Abstract
Purpose
There have been no large-scale studies on whether metformin therapy might have a potential benefit for lowering mortality. Thus, this study aimed to investigate the association between prior metformin therapy and the development of sepsis as well as the association between prior metformin therapy and 30-day mortality in sepsis patients.
Methods
We evaluated adult diabetes patients registered in the 2010 sample cohort database of the National Health Insurance Service in South Korea. Diabetes was identified according to the International Classification of Disease-10 diagnostic system (E10–E14). The cohorts were divided into the metformin user group (i.e., those who had been prescribed continuous oral metformin over a period of ≥ 90 days) and the control group (i.e., all other individuals). The primary endpoint was the development of sepsis between 2011 and 2015, and the secondary endpoint was 30-day mortality among diabetes patients diagnosed with sepsis.
Results
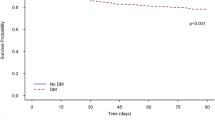
In total, 77,337 patients (34,041 in the metformin user group and 43,296 in the control group) were included in the analysis, among whom 2512 patients (3.2%) were diagnosed with sepsis between 2011 and 2015. After propensity score adjustment, metformin use was not significantly associated with both the risk of sepsis (OR: 0.92, 95%CI 0.82–1.03; P = 0.143) and the risk of 30-day mortality after diagnosis of sepsis (OR: 0.94, 95%CI 0.75–1.17; P = 0.571).
Conclusions
Prior metformin therapy was not significantly associated with the risk of sepsis and 30-day mortality after diagnosis of sepsis among diabetes patients.

Similar content being viewed by others
References
Shankar-Hari M, Phillips GS, Levy ML, Seymour CW, Liu VX, Deutschman CS, Angus DC, Rubenfeld GD, Singer M, Sepsis Definitions Task Force. Developing a new definition and assessing new clinical criteria for septic shock: for the third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA. 2016;315(8):775–87.
Rhee C, Dantes R, Epstein L, Murphy DJ, Seymour CW, Iwashyna TJ, Kadri SS, Angus DC, Danner RL, Fiore AE, Jernigan JA, Martin GS, Septimus E, Warren DK, Karcz A, Chan C, Menchaca JT, Wang R, Gruber S, Klompas M, Program CDCPE. Incidence and trends of sepsis in US hospitals using clinical vs claims data, 2009–2014. JAMA. 2017;318(13):1241–9.
Hajj J, Blaine N, Salavaci J, Jacoby D. The “Centrality of Sepsis”: a review on incidence, mortality, and cost of care. Healthcare (Basel). 2018;6(3).
Nguyen AT, Tsai CL, Hwang LY, Lai D, Markham C, Patel B. Obesity and mortality, length of stay and hospital cost among patients with sepsis: a nationwide inpatient retrospective cohort study. PLoS ONE. 2016;11(4):e0154599.
Page DB, Donnelly JP, Wang HE. Community-, healthcare-, and hospital-acquired severe sepsis hospitalizations in the university healthsystem consortium. Crit Care Med. 2015;43(9):1945–51.
Bailey CJ. Metformin: historical overview. Diabetologia. 2017;60(9):1566–76.
Kim SA, Choi HC. Metformin inhibits inflammatory response via AMPK-PTEN pathway in vascular smooth muscle cells. Biochem Biophys Res Commun. 2012;425(4):866–72.
Kuo CL, Ho FM, Chang MY, Prakash E, Lin WW. Inhibition of lipopolysaccharide-induced inducible nitric oxide synthase and cyclooxygenase-2 gene expression by 5-aminoimidazole-4-carboxamide riboside is independent of AMP-activated protein kinase. J Cell Biochem. 2008;103(3):931–40.
Liu G, Wu K, Zhang L, Dai J, Huang W, Lin L, Ge P, Luo F, Lei H. Metformin attenuated endotoxin-induced acute myocarditis via activating AMPK. Int Immunopharmacol. 2017;47:166–72.
Yuan H, Li L, Zheng W, Wan J, Ge P, Li H, Zhang L. Antidiabetic drug metformin alleviates endotoxin-induced fulminant liver injury in mice. Int Immunopharmacol. 2012;12(4):682–8.
An H, He L. Current understanding of metformin effect on the control of hyperglycemia in diabetes. J Endocrinol. 2016;228(3):R97–106.
