Abstract
Purpose
Recent work showed an increased risk of cervical and lumbar intervertebral disc (IVD) herniations in astronauts. The European Space Agency asked the authors to advise on the underlying pathophysiology of this increased risk, to identify predisposing factors and possible interventions and to suggest research priorities.
Methods
The authors performed a narrative literature review of the possible mechanisms, and conducted a survey within the team to prioritize research and prevention approaches.
Results and conclusions
Based on literature review the most likely cause for lumbar IVD herniations was concluded to be swelling of the IVD in the unloaded condition during spaceflight. For the cervical IVDs, the knowledge base is too limited to postulate a likely mechanism or recommend approaches for prevention. Basic research on the impact of (un)loading on the cervical IVD and translational research is needed. The highest priority prevention approach for the lumbar spine was post-flight care avoiding activities involving spinal flexion, followed by passive spinal loading in spaceflight and exercises to reduce IVD hyper-hydration post-flight.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The problem: disc herniations in astronauts
A recent publication [1] from the National Aeronautics and Space Administration (NASA) showed an increased risk of intervertebral disc (IVD) herniations in US astronauts (Fig. 1). 10.0 % (32 of a total of 321) of all US astronauts were diagnosed with IVD herniation after spaceflight compared to 3.5 % (34 of a total of 983) in the control, Earth-bound, population. The incidence was highest immediately upon returning to Earth, with 7 herniations occurring within 1 week of return to Earth (19 % of all post-flight herniations) and 14 herniations in total occurring within the first year. IVD herniations occurred in both the cervical and lumbar regions, with the incidence markedly increased at the cervical spine (21.4 times higher incidence rate than in the control population versus 2.8 times higher at the lumbar spine).
A herniated nucleus pulposus of an astronaut at surgery. This intraoperative photo shows the L2/3 intervertebral disc of an astronaut whose symptoms started shortly after spaceflight. The herniation is atypical in that the disruption of the annulus (a) occurred as a sharp horizontal tear just at the border between the disc and the vertebral body (see dark gap marked with line at b) and the nucleus pulposus herniated out through this space. The herniated nucleus pulposus has already been removed before this photo was taken. There is a retractor (c) retracting the ligamentum flavum at the top of the image. The gel-like material (d) is fat tissue
The European Space Agency (ESA) asked the authors to advise on why this higher rate of IVD herniations occurs in astronauts and what could be done about it. The main question is not why astronauts get herniations but rather: Why do they get more than the general population? How come this increase persists often months after the spaceflight is over? Also, why do so many herniations affect the cervical spine? An additional question of interest is: What does the astronaut experience tell us about IVD herniation in the normal population? The current paper reports on our literature review, discussions, conclusions and suggestions for future work.
What are the likely mechanisms?
Increase in lumbar disc hydration as a result of reduced spinal loads in space
In our opinion, the most probable mechanism, at least for the lumbar spine, is swelling of the IVD in the unloaded condition during spaceflight. The overhydrated disc is then vulnerable to an IVD herniation, especially when the pressures on the lumbar spine are increased under flexion during or immediately after spaceflight. This mechanism is supported by mechanical experiments on cadaveric and animal spines which have shown that severe loading in combined bending and compression can cause apparently normal lumbar IVDs to herniate [2, 3], and that herniation is more likely when the IVDs are fully hydrated [4].
Increased IVD swelling during spaceflight has not been shown directly. However, IVD swelling in spaceflight can be inferred from increases in spine length [5] and body height [6] during spaceflight and from IVD swelling observed in vivo following bed rest (Fig. 2) for a few hours [7, 8], overnight [9–12], and for a number of weeks [13–19]. At the end of overnight bed-rest, lumbar intradiscal pressure is increased [20]. Flattening of the lumbar lordosis could explain increases in spinal length despite swelling of the IVD, but flattening of the lumbar lordosis has not been consistently found in bed rest studies. At the end of prolonged bed rest, decreases [17, 21], no change [14, 22], and increases [16] of the lumbar lordosis have been seen.
Expansion of the lumbar L4/5 IVD due to spaceflight simulation (bed rest). MR images of 23 subjects of an earlier 60-d bed rest study [21], and have not been published previously. In this dataset, whole IVD volume increased by mean (SD) 6.0 (3.3) %, IVD height increased by 7.0 (3.6) % and the anteroposterior IVD dimension decreased by 1.6 (1.9) %. This can be seen in the increase in height of the IVD anteriorly and posteriorly and by the “drawing in” of the anterior aspect of the IVD. The bottom image overlaps the two images and exemplifies this. The geometry was averaged between all 23 subjects
This physical mechanism (Fig. 3) could explain why astronauts are at increased risk of herniation either during spaceflight, and/or after return to Earth. The time course of recovery of IVD size (Fig. 4) and hydration after actual or simulated spaceflight is not clear. The only published work to date after spaceflight showed no significant differences in sagittal plane disc area and lumbar spine length of four astronauts measured 24 h after 8 days spaceflight [19]. Diurnal studies [23] suggest that reductions of IVD size upon rising are normally a rapid process (on the order of hours). However, data from spaceflight simulation (bed rest) have shown IVD volume and/or height to be still increased at follow-up compared to before bed rest 7 days [18], 5 months [14] and even up to 2 years [24] after prolonged bed rest. In line with this, in vitro data [25] suggest that an unloaded disc becomes hyper-swollen disc and may lose fluid relatively slowly when loading is resumed.
