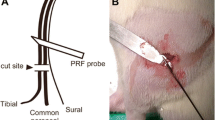
Abstract
Pathomechanisms of injured-nerve pain have not been fully elucidated. Radicular pain and chronic constriction injury models have been established; however, producing these models is complicated. A sciatic nerve-pinch injury is easy to produce but the reliability of this model for evaluating pain behavior has not been examined. The current study evaluated pain-related behavior and change in pain markers in the dorsal root ganglion (DRG) of rats in a simple, sciatic nerve-pinch injury model. In the model, the sciatic nerve was pinched for 2 s using forceps (n = 20), but not injured in sham-operated animals (n = 20). Mechanical and thermal hyperalgesia were measured every second day for 2 weeks using von Frey filaments and a Hargreaves device. Calcitonin gene-related peptide (CGRP), activating transcription factor-3 (ATF-3), phosphorylated p38 mitogen activated protein (Map) kinase (p-p38), and nuclear factor-kappa B (NF-κB; p65) expression in L5 DRGs were examined at 4 and 7 days after surgery using immunohistochemistry. The proportion of neurons immunoreactive for these markers was compared between the two groups. Mechanical (during 8 days) and thermal hyperalgesia (during 6 days) were found in the pinch group rats, but not in the sham-operated animals (p < 0.05); however, hyperalgesia was not significant from days 10 to 14. CGRP, ATF-3, p-p38, and NF-κB expression in L5 DRGs was upregulated in the nerve-injured rats compared with the sham-operated rats (p < 0.01). Our results indicate that a simple sciatic nerve pinch produced pain-related behavior. Upregulation of the pain-marker expression in the nerve-injury model suggested it could be used as a model of pain. However, it was not considered as suitable for long-term studies.







Similar content being viewed by others
References
Averill S, Michael GJ, Shortland PJ, Leavesley RC, King VR, Bradbury EJ, McMahon SB, Priestley JV (2004) NGF and GDNF ameliorate the increase in ATF3 expression which occurs in dorsal root ganglion cells in response to peripheral nerve injury. Eur J Neurosci 19:1437–1445
Baeuerle PA, Baltimore D (1996) NF-κB: ten years after. Cell 87:13–20
Barnes PJ, Karin M (1997) Nuclear factor-κB—a pivotal transcription factor in chronic inflammatory diseases. N Engl J Med 336:1066–1071
Bennett GJ (1993) An animal model of neuropathic pain: a review. Muscle Nerve 16:1040–1048
Bester H, Beggs S, Woolf CJ (2000) Changes in tactile stimuli-induced behavior and c-Fos expression in the superficial dorsal horn and in parabrachial nuclei after sciatic nerve pinch. J Comp Neurol 428:45–61
Chan CF, Sun WZ, Lin JK, Lin-Shiau SY (2000) Activation of transcription factors of nuclear factor kappa B, activator protein-1 and octamer factors in hyperalgesia. Eur J Pharmacol 402:61–68
Delcroix JD, Averill S, Fernandes K, Tomlinson DR, Priestley JV, Fernyhough P (1999) Axonal transport of activating transcription factor-2 is modulated by nerve growth factor in nociceptive neurons. J Neurosci RC 24:1–7
George A, Buehl A, Sommer C (2004) Wallerian degeneration after pinch injury of rat sciatic nerve increases endo- and epineurial tumor necrosis factor-alpha protein. Neurosci Lett 372:215–219
Hai T, Hartman MG (2001) The molecular biology and nomenclature of the activating transcription factor/cAMP responsive element binding family of transcription factors: activating transcription factor proteins and homeostasis. Gene 273:1–11
Hökfelt T (1991) Neuropeptides in perspective: the last ten years. Neuron 7:867–879
Inoue G, Ohtori S, Aoki Y, Ozawa T, Doya H, Saito T, Ito T, Akazawa T, Moriya H, Takahashi K (2006) Exposure of the nucleus pulposus to the outside of the anulus fibrosus induces nerve injury and regeneration of the afferent fibers innervating the lumbar intervertebral discs in rats. Spine 31:1433–1438
Ito T, Ohtori S, Inoue G, Koshi T, Doya H, Ozawa T, Saito T, Moriya H, Takahashi K (2007) Glial phosphorylated p38 MAP kinase mediates pain in a rat model of lumbar disc herniation and induces motor dysfunction in a rat model of lumbar spinal canal stenosis. Spine 32:159–167
