OL, Tomasi SO, Baurle B et al (2013) Lumbar total disc replacement: correlation of clinical outcome and radiological parameters. Acta Neurochir (Wien) 155:1923–1930
McAfee PC, Cunningham BW, Devine J et al (2003) Classification of heterotopic ossification (HO) in artificial disk replacement. J Spinal Disord Tech 16:384
Lemaire JP, Carrier H, Sariali el-H et al (2005) Clinical and radiological outcomes with the Charité artificial disc: a 10-year minimum follow-up. J Spinal Disord Tech 18:353–359
David T (2007) Long-term results of one-level lumbar arthroplasty: minimum 10-year follow-up of the CHARITE artificial disc in 106 patients. Spine 32:661–666
Tropiano P, Huang RC, Girardi FP et al (2005) Lumbar total disc replacement. Seven to eleven-year follow-up. J Bone Joint Surg Am 87-A:490–496
Park CK, Ryu KS, Lee KY et al (2012) Clinical outcome of lumbar total disc replacement using ProDisc-L in degenerative disc disease: minimum 5-year follow-up results at a single institute. Spine 37:672–677
Putzier M, Funk JF, Schneider SV et al (2006) Charité total disc replacement–clinical and radiographical results after an average follow-up of 17 years. Eur Spine J 15:183–195
Meir AR, Freeman BJ, Fraser RD et al (2013) Ten-year survival and clinical outcome of the AcroFlex lumbar disc replacement for the treatment of symptomatic disc degeneration. Spine J 13:13–21
Huang RC, Girardi FP, Cammisa FP Jr et al (2003) Long-term flexion-extension range of motion of the prodisc total disc replacement. J Spinal Disord Tech 16:435–440
Ishihara H, Osada R, Kanamori M et al (2001) Minimum 10-year follow-up study of anterior lumbar interbody fusion for isthmic spondylolisthesis. J Spinal Disord 14:91–99
Godde S, Fritsch E, Dienst M et al (2003) Influence of cage geometry on sagittal alignment in instrumented posterior lumbar interbody fusion. Spine 28:1693–1699
Goldstein JA, Macenski MJ, Griffith SL et al (2001) Lumbar sagittal alignment after fusion with a threaded interbody cage. Spine 26:1137–1142
Lazennec JY, Ramare S, Arafati N et al (2000) Sagittal alignment in lumbosacral fusion: relations between radiological parameters and pain. Eur Spine J 9:47–55
Kong Chao Lu, Shibao Hai Yong et al (2014) Radiological changes after Activ L total disc replacement and its correlation with clinical outcome. Chinese Journal of Spine and Spinal Cord 24:193–198
Long W, Ge C, Hong-qi Z et al (2013) Lumbar lordosis after disc replacement. ZhongguoZuzhiGongchengYanjiu 17: 5446–5451
Tournier C, Aunoble S, Le Huec JC et al (2007) Total disc arthroplasty: consequences for sagittal balance and lumbar spine movement. Eur Spine J 16:411–421
Yue JJ, Mo FF (2010) Clinical study to evaluate the safety and effectiveness of the Aesculap Activ-L artificial disc in the treatment of degenerative disc disease. BMC Surg 10:14
van den Eerenbeemt KD, Ostelo RW, van Royen BJ et al (2010) Total disc replacement surgery for symptomatic degenerative lumbar disc disease: a systematic review of the literature. Eur Spine J 19:1262–1280
Wei J, Song Y, Sun L et al (2013) Comparison of artificial total disc replacement versus fusion for lumbar degenerative disc disease: a meta-analysis of randomized controlled trials. Int Orthop 37:1315–1325
Siepe CJ, Heider F, Wiechert K et al (2014) Mid- to long-term results of total lumbar disc replacement: a prospective analysis with 5- to 10-year follow-up. Spine J 14:1417–1431
Cinotti G, David T, Postacchini F (1996) Results of disc prosthesis after a minimum follow-up period of 2 years. Spine 21:995–1000
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Lu, Sb., Hai, Y., Kong, C. et al. An 11-year minimum follow-up of the Charite III lumbar disc replacement for the treatment of symptomatic degenerative disc disease. Eur Spine J 24, 2056–2064 (2015). https://doi.org/10.1007/s00586-015-3939-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-015-3939-5