Abstract
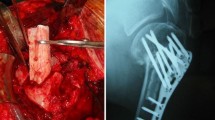
The objective of this study was to establish the efficacy and safety of porous bioactive titanium metal for use in a spinal fusion device, based on a prospective human clinical trial. A high-strength spinal interbody fusion device was manufactured from porous titanium metal. A bioactive surface was produced by simple chemical and thermal treatment. Five patients with unstable lumbar spine disease were treated surgically using this device in a clinical trial approved by our Ethics Review Committee and the University Hospital Medical Information Network. Clinical and radiological results were reported at the minimum follow-up period of 1 year. The optimal mechanical strength and interconnected structure of the porous titanium metal were adjusted for the device. The whole surface of porous titanium metal was treated uniformly and its bioactive ability was confirmed before clinical use. Successful bony union was achieved in all cases within 6 months without the need for autologous iliac crest bone grafting. Two specific findings including an anchoring effect and gap filling were evident radiologically. All clinical parameters improved significantly after the operation and no adverse effects were encountered during the follow-up period. Although a larger and longer-term follow-up clinical study is mandatory to reach any firm conclusions, the study results show that this porous bioactive titanium metal is promising material for a spinal fusion device.






Similar content being viewed by others
References
Banwart JC, Asher MA, Hassanein RS (1995) Iliac crest bone graft harvest donor site morbidity. A statistical evaluation. Spine 20:1055–1060
Bloebaum RD, Beeks D, Dorr LD, Savory CG, DuPont J, Hofmann AA (1994) Complications with hydroxyapatite particulate separation in total hip arthroplasty. Clin Orthop 298:19–26
Boden SD, Zdeblick TA, Sandu HS, Heim SE (2000) The use of rhBMP-2 in interbody fusion cages: definitive evidence of osteoinduction in humans: a preliminary report. Spine 25:376–381
Brantigan JW, Cunningham BW, Warden K, McAfee PC, Steffee AD (1993) Compression strength of donor bone for posterior lumbar interbody fusion. Spine 18:1213–1221
De Groot K, Geesink R, Klein CP, Serekian P (1987) Plasma sprayed coatings of hydroxyapatite. J Biomed Mater Res 21:1375–1381
Desogus N, Ennas F, Leuze R et al (2005) Posterior lumbar interbody fusion with PEEK cages: personal experience with 20 patients. J Neurosurg Sci 49:137–141
Ducheyne P, Qiu Q (1999) Bioactive ceramics: the effect of surface reactivity on bone formation and bone cell function. Biomaterials 20:2287–2303
Fujibayashi S, Shikata J, Tanaka C, Matsushita M, Nakamura T (2001) Lumbar posterolateral fusion with biphasic calcium phosphate ceramic. J Spinal Disord 14:214–221
Fujibayashi S, Nakamura T, Nishiguchi S, Tamura J, Uchida M, Kim HM et al (2001) Bioactive titanium: effect of sodium removal on the bone-bonding ability of bioactive titanium prepared by alkali and heat treatment. J Biomed Mater Res 56:562–570
Fujibayashi S, Neo M, Kim HM, Kokubo T, Nakamura T (2004) Osteoinduction of porous bioactive titanium metal. Biomaterials 25:443–450
Fujibayashi S, Neo M, Takemoto M, Ota M, Nakamura T (2010) Paraspinal-approach transforaminal lumbar interbody fusion for the treatment of lumbar foraminal stenosis. J Neurosurg Spine 13:500–508
Hench LL (1998) Bioactive materials: the potential for tissue regeneration. J Biomed Mater Res 41:511–518
Jost B, Cripton PA, Lund T, Oxland TR, Lippuner K, Jaeger P et al (1998) Compressive strength of interbody cages in lumbar spine: the effect of cage shape, posterior instrumentation and bone density. Eur Spine J 7:132–141
Kawanabe K, Ise K, Goto K, Akiyama H, Nakamura T, Kaneuji A et al (2009) A new cementless total hip arthroplasty with bioactive titanium porous-coating by alkaline and heat treatment: average 4.8-year results. J Biomed Mater Res Part B: Appl Biomater 90B:476–481
Kim HM, Kokubo T, Fujibayashi S, Nishiguchi S, Nakamura T (2000) Bioactive macroporous titanium surface layer on titanium substrate. J Biomed Mater Res 52:553–557
Kokubo T (1991) Bioactive glass ceramics: properties and applications. Biomaterials 12:155–163
