Abstract
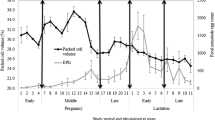
The main objective of the present study was to evaluate the biochemical profile of the Lacaune ewes naturally infected by gastrointestinal parasites in the pre- and post-partum period. Ewes with similar age and weight, primiparas in the final stage of gestation, totaling 24 animals were selected. Sampling days were 10 and 2 for pre-partum and 2, 6, and 12 days for post-partum to evaluate total proteins, albumin, globulin, cholesterol, triglycerides, and urea levels, in addition to thiobarbituric acid reactive substances (TBARS), reactive oxygen species (ROS), and glutathione S-transferase (GST). Fecal examination by McMaster method was performed during bleeding, and based on the number of eggs per gram of feces (EPG), the animals were divided into two groups: A (sheep with EPG <1000) and B (sheep with EPG >1000) to evaluate the effect of the parasitic load. Statistical analyses of biochemical parameters and EPG were performed taking into account groups, evaluated each time of sampling and over time, as well as without considering the groups, comparing pre- and post-partum and over time. It was possible to verify that the influence of time had a significant (P < 0.01) impact on cholesterol, triglycerides, total proteins, albumin, globulin, TBARS, and GST specifically among sampling time. When time of collection was considered, there were some differences between groups A and B for the variables: triglycerides (day 6 post-partum) and globulin (day 2 pre-partum). It is concluded that the parasitic load did influence seric levels of the urea, triglycerides, and globulins on a determined time.




Similar content being viewed by others
References
Ali SF, LeBel CP, Bondy SC (1992) Reactive oxygen species formation as a biomarker of methylmercury and trimethyltin neurotoxicity. Neurotoxicol 113:637–648
Bass DA, Parce JW, Dechatelet LR, Szejda P, Seeds MC, Thomas M (1983) Flow cytometric studies of oxidative product formation by neurotrophils: a graded response to membrane stimulation. J Immunol 1130:1910–1917
Bomfim MAD, Barros NN (2015) Nutrição de Cabras e Ovelhas no Pré e Pós-Parto. Embrapa caprinos. http://ainfo.cnptia.embrapa.br/digital/bitstream/item/35242/1/AAC-Nutricao-de-cabras-e-ovelhas-no-pre-e-pos-parto.pdf.
Brito MA, Diaz Gonzalez FH, Ribeiro LAO, Campos R, Lacerda LA, Barbosa PR, Bergmann GP (2006) Composição do sangue e do leite em ovinos leiteiros do sul do Brasil: variações na gestação e lactação. Ciênc Rural 36:942–948
Carvalho CTG (2013) Perfil metabólico e desempenho produtivo de ovelhas Santa Inês suplementadas com ionóforo durante o período de transição. Programa de Pós-graduação em Zootecnia, São Cristovão
Duffield TF, Rabiee AR, Lean IJ (2008) A meta-analysis of the impacto f monensinin lactating dairy cattle. Part2. Production effects. J Dairy Sci 91:1347–1360
Enevoldsen C, Kristensen T (1997) Estimation of body weight from body size measurements and body condition scores in dairy cows. J Ani Sci 80:1988–1995
Feijó JO, Perazzoli D, Silva LGC, Aragão RB, Martins CF, Pereira RA, Ferreira MB, Pino FABD, Rabassa VR, Corrêa MN (2014) Avaliação de parâmetros bioquímicos clínicos de ovelhas do grupo genético pantaneiro gestantes e não gestantes. Braz J Vet Res Anim Sci 51:156–165
Freeman BA, Crapo JD (1982) Biology of disease: free radicals and tissue injury. Lab Investig 47:412–426
Gonzales FHD, Campos R (2003) In: Gonzales FHD, Campos R (eds) Indicadores metabólico-nutricional do leite. Símposio de Patologia Clínica Veterinária da Região Sul do Brasil, Porto Alegre, pp 31–47
González FHD, Scheffer JFS (2002) Perfil sanguíneo: ferramenta de análise clínica, metabólica e nutricional. http://hdl.handle.net/10183/13177
González FHD, Silva SC (2006) Introdução à bioquímica clínica veterinária. Editora UFRGS, Porto Alegre, p 357
Gordon HM, Whitlock HV (1939) A new technique for counting nematode eggs in sheep faeces. J Commnw Sci 12:50–52
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione S-transferases. The first enzymatic step in mercapturic acid formation. J Biol Chem 249:7130–7139
Hoffman PC, Esser NM, Bauman LM, Denzine SL, Engstrom M, Chester-Jones H (2001) Effect of dietary protein on growth and nitrogen balance of Holstein heifers. J Dairy Sci 84:843–847
Instituto Brasileiro De Geografia E Estatística (2014) IBGE. Pesquisa pecuária municipal. http://www.ibge.gov.br.