A mechanical mechanism due to increased IVD size and hydration? On Earth, loading of the spine in daily life limits the water content of the IVD nucleus (“normohydration”), and hence the pressure within it. The IVD is, however, most vulnerable to herniation in flexion and compression. During and after spaceflight (“hyperhydration”), the lumbar disc is swollen, annular structures are stretched, and nucleus pressure increased. Then, in flexion the strain in, and thinning of, the posterior annulus, is even greater and the nucleus can more easily herniate through the posterior annulus. This, we presume, is the most likely mechanism to explain increased risk of IVD herniation in the first days after spaceflight
It may take some time for an IVD to return to pre-spaceflight dimensions. Height of a human IVD with daily loading cycles after the IVD has been allowed to expand (stored unloaded in a saline bath for several minutes). The IVD was loaded with a dynamic axial load for 16 h at 374 (64) kPa and unloaded for 8 h at 8 a.m. at 74 (7) kPa, for 10 days. These example data show how it will probably take some time for the IVD to return to pre-spaceflight dimensions. Data from bed rest studies [14, 18, 24] indicate it can take a number of weeks, or even months, for this to occur. Image kindly provided by the authors of Ref. [74]
Mechanisms for the cervical spine unclear: basic research is needed
For the cervical spine little data on possible herniation mechanisms are available. There are no published data from astronauts on changes in the cervical IVDs during spaceflight and while spinal length does increase during spaceflight [5, 6], it is not clear which spinal region contributes to this lengthening. Data from studies on Earth show that some degree of cervical spine lengthening occurs with recumbency [26, 27]. There are a number of differences between cervical and lumbar discs: in composition [28], anatomy [29] and biomechanical characteristics [30]. However, these data do not help us understand why the risk of IVD herniation is much more elevated at the cervical spine than at the lumbar spine in astronauts. Overall, a mechanism of increased cervical IVD injury risk due to increased IVD size and hydration may be possible, but there are no published data available to substantiate this hypothesis. Basic research on the cervical IVDs is needed.
Muscle changes
Muscle weakness and/or dysfunction may lead to risk of injury and may also predispose to herniation. It has been established that muscle atrophy occurs around the lumbar spine during spaceflight [31]. Data from bed rest on Earth suggest that the time course of lumbar muscle size recovery [14, 16, 32, 33] and the recovery of lumbar extension torque [16] is within “weeks” after bed rest. However, muscle function, as measured by superficial electromyography recordings [34, 35], may take “months” to recover. Overall, lumbar muscle weakness and dysfunction may well contribute to initial risk and may present an increased injury risk for weeks to months. Furthermore, on the basis of data on the (disturbed) control of posture after spaceflight [36, 37], it is reasonable to assume that altered sensorimotor control after spaceflight may lead to decreased protection of the IVD by the muscular system.
At the cervical spine, the (neuro)muscular adaptation is much less clear, again due to a paucity of data. What we do know is that no atrophy of the cervical extensor muscles was seen in nine astronauts measured after spaceflight [31]. Data from studies of neck pain suggest that the deep cervical extensors [38] and deep cervical flexors [39] may be important for protecting the spine, yet muscle groups such as these have not been examined in astronauts in any detail. In prolonged bed rest, hypertrophy of most of the cervical muscles was seen [40], but as argued in that publication, prolonged bed rest might not be a good model for the effects of spaceflight on the cervical spine region. Further data are required on the response of the cervical spine musculature to weightlessness.
Ergonomic issues
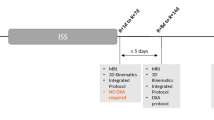
There are some ergonomic issues specific to astronauts that likely impact IVD injury risk (Figs. 5, 6). Apollo-era and Shuttle astronauts had [1] a higher incidence of IVD injury than International Space Station (ISS) and Mir astronauts. Shuttle astronauts landed in a sitting-type position similar to landing in a commercial aeroplane. Apollo astronauts landed in a more horizontal position: the reader should imagine sitting in a very large baby car seat whilst being positioned in a reclining position. Mir and ISS astronauts coming down with the Soyuz vehicle also land in a more horizontal position in a moulded seat liner that is designed specifically for each crew member. In the US astronaut program, no spinal injuries are definitively known to have occurred during re-entry, however. Post-flight, ISS astronauts do not stand up and walk around or perform risky (flexion) activities like earlier Shuttle and Apollo astronauts did. ISS astronauts also spend much longer time in lying or reclining post-flight than Shuttle astronauts (Figs. 5, 6). So, ergonomic factors likely play a very important role in lumbar IVD herniation risk. Specifically, loading the spine—as in walking, and performing risky movements such as spinal flexion activities when the IVDs are most vulnerable—will most probably increase the risk for IVD herniations.