Escobar DA, Botero-Quintero AM, Kautza BC, Luciano J, Loughran P, Darwiche S, Rosengart MR, Zuckerbraun BS, Gomez H. Adenosine monophosphate-activated protein kinase activation protects against sepsis-induced organ injury and inflammation. J Surg Res. 2015;194(1):262–72.
Liu Z, Bone N, Jiang S, Park DW, Tadie JM, Deshane J, Rodriguez CA, Pittet JF, Abraham E, Zmijewski JW. AMP-activated protein kinase and glycogen synthase kinase 3beta modulate the severity of sepsis-induced lung injury. Mol Med. 2016;21(1):937–50.
Mulchandani N, Yang WL, Khan MM, Zhang F, Marambaud P, Nicastro J, Coppa GF, Wang P. Stimulation of brain AMP-activated protein kinase attenuates inflammation and acute lung injury in sepsis. Mol Med. 2015;21:637–44.
Liang H, Ding X, Li L, Wang T, Kan Q, Wang L, Sun T. Association of preadmission metformin use and mortality in patients with sepsis and diabetes mellitus: a systematic review and meta-analysis of cohort studies. Crit Care. 2019;23(1):50.
Doenyas-Barak K, Beberashvili I, Marcus R, Efrati S. Lactic acidosis and severe septic shock in metformin users: a cohort study. Crit Care. 2016;20:10.
Green JP, Berger T, Garg N, Suarez A, Hagar Y, Radeos MS, Panacek EA. Impact of metformin use on the prognostic value of lactate in sepsis. Am J Emerg Med. 2012;30(9):1667–733.
Jochmans S, Alphonsine JE, Chelly J, Vong LVP, Sy O, Rolin N, Ellrodt O, Monchi M, Vinsonneau C. Does metformin exposure before ICU stay have any impact on patients' outcome? A retrospective cohort study of diabetic patients. Ann Intensive Care. 2017;7(1):116.
Park J, Hwang SY, Jo IJ, Jeon K, Suh GY, Lee TR, Yoon H, Cha WC, Sim MS, Carriere KC, Yeon S, Shin TG. Impact of metformin use on lactate kinetics in patients with severe sepsis and septic shock. Shock. 2017;47(5):582–7.
van Vught LA, Scicluna BP, Hoogendijk AJ, Wiewel MA, Klein Klouwenberg PM, Cremer OL, Horn J, Nurnberg P, Bonten MM, Schultz MJ, van der Poll T. Association of diabetes and diabetes treatment with the host response in critically ill sepsis patients. Crit Care. 2016;20(1):252.
Lee J, Lee JS, Park SH, Shin SA, Kim K. Cohort profile: the national health insurance service-national sample cohort (NHIS-NSC), South Korea. Int J Epidemiol. 2017;46(2):e15.
Arfe A, Corrao G. The lag-time approach improved drug-outcome association estimates in presence of protopathic bias. J Clin Epidemiol. 2016;78:101–7.
Rosenbaum PR, Rubin DB. Reducing bias in observational studies using subclassification on the propensity score. J Am Stat Assoc. 1984;79(387):516–24.
Levesque LE, Hanley JA, Kezouh A, Suissa S. Problem of immortal time bias in cohort studies: example using statins for preventing progression of diabetes. BMJ. 2010;340:b5087.
Mi X, Hammill BG, Curtis LH, Lai EC, Setoguchi S. Use of the landmark method to address immortal person-time bias in comparative effectiveness research: a simulation study. Stat Med. 2016;35(26):4824–36.
Huang J, Liu K, Zhu S, Xie M, Kang R, Cao L, Tang D. AMPK regulates immunometabolism in sepsis. Brain Behav Immun. 2018;72:89–100.
Suissa S, Azoulay L. Metformin and the risk of cancer: time-related biases in observational studies. Diabetes Care. 2012;35(12):2665–733.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they do not have any conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
540_2020_2753_MOESM2_ESM.docx
e-Appendix 2. Logistic regression analysis for development of sepsis in PS-matched cohort according to survival time PS, propensity score (DOCX 13 kb)
About this article
Cite this article
Oh, T.K., Song, IA. Association between prior metformin therapy and sepsis in diabetes patients: a nationwide sample cohort study. J Anesth 34, 358–366 (2020). https://doi.org/10.1007/s00540-020-02753-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00540-020-02753-3