Abstract
Background
Pancreatic neuroendocrine tumors (PanNETs) are indolent pancreatic tumors derived from neuroendocrine cells in pancreatic islets. To date, reliable predictors for identifying patients at high risk for recurrence after curative cancer resection are lacking. We aimed to determine independent predictors for high-risk PanNETs and patient outcomes after surgery.
Methods
We analyzed relevant clinicopathological parameters in 319 consecutive patients of derivation cohort 1 and 106 patients of validation cohort 2 who underwent pancreatectomy and were diagnosed with PanNETs. Association of tumor characteristics with recurrence-free survival (RFS) and overall survival (OS) was evaluated using Cox regression.
Results
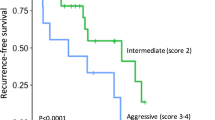
PanNET grade 3 (G3), pancreatic duct dilatation, and perineural invasion were independent prognostic factors for RFS and were significantly associated with early recurrence (within 1.5 years) of PanNETs after curative resection (P = 0.019, P < 0.001, and P < 0.001, respectively). Using these factors, we established a novel risk factor panel (R-panel), which predicted early recurrence (P < 0.001, HR = 15.02, 95% CI 5.76–39.19). Predictive accuracy of this R-panel was favorable, with a C-index of 0.853, higher than AJCC TNM staging (0.713). We further built an integrated staging system combining R-panel scoring and TNM staging, which improved predictive probability of TNM staging. Finally, we showed that adjuvant therapy with long-acting somatostatin analogs (SSAs) significantly reduced postoperative recurrence (P < 0.001) and prolonged long-term survival (P = 0.021) in patients with the above risk factors.
Conclusion
We identified a novel risk factor panel, which includes PanNET G3, pancreatic duct dilatation, and perineural invasion; this panel predicted early recurrence of PanNETs after curative resection. Patients with these risk factors can benefit from adjuvant therapy with SSAs.



Similar content being viewed by others
References
Rindi G, Klimstra DS, Abedi-Ardekani B, et al. A common classification framework for neuroendocrine neoplasms: an international agency for research on cancer (IARC) and world health organization (WHO) expert consensus proposal. Mod Pathol. 2018;31:1770–86.
Lloyd RV, Osamura R, Kloppel G, et al. WHO classification of tumours of endocrine organs. Lyon, France: IARC Press; 2017.
Cives M, Strosberg JR. Gastroenteropancreatic neuroendocrine tumors. CA Cancer J Clin. 2018;68:471–87.
Partelli S, Bartsch DK, Capdevila J, et al. ENETS consensus guidelines for standard of care in neuroendocrine tumours: surgery for small intestinal and pancreatic neuroendocrine tumours. Neuroendocrinology. 2017;105:255–65.
Luo G, Javed A, Strosberg JR, et al. Modified staging classification for pancreatic neuroendocrine tumors on the basis of the American joint committee on cancer and european neuroendocrine tumor society systems. J Clin Oncol. 2017;35:274–80.
Knigge U, Capdevila J, Bartsch DK, et al. ENETS consensus recommendations for the standards of care in neuroendocrine neoplasms: follow-up and documentation. Neuroendocrinology. 2017;105:310–9.
National comprehensive cancer network clinical practice guidelines in oncology (NCCN guidelines®). Neuroendocrine and adrenal tumors. Version 1.2019. http://www.nccn.org. Accessed 20 June 2020
Genc CG, Jilesen AP, Partelli S, et al. A new scoring system to predict recurrent disease in grade 1 and 2 nonfunctional pancreatic neuroendocrine tumors. Ann Surg. 2018;267:1148–54.
Sallinen VJ, Le Large TYS, Tieftrunk E, et al. Prognosis of sporadic resected small (≤ 2 cm) nonfunctional pancreatic neuroendocrine tumors—a multi-institutional study. HPB (Oxford). 2018;20:251–9.
Nanno Y, Matsumoto I, Zen Y, et al. Pancreatic duct involvement in well-differentiated neuroendocrine tumors is an independent poor prognostic factor. Ann Surg Oncol. 2017;24:1127–33.
Gao H, Liu L, Wang W, et al. Novel recurrence risk stratification of resected pancreatic neuroendocrine tumor. Cancer Lett. 2018;412:188–93.
Cai L, Michelakos T, Deshpande V, et al. Role of tumor-associated macrophages in the clinical course of pancreatic neuroendocrine tumors (PanNETs). Clin Cancer Res. 2019;25:2644–55.
Xu JZ, Wang WQ, Zhang SR, et al. Intrinsic contact between T and N classifications in resected well-moderately differentiated locoregional pancreatic neuroendocrine neoplasms. Ann Surg Oncol. 2018;25:647–54.
Amin MB, Edge SB, Greene FL, et al. AJCC cancer staging manual. 8th ed. New York: Springer; 2018.
WHO Classification of Tumours Editorial Board. Digestive system tumours, WHO classification of tumours. 5th ed. Lyon, France: IARC Press; 2019.
Tang LH, Basturk O, Sue JJ, et al. A practical approach to the classification of WHO grade 3 (G3) well-differentiated neuroendocrine tumor (WD-NET) and poorly differentiated neuroendocrine carcinoma (PD-NEC) of the pancreas. Am J Surg Pathol. 2016;40:1192–202.
Pavel M, Oberg K, Falconi M, et al. Gastroenteropancreatic neuroendocrine neoplasms: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2020;31:844–60.
