Abstract
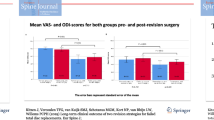
Low back pain (LBP) due to degenerative disc disease (DDD) is a common condition that can be treated along a continuum of care: from conservative therapies to several surgical choices. Nucleus arthroplasty is an emerging technology that could potentially fill part of the gap in the spine continuum of care. The introduction of recent technologies that allow the replacement of the degenerated disc nucleus using prosthetic devices may be considered an additional therapeutic tool that can be used by the surgeon in selected cases of LBP due to DDD. Nucleus arthroplasties are designed to treat early stages of DDD, which are one of the most common spinal disorders in the population under 65 years of age. NUBAC™ is the first articulating nucleus disc prosthesis, designed to optimally respect the lumbar anatomy, kinematics, and biomechanics, constructed in unique two-piece manufactured from polyetheretherketone (PEEK) with an inner ball/socket articulation. The optimal indications for NUBAC™ implantation are: disc height >5 mm, degenerative disc changes at an early stage (Pfirmann 2, 3), single level affection, integrity of posterior facet joints, lack of local anatomical contraindication, failure of conservative treatment for at least 6 months. From December 2006 to January 2009, a total of 39 patients underwent nucleus disc arthroplasty with NUBAC™ device. 22 cases have 2-year follow up. There have been no major intra-operative or post-operative vascular or neurological complications in this series. The data showed that there were significant decreases in both Visual Analogue Scale (VAS) and Oswestry Disability Index (ODI) after the procedure, with a meaningful improvement of symptoms in all patients. Although preliminary, the initial results are encouraging. The absence of any major intra-operative and post-operative complications supports the design rationale of the NUBAC™, being less invasive comparing to total disc replacement (TDA) and with a low rate of surgical risk. The effectiveness of data as seen in 2-year follow-up on both VAS and ODI have also suggested that the NUBAC™ could be considered a viable treatment option for patients with LBP caused by DDD.




Similar content being viewed by others
References
Kuslich SD, Ulstrom CL, Griffith SL, Ahern JW, Dowdle JD (1998) The Bagby and Kuslich method of lumbar interbody fusion. History, techniques, and 2-year follow-up results of a United States prospective, multicenter trial. Spine 23:1267–1279
Lehman TR, Spratt KF, Tozzi JE (1987) Long-term follow-up of lower lumbar fusion patients. Spine 12:97–104
Lemaire JP, Skalli W, Lavaste F (1997) Intervertebral disc prosthesis. Results and prospects for the year 2000. Clin Orthop 337:64–76
Enker P, Steffee AD (1997) Total disc replacement. In: Bridwell KH, DeWald RL (eds) The textbook of spinal surgery, 2nd edn. Lippincott-Raven, Philadelphia, pp 2275–2288
Hsu KY, Zucherman J, White A, Reynolds J, Goldwaite N (1988) Deterioration of motion segments adjacent to lumbar spine fusions. Trans N Am Spine Soc 12:605–606
Rahm MD, Hall BH (1996) Adjacent-segment degeneration after lumbar fusion with instrumentation: a retrospective study. J Spinal Disord 9:392–400
Bao QB, Yuan HA (2002) New technologies in spine: nucleus replacement. Spine 27:1245–1247
Bao QB, Yuan HA (2002) Prosthetic disc replacement: the future? Clin Orthop 394:139–145
Bao QB, Songer M, Pimenta L, Werner D, Reyes-Sanchez A, Balsano M, Agrillo U, Coric D, Davenport K, Yuan H (2007) Nubac disc artyhroplasty: preclinical studies and preliminary safety and efficacy evaluations. SAS J 1:36–45
Bertagnoli R, Vazquez RJ (2003) The anterolateral transpsoatic approach (ALPA). A new technique for implanting prosthetic disc nucleus devices. J Spinal Disord 16:398–404
Hedman TP, Kostuik JP, Fernie GR, Hellier WG (1991) Design of an intervertebral disc prosthesis. Spine 16(Suppl 6):S256–S260
Enker P, Steffee A, Mcmillan C, Keppler L, Biscup R, Miller S (1993) Artificial disc replacement. Preliminary report with a 3-year minimum follow-up. Spine 18:1061–1070
Fernstrom U (1966) Arthroplasty with intercorporal endoprothesis in herniated disc and in painful disc. Acta Chir Scand (Suppl) 357:154–159
Kostuik JP (1997) Intervertebral disc replacement. In: Bridwell KH, DeWald RL (eds) The textbook of spinal surgery, 2nd edn. Lippincott-Raven, Philadelphia, pp 2257–2266
Rothman RH, Simeone FA, Bernini PM (1982) Lumbar disc disease. In: Rothman RH, Simeone FA (eds) The spine, 2nd edn. WB Saunders, Philadelphia, pp 508–645
Weinstein JN (ed) (1992) Clinical efficacy and outcome in the diagnosis and treatment of low back pain. Raven Press, New York
Lee CK (1988) Accelerated degeneration of the segment adjacent to a lumbar fusion. Spine 13:375–377
Ray CD (1997) Threaded titanium cages for lumbar interbody fusions. Spine 22:667–680
Lee CK, Langrana NA, Parsons JR, Zimmerman MC (1991) Development of a prosthetic intervertebral disc. Spine 16(Suppl 6):S253–S255
Nachemson AL (1992) Challenge of the artificial disc. In: Weinstein JN (ed) Clinical efficacy and outcome in the diagnosis and treatment of low back pain. Raven Press, New York
Ray CD, Schönmayr R, Kavanagh SA, Assell R (1999) Prosthetic disc nucleus implants. Riv Neuroradiol 12(Suppl 1):157–162
Schönmayr R, Busch C, Lotz C, Lotz-Metz G (1999) Prosthetic disc nucleus implants: the Wiesbaden feasibility study. 2 years follow-up in ten patients. Riv Neuroradiol 12(Suppl 1):163–170
Ozgur BM, Aryan HE, Pimenta L, Taylor WR (2006) Extreme lateral interbody fusion (XLIF): a novel surgical technique for anterior lumbar interbody fusion. Spine J 6:435–443
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Balsano, M., Zachos, A., Ruggiu, A. et al. Nucleus disc arthroplasty with the NUBAC™ device: 2-year clinical experience. Eur Spine J 20 (Suppl 1), 36–40 (2011). https://doi.org/10.1007/s00586-011-1752-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-011-1752-3