Abstract
Background
The efficacy of neoadjuvant chemoradiotherapy (NACRT) and subset of pancreatic ductal adenocarcinoma (PDAC) patients who are most likely to benefit from this strategy remain elusive. The aim of this study was to investigate the effects of NACRT in patients with resectable (R) or borderline resectable (BR) adenocarcinoma of the pancreatic head. BR diseases were classified into two groups: lesions involving exclusively the portal vein system (BR-PV) and those abutting the major artery (BR-A).
Methods
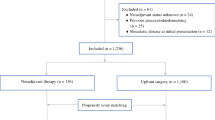
A total of 504 patients treated with curative intent for PDAC were analyzed (R, n = 273; BR-PV, n = 129; BR-A, n = 102). Patients who underwent upfront surgery and those who underwent NACRT followed by surgery were compared using propensity score-matched and inverse probability of treatment-weighted analyses (UMIN000019719).
Results
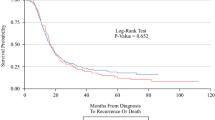
No significant differences were noted in the incidences of curative resection among the three categories (R, BR-PV and BR-A). Propensity score-weighted logistic regression analysis revealed that the incidence of pathologically positive resection margins was reduced by NACRT only for BR patients. Among the propensity score-matched patients, NACRT rather than upfront surgery significantly prolonged the median survival time of BR-PV patients (28.4 vs. 20.1 months; P = 0.044) but not that of R-PDAC patients (28.6 vs. 33.7 months; P = 0.960). NACRT prolonged the median survival time of BR-A patients (18.1 vs. 10.0 months; P = 0.046), but the results remained unsatisfactory.
Conclusions
These findings suggest that NACRT improves R0 rates and increases the survival of patients with BR-PV adenocarcinoma of the pancreatic head but not that of patients with R-PDAC.


Similar content being viewed by others
References
Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA Cancer J Clin. 2012;62:10–29. doi:10.3322/caac.20138.
Wray CJ, Ahmad SA, Matthews JB, et al. Surgery for pancreatic cancer: recent controversies and current practice. Gastroenterol. 2005;128:1626–41. doi:10.1053/j.gastro.2005.03.035.
Tempero MA, Malafa MP, Behrman SW, et al. Pancreatic adenocarcinoma, version 2.2014: featured updates to the NCCN guidelines. J Natl Compr Canc Netw. 2014;12:1083–93.
Talamonti MS, Small W Jr, Mulcahy MF, et al. A multi-institutional phase II trial of preoperative full-dose gemcitabine and concurrent radiation for patients with potentially resectable pancreatic carcinoma. Ann Surg Oncol. 2006;13:150–8. doi:10.1245/ASO.2006.03.039.
Pisters PW, Wolff RA, Janjan NA, et al. Preoperative paclitaxel and concurrent rapid-fractionation radiation for resectable pancreatic adenocarcinoma: toxicities, histologic response rates, and event-free outcome. J Clin Oncol. 2002;20:2537–44. doi:10.1200/JCO.2002.11.064.
Hoffman JP, Lipsitz S, Pisansky T, et al. Phase II trial of preoperative radiation therapy and chemotherapy for patients with localized, resectable adenocarcinoma of the pancreas: an Eastern Cooperative Oncology Group Study. J Clin Oncol. 1998;16:317–23.
Varadhachary GR, Wolff RA, Crane CH, et al. Preoperative gemcitabine and cisplatin followed by gemcitabine-based chemoradiation for resectable adenocarcinoma of the pancreatic head. J Clin Oncol. 2008;26:3487–95. doi:10.1200/JCO.2007.15.8642.
Evans DB, Varadhachary GR, Crane CH, et al. Preoperative gemcitabine-based chemoradiation for patients with resectable adenocarcinoma of the pancreatic head. J Clin Oncol. 2008;26:3496–502. doi:10.1200/JCO.2007.15.8634.
Katz MH, Pisters PW, Evans DB, et al. Borderline resectable pancreatic cancer: the importance of this emerging stage of disease. J Am Coll Surg. 2008;206:833–46. doi:10.1016/j.jamcollsurg.2007.12.020.
Golcher H, Brunner TB, Witzigmann H, et al. Neoadjuvant chemoradiation therapy with gemcitabine/cisplatin and surgery versus immediate surgery in resectable pancreatic cancer: results of the first prospective randomized phase II trial. Strahlenther Onkol. 2015;191:7–16. doi:10.1007/s00066-014-0737-7.