The ergonomics of being a space traveller. Apollo and Shuttle astronauts (top panels) walked immediately after landing, whereas astronauts and cosmonauts coming back with the Soyuz vessel (bottom panels) were carried out of the capsule. The data presented by Johnston et al. [1] suggested that those astronauts who performed upright activities immediately after spaceflight were at a higher risk of IVD herniation. Photo credits: NASA (top left and right, bottom right), ESA (bottom left)
Extraction of an astronaut from the Soyuz vehicle on the 1st of July 2012. An astronaut is being taken out of the Soyuz capsule. Note that he is in a fully flexed position. This is not ideal for the spine, but given the physical setup of the vehicle, this position may be unavoidable. Most important is that the helpers bear most of the load and that the astronaut himself does not generate high internal muscle forces. The helper should probably watch his back as well too, however. Photo credit: ESA
Cumulative injury to the IVD prior to spaceflight
The increased IVD herniation incidence in astronauts could result from cumulative IVD injury occurring prior to the spaceflights. Johnston et al. [1] observed an increased risk of IVD herniation upon entering the astronaut program. This could potentially be due to pre-existing conditions or the training program itself.
Since a number of astronauts had previously been fighter pilots, Johnston et al. [1] evaluated whether this prior history may be a predictive factor for IVD herniation, but they did not find any significant relationship. Performing high-G manoeuvres [41], pilot ejection [42], and helicopter vibration [43] present risk factors for injury to the neck. Potentially, the resulting high-compressive forces on the spine could result in end plate fractures, then leading to adjacent IVD degeneration [44]. Alternatively, the IVD injuries could have occurred whilst astronauts were still fighter pilots and that the injuries were only picked up as part of medical management once they entered the astronaut program. However, the longitudinal data available [45] and meta-analysis of cross-sectional data [46] suggest that the majority of degenerative changes in the IVDs of fighter pilots are due to normal ageing and not the fighter pilot occupation per se.
Furthermore, the astronaut selection process might “select” people at higher risk of IVD problems. For example, in the Finnish Air Force, one of the selection criteria for fighter pilots is peak power on a cycling ergometry test. Experience (unpublished observations; R. Sovelius) has been that applicants who are weightlifters or ice-hockey players typically perform better on these tests. Weightlifters typically show more degenerative IVD changes [47]. It could be that persons selected for astronaut training, due to their previous sporting pursuits, are at risk for IVD problems.
Long-term effect of spaceflight on spinal tissues
Spaceflights may have an effect on IVD physiology and, in the long run, possibly also have an impact on the rate of IVD degeneration. This may thereby have an influence on medium to long-term IVD injury risk. Animal models investigating the IVD in spaceflight [48, 49], hindlimb suspension [50–53] and tail vertebra immobilisation [54, 55] have typically [48–53, 55], but not always [54], found losses in glycosaminoglycan content. However, the extent to which animal models can be used as a model of human IVD changes in spaceflight remains an open question [56]. It is also unclear to what extent such changes in IVD composition occur in humans in unloading, and further if and how any such changes affect the herniation risk. In vitro work has shown that both glycosaminoglycan synthesis rates fall [57–59] and production of proteases is able to degrade the IVD increases [60] with the decrease in osmolarity arising from IVD swelling. Such changes in cellular activity could be responsible for the loss of glycosaminoglycans seen in fast-metabolising small animal IVDs. However, in the much larger and relatively acellular human IVDs, the half-life of the proteoglycan aggrecan, a major component of the IVD matrix and responsible for regulating IVD swelling pressure [61], is around 12 years for normal IVDs and 8 years for degenerate IVDs [62]. Hence, spaceflight appears too short for such changes in cellular activity to influence human IVD composition noticeably, at least during the period when susceptibility to herniation is increased.
Vertebral body bone loss in spaceflights may influence the IVD. A loss of lumbar spine bone mineral density has been demonstrated in astronauts after a spaceflight [63]. Clinically, it is observed that people with lower bone density have larger IVDs [64] (Fig. 7). Presumably the lower bone strength permits the deformation of the vertebral end plate and expansion of the IVDs. Also, anchoring of annulus fibrosus fibres into the vertebrae may be less strong when bone mass is lost: a significant fraction of herniations occurs as a result of failure at the end plate junction [65]. These factors may play a role in the injury risk of the IVD.
What role do bone loss and end plate changes play in the IVD in spaceflight? People with osteoporosis have larger IVDs [64]. With losses in bone, the structure of the vertebra changes and the IVD “expands” into this space. This may play a role in increases in IVD size during spaceflight. There is some evidence to support this hypothesis: blinded radiological examination of the thoracic IVDs during bed rest [40] showed “bowing” of the vertebral end plate in two subjects at the end of bed rest. The image stems from material from an earlier project [75], but has not been published elsewhere
Essentially nothing is known about the adaptation of the spinal ligamentous system to weightlessness. IVD swelling increases tension in the intervertebral ligaments, so that they may provide greater resistance to spinal flexion [4, 66]. Increased ligament tension would increase IVD compression, especially when the spine is flexed, and this could increase the risk of IVD herniations due to spaceflight. The role of the spinal ligaments in bending moments [67] or in proprioceptive function may be disturbed after spaceflight, but data to support this are lacking.