Kokubo T, Kushitani H, Sakka S, Kitsugi T, Yamamuro T (1990) Solutions able to reproduce in vivo surface-structure changes in bioactive glass-ceramic A-W. J Biomed Mater Res 24:721–734
Kokubo T, Miyaji F, Kim HM, Nakamura T (1996) Spontaneous formation of bonelike apatite layer on chemically treated titanium metals. J Am Ceram Soc 79:1127–1129
McClellan JW, Mulconrey DS, Forbes RJ, Fullmer N (2006) Vertebral bone resorption after transforaminal lumbar interbody fusion with bone morphogenetic protein (rhBMP-2). J Spinal Disord Tech 19:483–486
Modic MT, Steinberg PM, Ross JS, Masaryk TJ, Carter JR (1988) Degenerative disk disease: assessment of changes in vertebral body marrow with MR imaging. Radiology 166:193–199
Morscher EW, Hefti A, Aebi U (1998) Severe osteolysis after third body wear due to hydroxyapatite particles from acetabular cup coating. J Bone Joint Surg Br 80:267–272
Nachemson AL (1981) Disc pressure measurements. Spine 6:93–97
Otsuki B, Takemoto M, Fujibayashi S, Neo M, Kokubo T, Nakamura T (2006) Pore throat size and connectivity determine bone and tissue ingrowth into porous implants: three-dimensional micro-CT based structural analyses of porous bioactive titanium implants. Biomaterials 27:5892–5900
Takemoto M, Fujibayashi S, Neo M, Suzuki J, Kokubo T, Nakamura T (2005) Mechanical properties and osteoconductivity of porous bioactive titanium. Biomaterials 26:6014–6023
Takemoto M, Fujibayashi S, Neo M, Suzuki J, Matsushita T, Kokubo T, Nakamura T (2006) Osteoinductive porous titanium implants: effect of sodium removal by dilute HCl treatment. Biomaterials 27:2682–2691
Takemoto M, Fujibayashi S, Neo M, So K, Akiyama N, Matsushita T et al (2007) A porous bioactive titanium implant for spinal interbody fusion: an experimental study using a canine model. J Neurosurg Spine 7:435–443
Toth JM, Boden SD, Burkus JK MD, Badura JM, Peckham SM, McKay WF (2009) Short-term osteoclastic activity induced by locally high concentrations of recombinant human bone morphogenetic protein-2 in a cancellous bone environment. Spine 34:539–550
Tullberg T, Brandt B, Rydberg J, Fritzell P (1996) Fusion rate after posterior lumbar interbody fusion with carbon fiber implant: 1-year follow-up of 51 patients. Eur Spine J 5:178–182
Tullberg T (1998) Failure of a carbon fiber implant: a case report. Spine 23:1804–1806
Vaccaro AR, Lawrence JP, Patel T, Katz LD, Anderson DG, Fischgrund JS et al (2008) The safety and efficacy of OP-1 (rhBMP-7) as a replacement for iliac crest autograft in posterolateral lumbar arthrodesis. Spine 33:2850–2862
Vaidya R, Sethi A, Bartol S, Jacobson M, Coe C, Craig JG (2008) Complications in the use of rhBMP-2 in PEEK cages for interbody spinal fusions. J Spinal Disord Tech 21:557–562
Wen CE, Mabuchi M, Yamada Y, Shimojima K, Chino Y, Asahina T (2001) Processing of biocompatible porous Ti and Mg. Scripta Mater 45:1147–1153
Acknowledgments
The authors thank Hisashi Kitagaki, Tsuneo Teraoka, of Osaka Yakin Co., for manufacturing and providing the porous titanium implants. They thank Seiji Yamaguchi, of Chubu University Biomedical Sciences, for treating the material chemically. They thank Shuji Higuchi, Masanori Fukushima, Satoshi Teramukai, Kenichi Yoshimura, Toshinori Murayama, Tomoko Yokota, Erika Hirata and Harue Tada, of Kyoto University’s translational research center, for help in protocol preparation, moderation of the clinical trial and data management. They also thank Takeshi Sakamoto, Makoto Yoshida and Masahiko Miyata for radiological assessments, and Masato Ota for surgical assistance.
This study was supported by a Grant in Aid for Scientific Research from the Japan Society for the Promotion of Science (No. 19200039). No benefits in any form have been or will be received from a commercial party related directly or indirectly to the subject of this manuscript. This manuscript has not been previously published and is not under consideration for publication elsewhere. The first two authors contributed equally to this study and preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fujibayashi, S., Takemoto, M., Neo, M. et al. A novel synthetic material for spinal fusion: a prospective clinical trial of porous bioactive titanium metal for lumbar interbody fusion. Eur Spine J 20, 1486–1495 (2011). https://doi.org/10.1007/s00586-011-1728-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-011-1728-3