Abstract
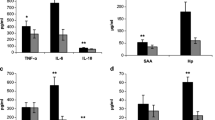
Monitoring of changes in acute phase proteins following the treatment of endometritis can help us to improve treatment protocols. In total, 81 dairy cows affected by clinical endometritis were selected in 30 days in milk (DIM) and treated with hyperimmune serum. Cervical mucus cytology was used to determine the neutrophil percentage. Levels of amyloid A (SAA) and haptoglobin (Hp) in blood serum were measured just before and 2 weeks after the treatment. The cows were timed-inseminated with Heat Synch following the treatment, and pregnancy was diagnosed 35 ± 3 days after AI. Variables such as parturition (abnormal and normal), 305 days milk production, pregnancy status, number of AI, endometritis score, days open, and parity were considered in statistical analysis. Concentrations of SAA and Hp were significantly lower after the treatment of cows affected by endometritis (P < 0.05). Pre- and post-treatment cows with < 9000kg milk production during 305 days had lower SAA than those cows with > 9000kg milk production. There was significantly higher Hp concentration in the pretreatment of cows that had been pregnant after the treatment of endometritis. Post-treatment cows with days open ≤ 100 days showed higher SAA than those cows with days open > 100 days. The results showed a positive significant correlation (r = 0.44, P = 0.02) between SAA and Hp in pre-treatment cows, but this correlation was not significant after the treatment (r = 0.01, P = 0.98). Treatment of cows affected by clinical endometritis with hyperimmune serum could decrease SAA and Hp, and factors such as milk production, pregnancy status, and days open affected their concentrations.


Similar content being viewed by others
References
Ahmadi M, Nazifi S, Ghaisari H (2006) Comparison of hormonal changes of estrous cycle with cytology of cervical mucosa and hematological parameters in dairy heifers. Comp Clin Pathol 15:94–97
Ahmadi MR, Hosseini A, Gheisari HR, Yavari M (2014) Preliminary trial in treatment of postpartum endometritis with intrauterine application of hyperimmune serum in dairy cows. Asian Pac J Trop Dis 4:S360–S365. https://doi.org/10.1016/S2222-1808(14)60471-0
Berkova N, Lemay A, Dresser DW, Fontaine JY, Kerizit J, Goupil S (2001) Haptoglobin is present in human endometrium and shows elevated levels in the decidua during pregnancy. Mol Hum Reprod 7:747–754
Beutler B, Hoebe K, Du X, Ulevitch RJ (2003) How we detect microbes and respond to them: the Toll-like receptors and their transducers. J Leukoc Biol 74:479–485. https://doi.org/10.1189/jlb.0203082
Biswal S, Das S, Balasubramanian S, Mohanty D, Sethy K, Dasgupta M (2014) Serum amyloid A and haptoglobin levels in crossbred cows with endometritis following different therapy. Vet World 7:1066–1070
Brodzki P, Kostro K, Brodzki A, Wawron W, Marczuk J, Kurek L (2015a) Inflammatory cytokines and acute-phase proteins concentrations in the peripheral blood and uterus of cows that developed endometritis during early postpartum. Theriogenology 84:11–18. https://doi.org/10.1016/j.theriogenology.2015.02.006
Brodzki P, Kostro K, Krakowski L, Marczuk J (2015b) Inflammatory cytokine and acute phase protein concentrations in the peripheral blood and uterine washings of cows with subclinical endometritis in the late postpartum period. Vet Res Commun 39:143–149. https://doi.org/10.1007/s11259-015-9635-4
Ceciliani F, Ceron JJ, Eckersall PD, Sauerwein H (2012) Acute phase proteins in ruminants. J Proteome 75:4207–4231. https://doi.org/10.1016/j.jprot.2012.04.004
Chan JP-W, Chu CC, Fung HP, Te Chuang S, Lin YC, Chu RM, Lee SL (2004) Serum haptoglobin concentration in cattle. J Vet Med Sci 66:43–46
Chan JP, Chang CC, Hsu WL, Liu WB, Chen TH (2010) Association of increased serum acute-phase protein concentrations with reproductive performance in dairy cows with postpartum metritis. Vet Clin Pathol 39:72–78. https://doi.org/10.1111/j.1939-165X.2009.00182.x
Dubuc J, Duffield TF, Leslie KE, Walton JS, LeBlanc SJ (2010) Risk factors for postpartum uterine diseases in dairy cows. J Dairy Sci 93:5764–5771. https://doi.org/10.3168/jds.2010-3429
Fischer C, Drillich M, Odau S, Heuwieser W, Einspanier R, Gabler C (2010) Selected pro-inflammatory factor transcripts in bovine endometrial epithelial cells are regulated during the oestrous cycle and elevated in case of subclinical or clinical endometritis. Reprod Fertil Dev 22:818–829. https://doi.org/10.1071/RD09120
Friendly M (1991) SAS system for statistical graphics. First Edition. SAS Publishing SAS Publishing
Galvão K et al (2010) Association between uterine disease and indicators of neutrophil and systemic energy status in lactating Holstein cows. J Dairy Sci 93:2926–2937
Galvão KN, Santos NR, Galvão JS, Gilbert RO (2011) Association between endometritis and endometrial cytokine expression in postpartum Holstein cows. Theriogenology 76:290–299. https://doi.org/10.1016/j.theriogenology.2011.02.006
Ghasemi F, Gonzalez-Cano P, Griebel PJ, Palmer C (2012) Proinflammatory cytokine gene expression in endometrial cytobrush samples harvested from cows with and without subclinical endometritis. Theriogenology 78:1538–1547. https://doi.org/10.1016/j.theriogenology.2012.06.022