How can we best improve our understanding of the problem?
The European Space Agency requested advice on research priorities to better understand the issue of IVD herniations in astronauts. In our discussions, some specific research questions were suggested and in other instances, general research approaches aimed at increasing our knowledge, albeit without a specific hypothesis, were put forward. Each of these had their scientific merit and each of them had differing levels of difficulty of implementation, financial cost and time cost. Research ideas were ranked by the authors and those gained wider support within the team were:
Using existing astronaut data Other data from astronaut medical records (e.g. body height) may give insight into spinal adaptation after spaceflight. Also, gaining more detailed information on the existing IVD herniation events (e.g. which vertebral level, location of the herniation, time of day of herniation, etc.) would be insightful. Although these data will likely be exhausted quickly, we consider it prudent to examine such data before planning new experiments.
Gaining basic information on the cervical spine A common theme in our discussions and review of the literature was that we lack even basic information on the impact of spaceflight on the cervical spine. For basic clinical, biomechanical and physiological sciences, research has been done mostly on the lumbar spine, and much less on the cervical spine. Topics could include examining diurnal variation in the cervical discs, understanding changes in neck musculature in spaceflight, and understanding loading patterns that cause cervical disc herniation.
Better understanding lumbar IVD diurnal changes Diurnal variation in the lumbar IVDs may be altered after spaceflight and more data on this will improve our understanding of lumbar IVD herniation risk after spaceflight. Also, using diurnal variation of lumbar IVDs is probably a useful model to understand the likely effects of spaceflight on the lumbar spine.
Minor modification of medical data collection in astronauts This could help to answer questions without doing complex studies. For example, consistent data collection on body height with standard equipment and information on body shape would be of use. Of course, any additional time taken from an astronaut’s schedule will be seen critically by mission organisers, so this may be a limiting factor.
Assessing astronauts that have already flown Depending on research question, this could be a comparatively low-cost method to test certain hypotheses without the efforts or cost of a spaceflight or an Earth-bound bed rest study. There are some practical impediments to this strategy, such as gaining access to the ex-astronauts, but it is nonetheless possible.
Larger modification of medical data collection in astronauts For example, is it possible to consider a whole-body magnetic resonance imaging scan pre- and post-flight? With, for example, three-dimensional sequences any plane of the body can be reconstructed to enable researchers to examine the body plane of interest. No matter how useful the data may be, the administrative and scheduling challenges of implementing new standard medical examinations will present a limiting factor.
There were other ideas that gained some support within the team. These represent lower priority options that could be targeted once the primary approaches are exhausted.
Understanding which loading protocols help to prevent IVD changes with unloading The available bed rest data on the impact of exercise in bed rest on the lumbar discs has been summarised in earlier work [16], but there are a number of open questions for the lumbar spine with a complete lack of information for the cervical spine.
Laboratory in vitro mechanical testing and/or computer modelling This approach also has the advantage of being relatively low cost and one that could deliver useful data fairly quickly (Fig. 8).
Finite element modelling for understanding the impact of spaceflight on the IVDs. The image stems from an earlier published work [76], but has not been published elsewhere. In this particular study, the finite element modelling was used to investigate which regions of the IVD are at highest risk of injury under various loading states and for different states of IVD degeneration. Such approaches can be used to understand changes in IVD strains and hence injury risk post-flight
Understanding changes at the vertebral end plate It is unclear how spaceflight and spine unloading might distort or affect the function of the end plate (see also Fig. 7).
Understanding changes in (eccentric) muscle function and sensorimotor control A better understanding of changes in muscle function will help us to guide rehabilitation processes better. Proprioception and motor processing are impacted in microgravity [36]. Similar to muscle function, the controller of motor output could play a role in preventing injury to the spine and a better understanding of this system is warranted.
Understanding why the lumbar IVDs remain larger for some time after spaceflight simulation (bed rest) Earlier findings have indicated that the lumbar IVDs remain larger than baseline levels some weeks or months after bed rest. It may be relevant to injury risk to understand why this effect occurs.
Understanding what impact changing exercise devices on the ISS has had A new “advanced resistive exercise device” has been put in use on the International Space Station. This research question asks what impact, if any, the new higher load device had on back pain and IVD injury risk post-flight.
What can we do to reduce the incidence of IVD herniations in astronauts?
One of the key issues is because our knowledge of the cervical spine is so limited, it is impossible to provide any recommendations for countermeasures against cervical IVD herniation without some basic spaceflight-related and clinical research.
Despite this, one primary suggestion to reduce incidence of IVD herniation, drawn predominately from our knowledge base of the lumbar spine involves management of the astronaut before and after landing. These recommendations include:
-
In general: avoid spinal flexion and compression activities. This includes care with daily tasks, such as when putting ones socks on, and also use of devices to assist in activities to avoid such positions.