Casadei R, Di Marco M, Ricci C, et al. Neoadjuvant chemoradiotherapy and surgery versus surgery alone in resectable pancreatic cancer: a single-center prospective, randomized, Controlled Trial which failed to achieve accrual targets. J Gastrointest Surg. 2015;19:1802–12. doi:10.1007/s11605-015-2890-4.
National Comprehensive Cancer Network. Clinical practice guidelines in oncology (NCCN guidelines). Pancreatic adenocarcinoma (version 2.2015). http://www.nccn.org/professionals/physician_gls/pdf/pancreatic.pdf. Accessed 1 November 2015.
Fujii T, Yamada S, Murotani K, et al. Inverse probability of treatment weighting analysis of upfront surgery versus neoadjuvant chemoradiotherapy followed by surgery for pancreatic adenocarcinoma with arterial abutment. Med (Baltimore). 2015;94:e1647. doi:10.1097/MD.0000000000001647.
Ikeda M, Okusaka T, Ito Y, et al. A phase I trial of S-1 with concurrent radiotherapy for locally advanced pancreatic cancer. Br J Cancer. 2007;96:1650–5. doi:10.1038/sj.bjc.6603788.
Fujii T, Nakao A, Yamada S, et al. Vein resections >3 cm during pancreatectomy are associated with poor 1-year patency rates. Surgery. 2015;157:708–15. doi:10.1016/j.surg.2014.12.002.
Nakao A, Kanzaki A, Fujii T, et al. Correlation between radiographic classification and pathological grade of portal vein wall invasion in pancreatic head cancer. Ann Surg. 2012;255:103–8. doi:10.1097/SLA.0b013e318237872e.
Yamada S, Fujii T, Sugimoto H, et al. Aggressive surgery for borderline resectable pancreatic cancer: evaluation of National Comprehensive Cancer Network guidelines. Pancreas. 2013;42:1004–10. doi:10.1097/MPA.0b013e31827b2d7c.
Fujii T, Kanda M, Kodera Y, et al. Preservation of the pyloric ring has little value in surgery for pancreatic head cancer: a comparative study comparing three surgical procedures. Ann Surg Oncol. 2012;19:176–83. doi:10.1245/s10434-011-1901-2.
Kanda M, Fujii T, Kodera Y, et al. Nutritional predictors of postoperative outcome in pancreatic cancer. Br J Surg. 2011;98:268–74. doi:10.1002/bjs.7305.
Fujii T, Sugimoto H, Yamada S, et al. Modified Blumgart anastomosis for pancreaticojejunostomy: technical improvement in matched historical control study. J Gastrointest Surg. 2014;18:1108–15. doi:10.1007/s11605-014-2523-3.
Fujii T, Nakao A, Murotani K, et al. Influence of food intake on the healing process of postoperative pancreatic fistula after pancreatoduodenectomy: a multi-institutional randomized controlled trial. Ann Surg Oncol. 2015;22:3905–12. doi:10.1245/s10434-015-4496-1.
Satoi S, Toyokawa H, Yanagimoto H, et al. Neo-adjuvant chemoradiation therapy using S-1 followed by surgical resection in patients with pancreatic cancer. J Gastrointest Surg. 2012;16:784–92. doi:10.1007/s11605-011-1795-0.
Satoi S, Yanagimoto H, Toyokawa H, et al. Surgical results after preoperative chemoradiation therapy for patients with pancreatic cancer. Pancreas. 2009;38:282–8. doi:10.1097/MPA.0b013e31819438c3.
Oettle H, Neuhaus P, Hochhaus A, et al. Adjuvant chemotherapy with gemcitabine and long-term outcomes among patients with resected pancreatic cancer: the CONKO-001 randomized trial. JAMA. 2013;310:1473–81. doi:10.1001/jama.2013.279201.
Maeda A, Boku N, Fukutomi A, et al. Randomized phase III trial of adjuvant chemotherapy with gemcitabine versus S-1 in patients with resected pancreatic cancer: Japan Adjuvant Study Group of Pancreatic Cancer (JASPAC-01). Jpn J Clin Oncol. 2008;38:227–9. doi:10.1093/jjco/hym178.
Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205–13. doi:10.1097/01.sla.0000133083.54934.ae.
International Union Against Cancer. TNM classification of malignant tumors. New York: Wiley-Blackwell, 7th edn; 2009.
Staley CA, Cleary KR, Abbruzzese JL, et al. The need for standardized pathologic staging of pancreaticoduodenectomy specimens. Pancreas. 1996;12:373–80. doi:10.1097/00006676-199605000-00009.