Jentzsch AM, Bachmann H, Furst P, Biesalski H (1996) Improved analysis of malondialdehyde in human body fluids. Free Rad Biol Med 20:251–256
Machado V, Da Silva AS, Schafer AS, Aires AR, Tonin AA, Oliveira CB, Hermes CL, Almeida TC, Moresco RN, Stefani LM, Lopes ST, Mendes RE, Schwertz CI, Leal ML (2014) Relationship between oxidative stress and pathological findings in abomasum of infected lambs by Haemonchus contortus. Pathol Res Pract 210:812–817
Madureira KM, Gomes V, Barcelos B, Zani BH, Shecaira CL, Costa CR, Benesi FJ (2013) Parâmetros hematológicos e bioquímicos de ovinos da raça Dorper. Semina: Cienc Agric 34:811–816
Moallem U, Rozov A, Gootwine E, Honig H (2012) Plasma concentrations of key metabolites and insulin in latepregnantewes carrying 1 to 5 fetuses. J Anim Sci 90:318–324
Nascimento PM, Morgado AA, Nunes GR, Nikolaus JP, Weigel RA, Lima AS, Storilho VM, Mori CS, Sucupira MCA (2014) Metabolismo oxidativo e perfil bioquímico de ovelhas santa inês no período periparto: efeito da suplementação parenteral com vitamina E. Semina: Cienc Agric 36:1397–1408
Pereira JV (1998) Bioquímica Clínica. Editora Universitária UFPB, João Pessoa
Perry BD, Mcdermott JJ, Randolph TF, Sones KR, Thornton PK (2002) Investing in animal health research to alleviate poverty. Nairobi, Kenya. Int Livest Res Instig 148:1–56
Pinheiro AC (1982) Verminose ovina. In: Curso sobre parasitoses dos ruminates. Florianópolis, Medicina Veterinaria, pp 61–75
Ramos JJ, Verde MT, Marca MC, Fermindez A (1994) Clinical chemical values and variations in Rasa Aragonesa ewes and lambs. Small Rum Res 13:133–139
Roberts FHS, O’sullivan PJ (1950) Methods for egg counts and larval cultures for Strongyles infesting the gastrointestinal tract of cattle. Aust J Agric Res 1:99–102
Russel AJF (1991) Nutrition of pregnant ewe. In: Boden D (ed) Sheep and goat practice. Baillière Trindall, London, pp 29–39
Santos CM (2013) Influência da raça e das fases reprodutivas no parasitismo gastrintestinal em matrizes ovinas. (Dissertação). Mestrado em Zootecnia, centro de ciências agrárias e biológicas, Universidade Estadual Vale do Acaraú, Sobral, 58f
Schalm OW (1961) Antioxidant-induced stress. Int J Mol Sci 13:2091–2109
Silva M, Lima WG, Silva ME, Pedrosa ML (2011) Efeito da estreptozotocina sobre os perfis glicêmico e lipídico e o estresse oxidativo em hamsters. Arq Bras Endocrinol Metab 55:1–8
Vieira LS (2005). Endoparasitoses gastrintestinais em caprinos e ovinos. Embrapa Caprinos. http://people.ufpr.br/∼freitasjaf/artigoscaprinos/endoparasitasgastrointestinais.pdf
Vieira LS (2008) Métodos alternativos de controle de nematóides gastrintestinais em caprinos e ovinos. Tec Cie Agrop 2:49–56
Acknowledgments
The authors thank the farm “Cabanha Chapecó” for providing animals for research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Committee
The experiment was approved by the Ethics Committee on Animal Research of Universidade do Estado de Santa Catarina (UDESC) under protocol number 1.28.15.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Balzan, A., Machado, G., Bottari, N.B. et al. Pre- and post-partum seric biochemical variables of Lacaune ewes naturally infected by gastrointestinal parasites. Comp Clin Pathol 25, 815–823 (2016). https://doi.org/10.1007/s00580-016-2268-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-016-2268-3