-
Spend more time in lying in the days after flight.
-
Consider some kind of orthosis or taping for the lumbar spine, and for the cervical spine a neck collar.
-
After flight: walking around gently, but otherwise further research is required on what kinds of exercise protocols are better for loading the disc when it is hyper-hydrated.
-
The duration for which such “care with spinal flexion activities” protocols should be implemented should last for a few weeks. However, further work is needed to better define this time frame.
As increase in disc hydration in space appears to be the major risk factor in the lumbar spine at least, mechanisms which attempt to counteract such changes could potentially lower herniation risk. Passive spinal loading throughout spaceflight using, for example, the “skinsuit” [68] could reduce IVD changes in-flight through continual loading of the spine. However, such compression suits can be, according to anecdotal reports, uncomfortable and compliance will need to be considered. Exercise approaches may also reduce (lumbar) IVD hyper-hydration and size. Currently the “fetal tuck” position is used by astronauts to relieve low back pain in spaceflight [69], caution should be applied when considering this as a standard countermeasure: this kind of spinal loading differs from the normal functional loading patterns of the spine. We suggest that axial loading of the spine in a neutral posture would be better. Furthermore, it would be beneficial to optimise the training programs on exercise devices on the ISS to avoid inappropriate loading and to consider post-flight exercise with partial unloading.
What can astronaut IVD herniations tell us about herniations in the normal population?
Astronauts are not a very large population, whereas IVD herniations in Earth-bound populations are an important and common diagnosis causing substantial disability. The astronaut data highlight risk factors for IVD herniation in the general population, specifically that risk of herniation likely varies with time of day, with risks possibly highest in the morning after waking from an overnight rest. Whilst there have been no direct studies on diurnal variation in incidence of herniation, there are data on back pain. One study on acute-onset low back pain [70] noted that, of those individuals who could recall when pain onset started, 46 % of these reported pain beginning between 8 a.m. and midday. Another study in hospital employees [71] reported that the mean time of day for low back injury was in the morning. Similarly a study [72] reported the highest rates of back injury in the “day shift” (7 a.m.–4 p.m.), but did not differentiate the time of the day any further. Finally, a crossover design intervention study [73] in people with recurrent low back pain showed that instruction to control spinal flexion activities in the early morning reduced number of days with pain, pain intensity and medication use.
Overall, the data from astronauts and bed rest support the idea that diurnal changes in the spine do influence injury risk in Earth-bound populations and suggest modalities for reducing these risks. The ideas are equally applicable to individuals subject to bed rest for medical reasons (e.g. post-surgery, or prolonged care in an intensive care unit). Furthermore, the findings also help to highlight risk window periods for activities such as heavy lifting and spine flexion.
The issue of IVD herniations in astronauts also highlights the role of mechanical loading in IVD herniation in general. Although we cannot rule out an impact of spaceflight on medium to long-term IVD degeneration post-flight in the relatively short duration of a spaceflight the IVD is not likely to degenerate. Thus, the spike of IVD herniation incidence observed by Johnston et al. [1] immediately after spaceflight emphasises the mechanics of internal IVD forces and influence of external forces on IVD herniation.
Concluding remarks
In conclusion, the current review examined available data trying to define reasons why astronauts are at a higher risk for IVD herniations. For the lumbar IVD, the most likely contributing factor is swelling of the IVDs under weightlessness. However, for the cervical IVDs, the database is so limited that we cannot postulate a likely mechanism or recommend approaches for prevention. Basic research on the cervical spine, such as the impact of spaceflight and simulation (e.g. diurnal variation) as well as basic clinical, biomechanical and physiological research on the cervical IVDs is required. To better understand the problem of IVD herniations in astronauts we further advise focussing first on using existing astronaut data and assessing astronauts that have already flown in case–control studies. Further suggestions include: modification of medical data collection within the realms possible within space agencies and examining diurnal lumbar IVD variation both in astronauts after spaceflight as well as validated ground-based models. In the Earth-bound population, the current work highlights the potential impact of diurnal variation on IVD injury risk and also the role of mechanical loading of the IVD for the risk of experiencing herniations in general.