Robins JM, Hernán MA, Brumback B. Marginal structural models and causal inference in epidemiology. Epidemiology. 2000;11:550–60. doi:10.1097/00001648-200009000-00011.
Bonjer HJ, Hop WC, Nelson H, et al. Laparoscopically assisted vs open colectomy for colon cancer: a meta-analysis. Arch Surg. 2007;142:298–303. doi:10.1001/archsurg.142.3.298.
Thorpe H, Jayne DG, Guillou PJ, et al. Patient factors influencing conversion from laparoscopically assisted to open surgery for colorectal cancer. Br J Surg. 2008;95:199–205. doi:10.1002/bjs.5907.
Gillen S, Schuster T, Meyer Zum Büschenfelde C, et al. Preoperative/neoadjuvant therapy in pancreatic cancer: a systematic review and meta-analysis of response and resection percentages. PLOS Med. 2010;7:e1000267. doi:10.1371/journal.pmed.1000267.
Abbott DE, Tzeng CW, Merkow RP, et al. The cost-effectiveness of neoadjuvant chemoradiation is superior to a surgery-first approach in the treatment of pancreatic head adenocarcinoma. Ann Surg Oncol. 2013;20(Suppl 3):S500–8. doi:10.1245/s10434-013-2882-0.
Franke AJ, Rosati LM, Pawlik TM, et al. The role of radiation therapy in pancreatic ductal adenocarcinoma in the neoadjuvant and adjuvant settings. Semin Oncol. 2015;42:144–62. doi:10.1053/j.seminoncol.2014.12.013.
Xu CP, Xue XJ, Liang N, et al. Effect of chemoradiotherapy and neoadjuvant chemoradiotherapy in resectable pancreatic cancer: a systematic review and meta-analysis. J Cancer Res Clin Oncol. 2014;140:549–59. doi:10.1007/s00432-013-1572-4.
Lee JH, Kang CM, Bang SM, et al. The role of neoadjuvant chemoradiation therapy in patients with borderline resectable pancreatic cancer with isolated venous vascular involvement. Med (Baltimore). 2015;94:e1233. doi:10.1097/MD.0000000000001233.
Sho M, Akahori T, Tanaka T, et al. Optimal indication of neoadjuvant chemoradiotherapy for pancreatic cancer. Langenbecks Arch Surg. 2015;400:477–85. doi:10.1007/s00423-015-1304-0.
Ishii H, Furuse J, Boku N, et al. Phase II study of gemcitabine chemotherapy alone for locally advanced pancreatic carcinoma: JCOG0506. Jpn J Clin Oncol. 2010;40:573–9. doi:10.1093/jjco/hyq011.
Ikeda M, Ioka T, Ito Y, et al. A multicenter phase II trial of S-1 with concurrent radiation therapy for locally advanced pancreatic cancer. Int J Radiat Oncol Biol Phys. 2013;85:163–9. doi:10.1016/j.ijrobp.2012.03.059.
Ferrone CR, Marchegiani G, Hong TS, et al. Radiological and surgical implications of neoadjuvant treatment with FOLFIRINOX for locally advanced and borderline resectable pancreatic cancer. Ann Surg. 2015;261:12–7. doi:10.1097/SLA.0000000000000867.
Paniccia A, Edil BH, Schulick RD, et al. Neoadjuvant FOLFIRINOX application in borderline resectable pancreatic adenocarcinoma: a retrospective cohort study. Med (Baltimore). 2014;93:e198. doi:10.1097/MD.0000000000000198.
Tokunaga J, Imanaka Y. Influence of length of stay on patient satisfaction with hospital care in Japan. Int J Qual Health Care. 2002;14:493–502. doi:10.1093/intqhc/14.6.493.
Kondo A, Zierler BK, Isokawa Y, et al. Comparison of outcomes and costs after hip fracture surgery in three hospitals that have different care systems in Japan. Health Policy. 2009;91:204–10. doi:10.1016/j.healthpol.2008.12.006.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
S. Satoi and K. Murotani contributed equally to this research.
Rights and permissions
About this article
Cite this article
Fujii, T., Satoi, S., Yamada, S. et al. Clinical benefits of neoadjuvant chemoradiotherapy for adenocarcinoma of the pancreatic head: an observational study using inverse probability of treatment weighting. J Gastroenterol 52, 81–93 (2017). https://doi.org/10.1007/s00535-016-1217-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-016-1217-x