Ji RR, Samad TA, Jin SX, Schmoll R, Woolf CJ (2002) p38 MAPK activation by NGF in primary sensory neurons after inflammation increases TRPV1 levels and maintains heat hyperalgesia. Neuron 36:57–68
Kato K, Kikuchi S, Shubayev VI, Myers RR (2009) Distribution and tumor necrosis factor-alpha isoform binding specificity of locally administered etanercept into injured and uninjured rat sciatic nerve. Neurosci 160:492–500
Kawakami M, Tamaki T, Weinstein JN, Hashizume H, Nishi H, Meller ST (1996) Pathomechanism of pain-related behavior produced by allografts of intervertebral disc in the rat. Spine 21:2101–2107
Kim SH, Chung JM (1992) An experimental model for peripheral neuropathy produced by segmental spinal nerve ligation in the rat. Pain 50:355–363
Kim SY, Bae JC, Kim JY, Lee HL, Lee KM, Kim DS, Cho HJ (2002) Activation of p38MAP kinase in the rat dorsal root ganglia and spinal cord following peripheral inflammation and nerve injury. Neuroreport 13:2483–2486
Ma W, Bisby MA (1998) Increased activation of nuclear factor kappa B in rat lumbar dorsal root ganglion neurons following partial sciatic nerve injuries. Brain Res 797:243–254
McCarthy PW, Lawson SN (1990) Cell type and conduction velocity of rat primary sensory neurons with calcitonin gene-related peptide-like immunoreactivity. Neuroscience 34:623–632
Miki K, Fukuoka T, Tokunaga A, Noguchi K (1998) Calcitonin gene-related peptide increase in the rat spinal dorsal horn and dorsal column nucleus following peripheral nerve injury: upregulation in a subpopulation of primary afferent sensory neurons. Neuroscience 82:1243–1252
Mizushima T, Obata K, Yamanaka H, Dai Y, Fukuoka T, Tokunaga A, Mashimo T, Noguchi K (2005) Activation of p38 MAPK in primary afferent neurons by noxious stimulation and its involvement in the development of thermal hyperalgesia. Pain 113:51–60
Obata K, Yamanaka H, Fukuoka T, Yi D, Tokunaga A, Hashimoto N, Yoshikawa H, Noguchi K (2003) Contribution of injured and uninjured dorsal root ganglion neurons to pain behavior and the changes in gene expression following chronic constriction injury of the sciatic nerve in rats. Pain 101:65–77
Ohtori S, Takahashi K, Chiba T, Yamagata M, Sameda H, Moriya H (2001) Phenotypic inflammation switch in rats shown by calcitonin gene-related peptide immunoreactive dorsal root ganglion neurons innervating the lumbar facet joints. Spine 26:1009–1013
Seltzer Z, Dubner R, Shir Y (1990) A novel behavioral model of neuropathic pain disorders produced in rats by partial sciatic nerve injury. Pain 43:205–218
Suzuki M, Inoue G, Gemba T, Watanabe T, Ito T, Koshi T, Yamauchi K, Yamashita M, Orita S, Eguchi Y, Ochiai N, Kishida S, Takaso M, Aoki Y, Takahashi K, Ohtori S (2009) Nuclear factor-kappa B decoy suppresses nerve injury and improves mechanical allodynia and thermal hyperalgesia in a rat lumbar disc herniation model. Eur Spine J 18:1001–1007
Svensson CI, Marsala M, Westerlund A, Calcutt NA, Campana WM, Freshwater JD, Catalano R, Feng Y, Protter AA, Scott B, Yaksh TL (2003) Activation of p38 mitogen-activated protein kinase in spinal microglia is a critical link in inflammation-induced spinal pain processing. J Neurochem 86:1534–1544
Takasaki I, Andoh T, Shiraki K, Kuraishi Y (2000) Allodynia and hyperalgesia induced by herpes simplex virus type-1 infection in mice. Pain 86:95–101
Wang R, Guo W, Ossipov MH, Vanderah TW, Porreca F, Lai J (2003) Glial cell line-derived neurotrophic factor normalizes neurochemical changes in injured dorsal root ganglion neurons and prevents the expression of experimental neuropathic pain. Neuroscience 121:815–824
Widmann C, Gibson S, Jarpe MB, Johnson GL (1999) Mitogen-activated protein kinase: conservation of a three-kinase module from yeast to human. Physiol Rev 79:143–180
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hirose, K., Iwakura, N., Orita, S. et al. Evaluation of behavior and neuropeptide markers of pain in a simple, sciatic nerve-pinch pain model in rats. Eur Spine J 19, 1746–1752 (2010). https://doi.org/10.1007/s00586-010-1428-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-010-1428-4