References
Johnston SL, Campbell MR, Scheuring R, Feiveson AH (2010) Risk of herniated nucleus pulposus among U.S. astronauts. Aviat Space Environ Med 81:566–574
Adams MA, Hutton WC (1982) Prolapsed intervertebral disc. A hyperflexion injury 1981 Volvo Award in basic science. Spine 7:184–191
Wade KR, Robertson PA, Thambyah A, Broom ND (2014) How healthy discs herniate: a biomechanical and microstructural study investigating the combined effects of compression rate and flexion. Spine 39:1018–1028. doi:10.1097/BRS.0000000000000262
Adams MA, Dolan P, Hutton WC (1987) Diurnal variations in the stresses on the lumbar spine. Spine 12:130–137
Young KS, Rajulu S (2011) The effects of microgravity on seated height (Spinal Elongation). NASA Report Number JSC–CN–25133
Thornton WE, Hoffer GW, Rummel JA (1977) Anthropometric changes and fluid shifts. In: Johnston RS, Dietlen LF (eds) Biomedical results from Skylab. NASA, Washington, DC, pp 330–338
Matsumura Y, Kasai Y, Obata H et al (2009) Changes in water content of intervertebral discs and paravertebral muscles before and after bed rest. J Orthop Sci 14:45–50. doi:10.1007/s00776-008-1288-5
Malko JA, Hutton WC, Fajman WA (1999) An in vivo magnetic resonance imaging study of changes in the volume (and fluid content) of the lumbar intervertebral discs during a simulated diurnal load cycle. Spine 24:1015–1022
Malko JA, Hutton WC, Fajiman WA (2002) An in vivo study of the changes in volume (and fluid content) of the lumber intervertebral disc after overnight bed rest and during an 8-hour walking protocol. J Spinal Disord Tech 15:157–163
Hutton WC, Malko JA, Fajman WA (2003) Lumbar disc volume measured by MRI: effects of bed rest, horizontal exercise, and vertical loading. Aviat Space Environ Med 74:73–78
Boos N, Wallin Å, Gbedegbegnon T et al (1993) Quantitative MR imaging of lumbar intervertebral disks and vertebral bodies: influence of diurnal water content variations. Radiology 188:351–354
Botsford DJ, Esses SI, Ogilvie-Harris DJ (1994) In vivo diurnal variation in intervertebral disc volume and morphology. Spine 19:935–940
Cao P, Kimura S, Macias BR et al (2005) Exercise within lower body negative pressure partially counteracts lumbar spine deconditioning associated with 28-day bed rest. J Appl Physiol 99:39–44
Belavý DL, Bansmann PM, Bohme G et al (2011) Changes in intervertebral disc morphology persist 5 months after 21-days bed-rest. J Appl Physiol 111:1304–1314
Belavý DL, Armbrecht G, Richardson CA et al (2011) Muscle atrophy and changes in spinal morphology: is the lumbar spine vulnerable after prolonged bed-rest? Spine 36:137–145
Belavý DL, Ohshima H, Bareille M-P et al (2011) Limited effect of fly-wheel and spinal mobilization exercise countermeasures on lumbar spine deconditioning during 90d bed-rest in the Toulouse LTBR study. Acta Astronaut 69:406–419
Belavý DL, Hides JA, Wilson SJ et al (2008) Resistive simulated weight bearing exercise with whole body vibration reduces lumbar spine deconditioning in bed-rest. Spine 33:E121–E131
Holguin N, Muir J, Rubin C, Judex S (2009) Short applications of very low-magnitude vibrations attenuate expansion of the intervertebral disc during extended bed rest. Spine J 9:470–477
Le Blanc AD, Evans HJ, Schneider VS et al (1994) Changes in intervertebral disc cross-sectional area with bed rest and space flight. Spine 19:812–817
Wilke HJ, Neef P, Caimi M et al (1999) New in vivo measurements of pressures in the intervertebral disc in daily life. Spine 24:755–762
Belavý DL, Armbrecht G, Gast U et al (2010) Countermeasures against lumbar spine deconditioning in prolonged bed-rest: resistive exercise with and without whole-body vibration. J Appl Physiol 109:1801–1811
Macias BR, Cao P, Watenpaugh DE, Hargens AR (2007) LBNP treadmill exercise maintains spine function and muscle strength in identical twins during 28-days simulated microgravity. J Appl Physiol 102:2274–2278
Tyrrell AR, Reilly T, Troup JD (1985) Circadian variation in stature and the effects of spinal loading. Spine 10:161–164
Belavý DL, Armbrecht G, Felsenberg D (2012) Incomplete recovery of lumbar intervertebral discs 2-years after 60-days bed-rest. Spine 37:1245–1251
Van der Veen AJ, Bisschop A, Mullender MG, Van Dieën JH (2013) Modelling creep behaviour of the human intervertebral disc. J Biomech 46:2101–2103. doi:10.1016/j.jbiomech.2013.05.026
Wing P, Tsang I, Gagnon F et al (1992) Diurnal changes in the profile shape and range of motion of the back. Spine 17:761–766
Reinach Y (1992) Die Wirbelsäule under dem Einfluß simulierter Mikrogravitation. Medical Doctoral Thesis. Insitut für Physiologie, Freie Universität Berlin