Hirvonen J, Huszenicza G, Kulcsar M, Pyörälä S (1999) Acute-phase response in dairy cows with acute postpartum metritis. Theriogenology 51:1071–1083
Hoffman LH, Winfrey VP, Blaeuer GL, Olson GE (1996) A haptoglobin-like glycoprotein is produced by implantation-stage rabbit endometrium. Biol Reprod 55:176–184
Humblet MF, Guyot H, Boudry B, Mbayahi F, Hanzen C, Rollin F, Godeau JM (2006) Relationship between haptoglobin, serum amyloid A, and clinical status in a survey of dairy herds during a 6-month period. Vet Clin Pathol 35:188–193
Huzzey J, Duffield T, LeBlanc S, Veira D, Weary D, Von Keyserlingk M (2009) Haptoglobin as an early indicator of metritis. J Dairy Sci 92:621–625
Ishikawa Y, Nakada K, Hagiwara K, Kirisawa R, Iwai H, Moriyoshi M, Sawamukai Y (2004) Changes in interleukin-6 concentration in peripheral blood of pre- and post-partum dairy cattle and its relationship to postpartum reproductive diseases. J Vet Med Sci 66:1403–1408. https://doi.org/10.1292/jvms.66.1403
Jain N (1986) Schalm’s veterinary haematology. Lea and Febiger, Philadelphia
Jeremejeva J, Orro T, Kask K (2015) Relationship between acute phase proteins and subsequent fertility of dairy cows after postpartum uterine inflammation and healthy cows. Vet Med Zoot 70:37–41
Kasimanickam RK, Kasimanickam VR, Olsen JR, Jeffress EJ, Moore DA, Kastelic JP (2013) Associations among serum pro- and anti-inflammatory cytokines, metabolic mediators, body condition, and uterine disease in postpartum dairy cows. Reprod Biol Endocrinol 11:103. https://doi.org/10.1186/1477-7827-11-103
Kaya S, Merhan O, Kacar C, Colak A, Bozukluhan K (2016) Determination of ceruloplasmin, some other acute phase proteins, and biochemical parameters in cows with endometritis. Vet World 9:1056–1062. https://doi.org/10.14202/vetworld.2016.1056-1062
Kim I-H, Kang H-G, Jeong J-K, Hur T-Y, Jung Y-H (2014) Inflammatory cytokine concentrations in uterine flush and serum samples from dairy cows with clinical or subclinical endometritis. Theriogenology 82:427–432. https://doi.org/10.1016/j.theriogenology.2014.04.022
Lavery K, Gabler C, Day J, Killian G (2004) Expression of haptoglobin mRNA in the liver and oviduct during the oestrous cycle of cows (Bos taurus). Anim Reprod Sci 84:13–26. https://doi.org/10.1016/j.anireprosci.2003.12.010
LeBlanc SJ (2008) Postpartum uterine disease and dairy herd reproductive performance: a review. Vet J 176:102–114
Markandeya N, Deshmukh V (1995) A study on antibiotic sensitivity pattern of bacterial isolates from repeat breeding cows. Int J Anim Sci 10:337–338
Pancarci SM, Jordan ER, Risco C, Schouten MJ (2002) Use of estradiol cypionate in a presynchronized timed artificial insemination program for lactating dairy cattle. J Dairy Sci 85:122–131
Petersen H, Nielsen J, Heegaard P (2004) Application of acute phase protein measurements in veterinary clinical chemistry. Vet Res 35:163–187
Schneider A, Corrêa M, Butler W (2013) Acute phase proteins in Holstein cows diagnosed with uterine infection. Res Vet Sci 95:269–271
Sheldon IM, Williams EJ, Miller AN, Nash DM, Herath S (2008) Uterine diseases in cattle after parturition. Vet J 176:115–121. https://doi.org/10.1016/j.tvjl.2007.12.031
Sheldon IM, Cronin J, Goetze L, Donofrio G, Schuberth HJ (2009) Defining postpartum uterine disease and the mechanisms of infection and immunity in the female reproductive tract in cattle. Biol Reprod 81:1025–1032. https://doi.org/10.1095/biolreprod.109.077370
Singh J, Murray RD, Mshelia G, Woldehiwet Z (2008) The immune status of the bovine uterus during the peripartum period. Vet J 175:301–309. https://doi.org/10.1016/j.tvjl.2007.02.003
Tóthová C, Nagy O, Seidel H, Konvičná J, Farkašová Z, Kováč G (2008) Acute phase proteins and variables of protein metabolism in dairy cows during the pre-and postpartal period. Acta Vet Brno 77:51–57
Tothova C, Nagy O, Kovac G (2014) Acute phase proteins and their use in the diagnosis of diseases in ruminants: a review. Vet Med 59:163–180
Turner ML, Healey GD, Sheldon IM (2012) Immunity and inflammation in the uterus. Reprod Domest Anim 47(Suppl 4):402–409. https://doi.org/10.1111/j.1439-0531.2012.02104.x
Wira CR, Fahey JV (2004) The innate immune system: gatekeeper to the female reproductive tract. Immunology 111:13–15
Acknowledgments
The authors would like to thank the Research Council of Shiraz University and School of Veterinary Medicine, Shiraz University, for financial and technical support of this study (grant no. 71-GR-VT-5).
Funding
This study was supported by School of Veterinary Medicine, Shiraz University (grant number 94GCU2M154630).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Rights and permissions
About this article
Cite this article
Ahmadi, M.R., Mogheiseh, A. & Nazifi, S. Changes in biomarkers serum amyloid A and haptoglobin following treatment of endometritis in dairy cows. Comp Clin Pathol 27, 1659–1665 (2018). https://doi.org/10.1007/s00580-018-2790-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-018-2790-6