Shrier I, Platt RW. Reducing bias through directed acyclic graphs. BMC Med Res Methodol. 2008;8:70.
Wang WQ, Liu L, Xu HX, et al. Infiltrating immune cells and gene mutations in pancreatic ductal adenocarcinoma. Br J Surg. 2016;103:1189–99.
Lee MR, Harris C, Baeg KJ, et al. Incidence trends of gastroenteropancreatic neuroendocrine tumors in the United States. Clin Gastroenterol Hepatol. 2018;17(11):2212–7.
Fan JH, Zhang YQ, Shi SS, et al. A nation-wide retrospective epidemiological study of gastroenteropancreatic neuroendocrine neoplasms in China. Oncotarget. 2017;8:71699–708.
Velayoudom-Cephise FL, Duvillard P, Foucan L, et al. Are G3 ENETS neuroendocrine neoplasms heterogeneous? Endocr Relat Cancer. 2013;20:649–57.
Basturk O, Yang Z, Tang LH, et al. The high-grade (WHO G3) pancreatic neuroendocrine tumor category is morphologically and biologically heterogenous and includes both well differentiated and poorly differentiated neoplasms. Am J Surg Pathol. 2015;39:683–90.
Fazio N, Milione M. Heterogeneity of grade 3 gastroenteropancreatic neuroendocrine carcinomas: new insights and treatment implications. Cancer Treat Rev. 2016;50:61–7.
Nunez-Valdovinos B, Carmona-Bayonas A, Jimenez-Fonseca P, et al. Neuroendocrine tumor heterogeneity adds uncertainty to the world health organization 2010 classification: real-world data from the spanish tumor registry (R-GETNE). Oncologist. 2018;23:422–32.
Rinke A, Gress TM. Neuroendocrine cancer, therapeutic strategies in G3 cancers. Digestion. 2017;95:109–14.
Bettini R, Partelli S, Boninsegna L, et al. Tumor size correlates with malignancy in nonfunctioning pancreatic endocrine tumor. Surgery. 2011;150:75–82.
Wong J, Fulp WJ, Strosberg JR, et al. Predictors of lymph node metastases and impact on survival in resected pancreatic neuroendocrine tumors: a single-center experience. Am J Surg. 2014;208:775–80.
Ausania F, Senra Del Rio P, Gomez-Bravo MA, et al. Can we predict recurrence in WHO G1–G2 pancreatic neuroendocrine neoplasms? Results from a multi-institutional Spanish study. Pancreatology. 2019;19:367–71.
Sennino B, Ishiguro-Oonuma T, Wei Y, et al. Suppression of tumor invasion and metastasis by concurrent inhibition of c-Met and VEGF signaling in pancreatic neuroendocrine tumors. Cancer Discov. 2012;2:270–87.
Kasajima A, Papotti M, Ito W, et al. High interlaboratory and interobserver agreement of somatostatin receptor immunohistochemical determination and correlation with response to somatostatin analogs. Hum Pathol. 2018;72:144–52.
Kaemmerer D, Peter L, Lupp A, et al. Molecular imaging with 68Ga-SSTR PET/CT and correlation to immunohistochemistry of somatostatin receptors in neuroendocrine tumours. Eur J Nucl Med Mol Imaging. 2011;38:1659–68.
Acknowledgements
The authors thank Prof. Jie Chen (Department of Gastroenterology, the First Affiliated Hospital, Sun Yat-sen University, Guangzhou, China), Prof. Cheng-Hao Shao (Department of Pancreatic-Biliary Surgery, Shanghai Changzheng Hospital, Second Military Medical University, Shanghai, China), and Prof. Jian Wang (Department of Biliary-Pancreatic Surgery, Renji Hospital, School of Medicine, Shanghai Jiao Tong University, Shanghai, China), three senior members of the Chinese Study Group for Neuroendocrine Tumor, for providing study materials. We also thank Dr. Kai-Zhou Jin, Ying Yang, and Ms. Huan-Yu Xia (Fudan University Shanghai Cancer Centre) for their assistance in collecting patient data. We thank Prof. Wei-Qi Sheng (Department of Pathology, Fudan University Shanghai Cancer Centre, China) and Prof. Hai-Ying Zeng (Department of Pathology, Zhongshan Hospital, Fudan University, China) for their assistance in the pathological diagnosis of PanNETs in all enrolled patients. Lastly, we thank Prof. Hai-Tao Chen (School of Public Health, Fudan University, China) and Dr. Miao Mo (Department of Clinical Statistics, Fudan University Shanghai Cancer Centre, China) for their assistance with statistical analyses.
Funding
This work was supported by grants from the National Science Foundation for Distinguished Young Scholars of China (81625016), the National Natural Science Foundation of China (81871941, 81872366, 81827807, 81802675, 81701630 and 81702341), the Outstanding Academic Leader Program of the “Technological Innovation Action Plan” in Shanghai Science and Technology Commission (18XD1401200), the Scientific Innovation Project of Shanghai Education Committee (2019-01-07-00-07-E00057), the Natural Science Foundation of Shanghai (19ZR1410800), Clinical and Scientific Innovation Project of Shanghai Hospital Development Center (SHDC12018109), and the Young Talented Specialist Training Program of Shanghai.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
None reported.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, WQ., Zhang, WH., Gao, HL. et al. A novel risk factor panel predicts early recurrence in resected pancreatic neuroendocrine tumors. J Gastroenterol 56, 395–405 (2021). https://doi.org/10.1007/s00535-021-01777-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-021-01777-0