Scott JE, Bosworth TR, Cribb AM, Taylor JR (1994) The chemical morphology of age-related changes in human intervertebral disc glycosaminoglycans from cervical, thoracic and lumbar nucleus pulposus and annulus fibrosus. J Anat 184(Pt 1):73–82
Mercer S, Bogduk N (1999) The ligaments and annulus fibrosus of human adult cervical intervertebral discs. Spine 24:619–626
Skrzypiec DM, Pollintine P, Przybyla A et al (2007) The internal mechanical properties of cervical intervertebral discs as revealed by stress profilometry. Eur Spine J 16:1701–1709. doi:10.1007/s00586-007-0458-z
Le Blanc A, Lin C, Shackelford L et al (2000) Muscle volume, MRI relaxation times (T2), and body composition after spaceflight. J Appl Physiol 89:2158–2164
Hides JA, Belavý DL, Stanton W et al (2007) MRI assessment of trunk muscles during prolonged bed rest. Spine 32:1687–1692
Hides JA, Lambrecht G, Richardson CA et al (2011) The effects of rehabilitation on the muscles of the trunk following prolonged bed rest. Eur Spine J 20:808–818
Belavý DL, Richardson CA, Wilson SJ et al (2007) Tonic to phasic shift of lumbo-pelvic muscle activity during 8-weeks of bed-rest and 6-months follow-up. J Appl Physiol 103:48–54
Belavý DL, Richardson CA, Wilson SJ et al (2007) Superficial lumbo-pelvic muscle overactivity and decreased co-contraction after 8-weeks of bed-rest. Spine 32:E23–E29
Roll R, Gilhodes JC, Roll JP et al (1998) Proprioceptive information processing in weightlessness. Exp Brain Res 122:393–402
Layne CS, Mulavara AP, McDonald PV et al (2001) Effect of long-duration spaceflight on postural control during self-generated perturbations. J Appl Physiol 90:997–1006
Schomacher J, Falla D (2013) Function and structure of the deep cervical extensor muscles in patients with neck pain. Man Ther 18:360–366. doi:10.1016/j.math.2013.05.009
Falla DL, Jull GA, Hodges PW (2004) Patients with neck pain demonstrate reduced electromyographic activity of the deep cervical flexor muscles during performance of the craniocervical flexion test. Spine 29:2108–2114
Belavý DL, Miokovic T, Armbrecht G, Felsenberg D (2013) Hypertrophy in the cervical muscles and thoracic discs in bed-rest? J Appl Physiol 115:586–596. doi:10.1152/japplphysiol.00376.2013
Jones JA, Hart SF, Baskin DS et al (2000) Human and behavioral factors contributing to spine-based neurological cockpit injuries in pilots of high-performance aircraft: recommendations for management and prevention. Mil Med 165:6–12
Werner U (1999) Ejection associated injuries within the German Air Force from 1981–1997. Aviat Space Environ Med 70:1230–1234
Byeon JH, Kim JW, Jeong HJ et al (2013) Degenerative changes of spine in helicopter pilots. Ann Rehabil Med 37:706–712. doi:10.5535/arm.2013.37.5.706
Pippig T, Kriebel J (2000) Prevalence of cervical and lumbar disc disorders in pilots of the German armed forces. Eur J Med Res 5:5–8
Sovelius R, Salonen O, Lamminen A et al (2008) Spinal MRI in fighter pilots and controls: a 13-year longitudinal study. Aviat Space Environ Med 79:685–688
Shiri R, Frilander H, Sainio M et al (2014) Cervical and lumbar pain and radiological degeneration among fighter pilots: a systematic review and meta-analysis. Occup Environ Med. doi:10.1136/oemed-2014-102268
Videman T, Sarna S, Battié MC et al (1995) The long-term effects of physical loading and exercise lifestyles on back-related symptoms, disability, and spinal pathology among men. Spine 20:699–709
Pedrini-Mille A, Maynard JA, Durnova GN et al (1992) Effects of microgravity on the composition of the intervertebral disk. J Appl Physiol 73:26S–32S
Sinha RK, Shah SA, Hume EL, Tuan RS (2002) The effect of a 5-day space flight on the immature rat spine. Spine J 2:239–243
Yasuoka H, Asazuma T, Nakanishi K et al (2007) Effects of reloading after simulated microgravity on proteoglycan metabolism in the nucleus pulposus and anulus fibrosus of the lumbar intervertebral disc: an experimental study using a rat tail suspension model. Spine (Phila Pa 1976) 32:E734–E740
Holguin N, Judex S (2010) Rat intervertebral disc health during hindlimb unloading: brief ambulation with or without vibration. Aviat Space Environ Med 81:1078–1084
Holguin N, Uzer G, Chiang F-P et al (2011) Brief daily exposure to low-intensity vibration mitigates the degradation of the intervertebral disc in a frequency-specific manner. J Appl Physiol 111:1846–1853. doi:10.1152/japplphysiol.00846.2011
Hutton WC, Yoon ST, Elmer WA et al (2002) Effect of tail suspension (or simulated weightlessness) on the lumbar intervertebral disc: study of proteoglycans and collagen. Spine (Phila Pa 1976) 27:1286–1290
Iatridis JC, Mente PL, Stokes IA et al (1999) Compression-induced changes in intervertebral disc properties in a rat tail model. Spine 24:996–1002
MacLean JJ, Lee CR, Grad S et al (2003) Effects of immobilization and dynamic compression on intervertebral disc cell gene expression in vivo. Spine (Phila Pa 1976) 28:973–981
Alini M, Eisenstein SM, Ito K et al (2008) Are animal models useful for studying human disc disorders/degeneration? Eur Spine J 17:2–19. doi:10.1007/s00586-007-0414-y
Bayliss MT, Urban JP, Johnstone B, Holm S (1986) In vitro method for measuring synthesis rates in the intervertebral disc. J Orthop Res 4:10–17. doi:10.1002/jor.1100040102
Ohshima H, Urban JP, Bergel DH (1995) Effect of static load on matrix synthesis rates in the intervertebral disc measured in vitro by a new perfusion technique. J Orthop Res 13:22–29. doi:10.1002/jor.1100130106
Ohshima H, Tsuji H, Hirano N et al (1989) Water diffusion pathway, swelling pressure, and biomechanical properties of the intervertebral disc during compression load. Spine 14:1234–1244
Cui Y, Lee S, Yu J, Urban JP (2012) MMP expression by intervertebral disc cells is responsive to changes in extracellular osmolarity. FASEB J 26:906.4
Johnstone B, Bayliss MT (1995) The large proteoglycans of the human intervertebral disc. Changes in their biosynthesis and structure with age, topography, and pathology. Spine (Phila Pa 1976) 20:674–684
Sivan SS, Tsitron E, Wachtel E et al (2006) Aggrecan turnover in human intervertebral disc as determined by the racemization of aspartic acid. J Biol Chem 281:13009–13014
Le Blanc A, Schneider V, Shackelford L et al (2000) Bone mineral and lean tissue loss after long duration space flight. J Musculoskelet Neuronal Interact 1:157–160
Kwok AWL, Wang Y-XJ, Griffith JF et al (2012) Morphological changes of lumbar vertebral bodies and intervertebral discs associated with decrease in bone mineral density of the spine: a cross-sectional study in elderly subjects. Spine 37:E1415–E1421. doi:10.1097/BRS.0b013e31826f561e
Rajasekaran S, Bajaj N, Tubaki V et al (2013) ISSLS Prize winner: the anatomy of failure in lumbar disc herniation: an in vivo, multimodal, prospective study of 181 subjects. Spine 38:1491–1500. doi:10.1097/BRS.0b013e31829a6fa6
Adams MA, Dolan P (1996) Time-dependent changes in the lumbar spine’s resistance to bending. Clin Biomech (Bristol, Avon) 11:194–200
Panjabi MM, Goel VK, Takata K (1982) Physiologic strains in the lumbar spinal ligaments. An in vitro biomechanical study 1981 Volvo Award in Biomechanics. Spine 7:192–203
Waldie JM, Newman DJ (2011) A gravity loading countermeasure skinsuit. Acta Astronaut 68:722–730. doi:10.1016/j.actaastro.2010.07.022
Wing PC, Tsang IKY, Susak L et al (1991) Back Pain and Spinal Changes in Microgravity. Orthop Clin North Am 22:255–262
Varma KM, Porter RW (1995) Sudden onset of back pain. Eur Spine J 4:145–147
Ryden LA, Molgaard CA, Bobbitt S, Conway J (1989) Occupational low-back injury in a hospital employee population: an epidemiologic analysis of multiple risk factors of a high-risk occupational group. Spine 14:315–320
Bigos SJ, Spengler DM, Martin NA et al (1986) Back injuries in industry: a retrospective study. II. Injury factors. Spine 11:246–251
Snook SH, Webster BS, McGorry RW et al (1998) The reduction of chronic nonspecific low back pain through the control of early morning lumbar flexion. A randomized controlled trial. Spine 23:2601–2607
Emanuel KS, Vergroesen P-PA, Kingma I, Smit TH (2014) Biomechanics of the degenerating human intervertebral disc: a 10-day axial loading study. In: Proceedings of European Cells and Materials, vol XV. Davos, Switzerland
Wilke H-J, Rohlmann F, Neidlinger-Wilke C et al (2006) Validity and interobserver agreement of a new radiographic grading system for intervertebral disc degeneration: part I. Lumbar spine. Eur Spine J 15:720–730. doi:10.1007/s00586-005-1029-9
Schmidt H, Kettler A, Heuer F et al (2007) Intradiscal pressure, shear strain, and fiber strain in the intervertebral disc under combined loading. Spine 32:748–755
Acknowledgments
We thank our colleagues at the European Space Agency, Dr. Oliver Angerer and Dr. Jennifer Ngo-An, for facilitating our discussions. DLB thanks Dr. Zully Ritter for assistance in generating Fig. 2. We acknowledge the assistance of our colleagues at the European Astronaut Centre for clarifying our questions regarding astronaut training and care. The “European Space Agency Topical Team: Intervertebral Disc Herniations in Astronauts” was supported by contract number 4000110441/14/NL/PG from the European Space Agency. ARH was supported by NASA grant NNX13AM89G.
Conflict of interest
All authors have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Belavy, D.L., Adams, M., Brisby, H. et al. Disc herniations in astronauts: What causes them, and what does it tell us about herniation on earth?. Eur Spine J 25, 144–154 (2016). https://doi.org/10.1007/s00586-015-3917-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-015-3917-y